CHEM 100 LEC: EXAM 4
1/72
Earn XP
Description and Tags
CHEM 100 LEC
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
True or False: Aromatic Hydrocarbons usually undergo Substitution Reaction
True
True or False: Aromatic Hydrocarbons usually undergo Addition Reaction
False
True or False: Aromatic Hydrocarbons always contain a Benzene Ring
True
True or False: Aromatic Hydrocarbons behave like Alkenes
False
What are Two Classes of Organic Compounds that could React Together to Form Esters?
Carboxylic acid and Alcohol
What is the Name of the Functional Group in Ketones?
Carbonyl
What is the Name of the Functional Group in Carboxylic Acids?
Carboxyl
What is an Alkane?
Saturated hydrocarbon with only single bonds
What is an Alkene?
Unsaturated hydrocarbon with at least one double bond
What is an Alkyne?
Unsaturated hydrocarbon with at least one triple bond
What is the General Formula for Alkanes?
CnH2n+2
What is the General Formula for Alkenes?
CnH2n
What is the General Formula for Alkynes?
CnH2n−2
Alcohol Functional Group?
C-O-H
Alcohol Suffix?
-ol
Ketone Suffix?
-one
Aldehyde Functional Group?
CHO
Aldehyde Suffix?
-anal
Carboxylic Acid Functional Group?
COOH
Carboxylic Acid suffix?
-oic acid
Ester Functional Group?
COO
Ester Suffix?
-oate
Ether Functional Group?
COC
Amine Functional Group?
NH2
Amine Suffix?
-amine
Alkene Functional Group?
C=C
Alkyne Functional Group?
C≡C
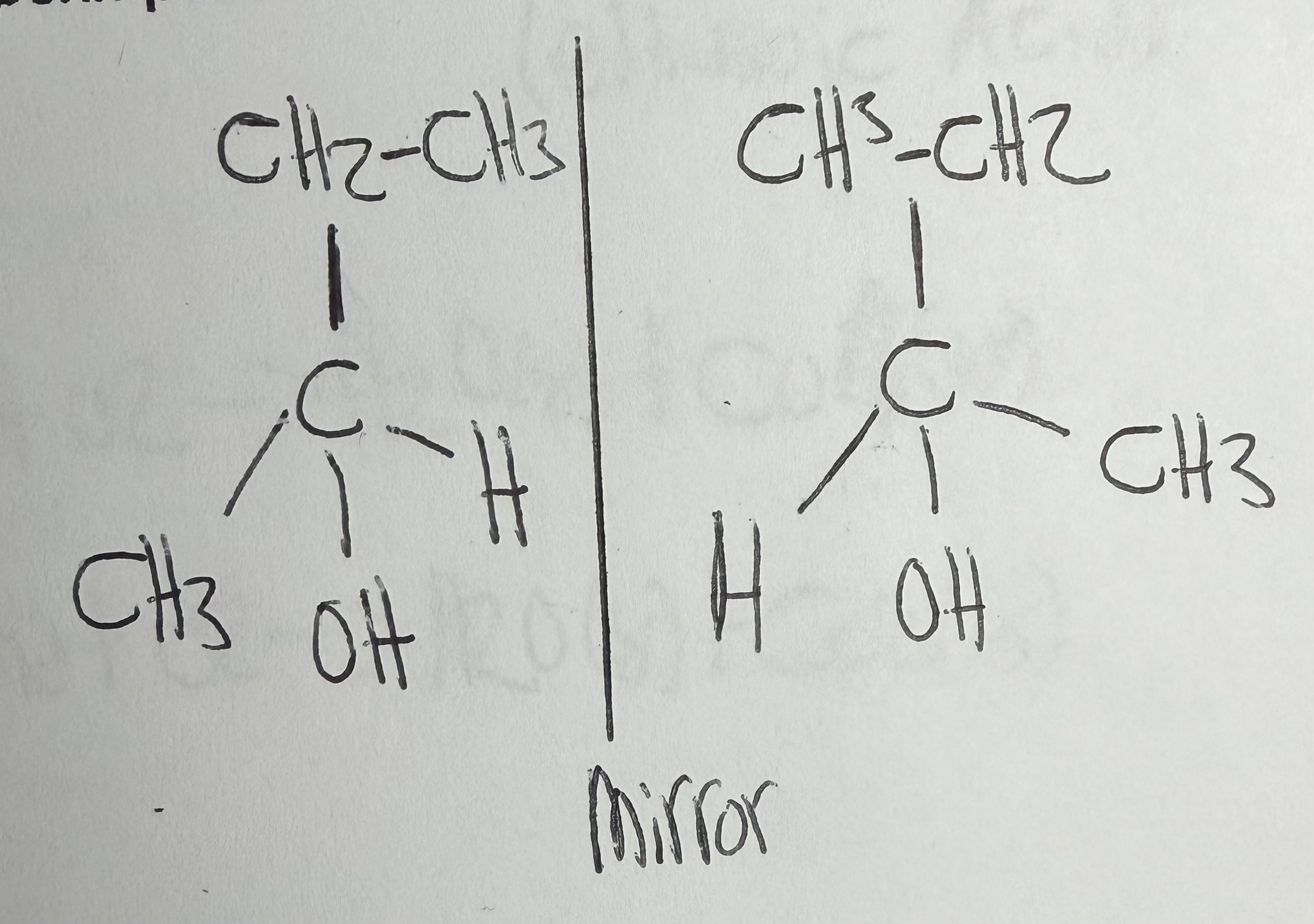
What is the Rule for Optical Isomerism?
A carbon must be attached to 4 different groups
Ketone Functional Group?
C=O in the middle
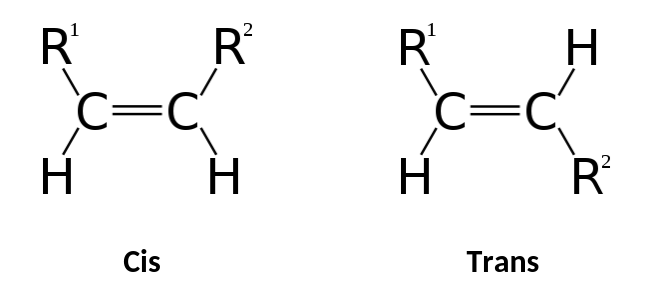
What is Cis?
Same side of a double bond
What is Trans?
Opposite sides of a double bond
What is the Difference between Cis and Trans?
Cis = Same side
Trans = Opposite side
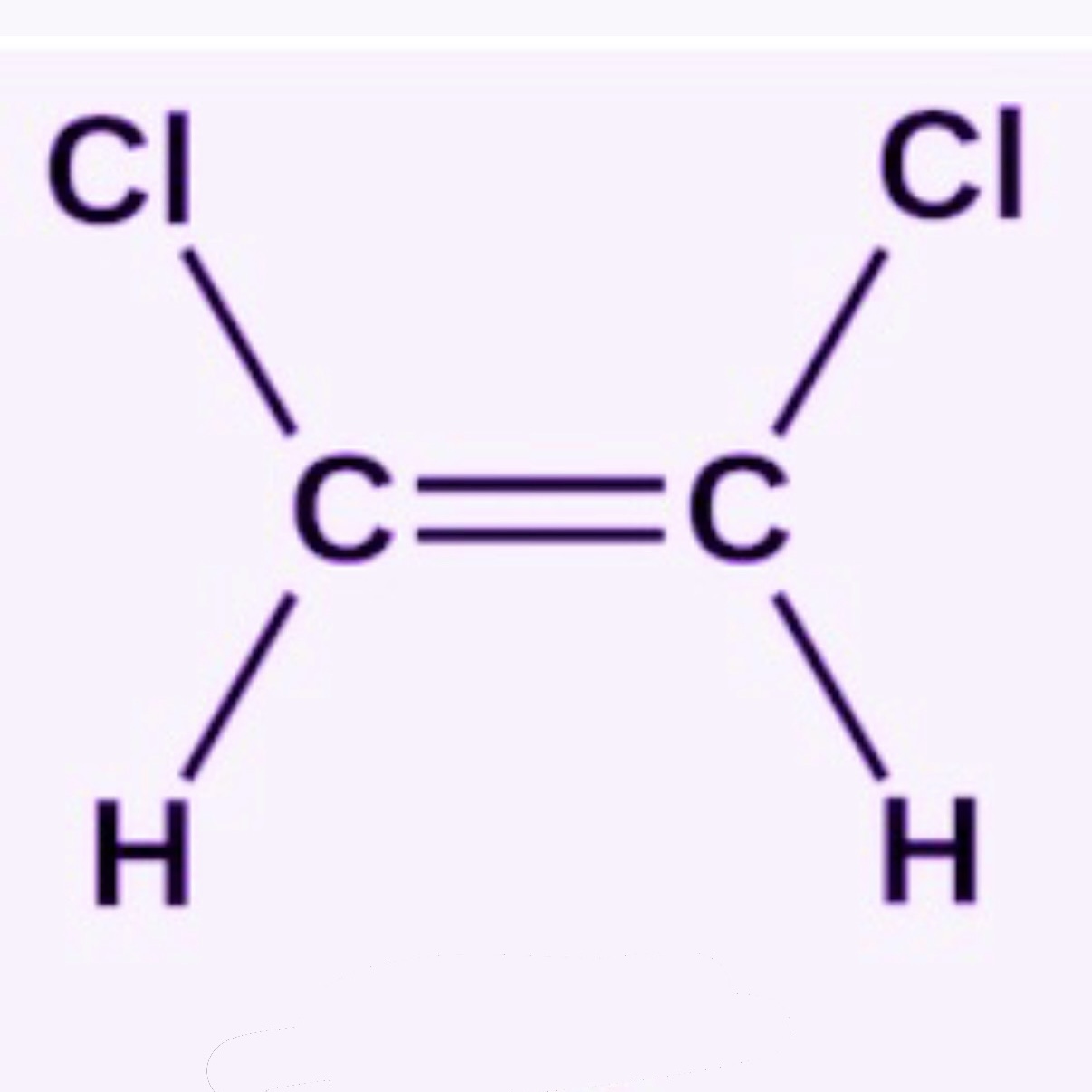
Cis
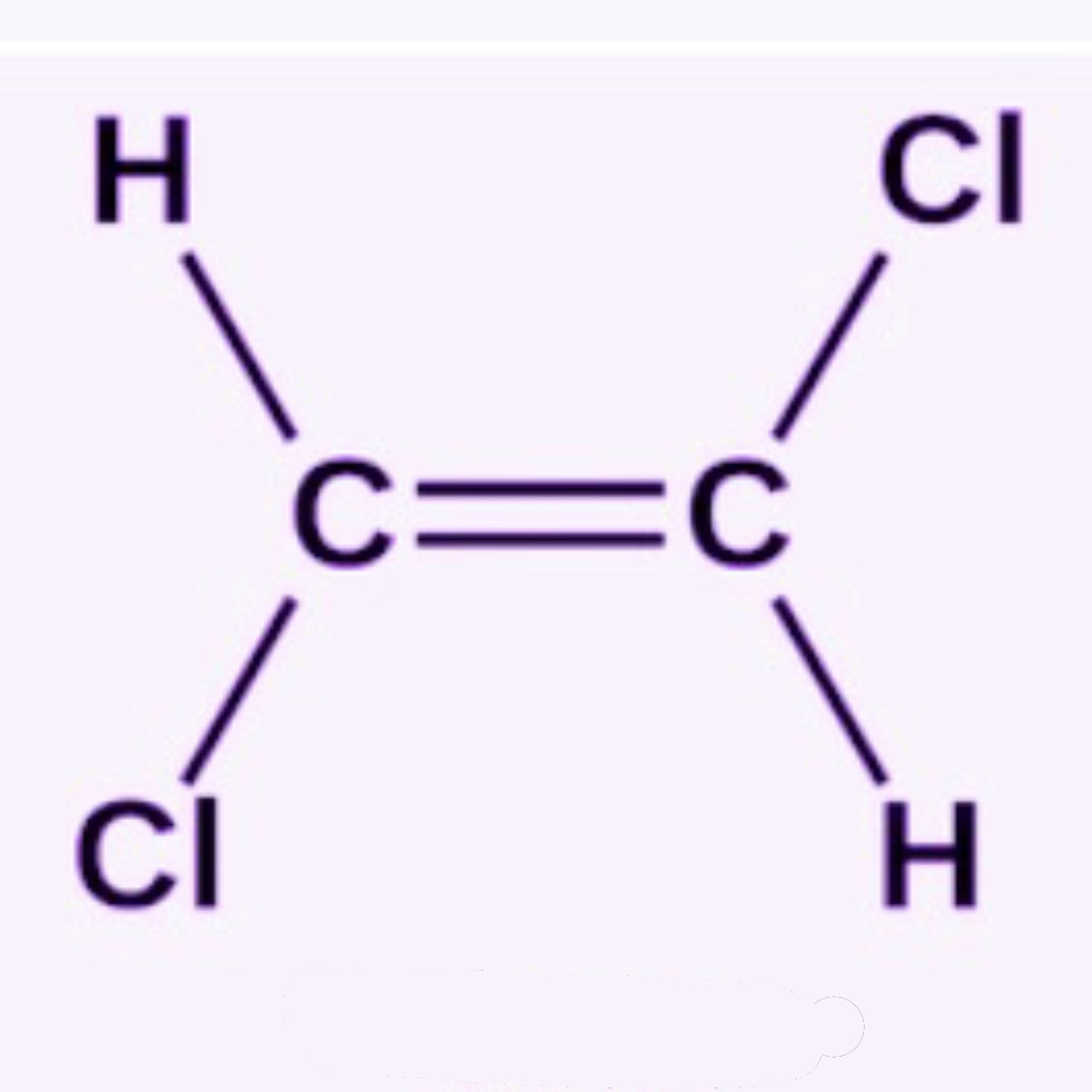
Trans
What is a Neutralization Reaction?
Acid + base reaction
What is a Condensation Reaction?
A reaction where two molecules join together and release water
What is a Hydrolysis Reaction?
A reaction where water breaks a molecule apart
What is an Addition Reaction?
A reaction where atoms add across a double bond
What is the IUPAC Naming Order?
Substituents (Ex. Methyl)
Parent Chain (Ex. Butan)
Functional Group (Ex. -one, ketone)
How many Bonds does Carbon usually Form?
4 bonds
How many Bonds does Hydrogen usually Form?
1 bond
How many Bonds does Oxygen usually Form?
2 bonds
How many Bonds does Sulfur usually Form?
2 bonds
How many bonds does Nitrogen usually Form?
3 bonds
How many Bonds do Halogens usually Form?
1 bond
What is the Name of the Functional Group in Alcohol?
Hydroxyl
What is the Name of the Functional Group in Aldehyde?
Carbonyl
What is the Name of the Functional Group in Amine?
Amino
Ether Suffix?
-oxy
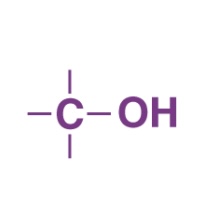
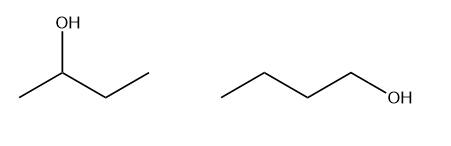
Primary Alcohol
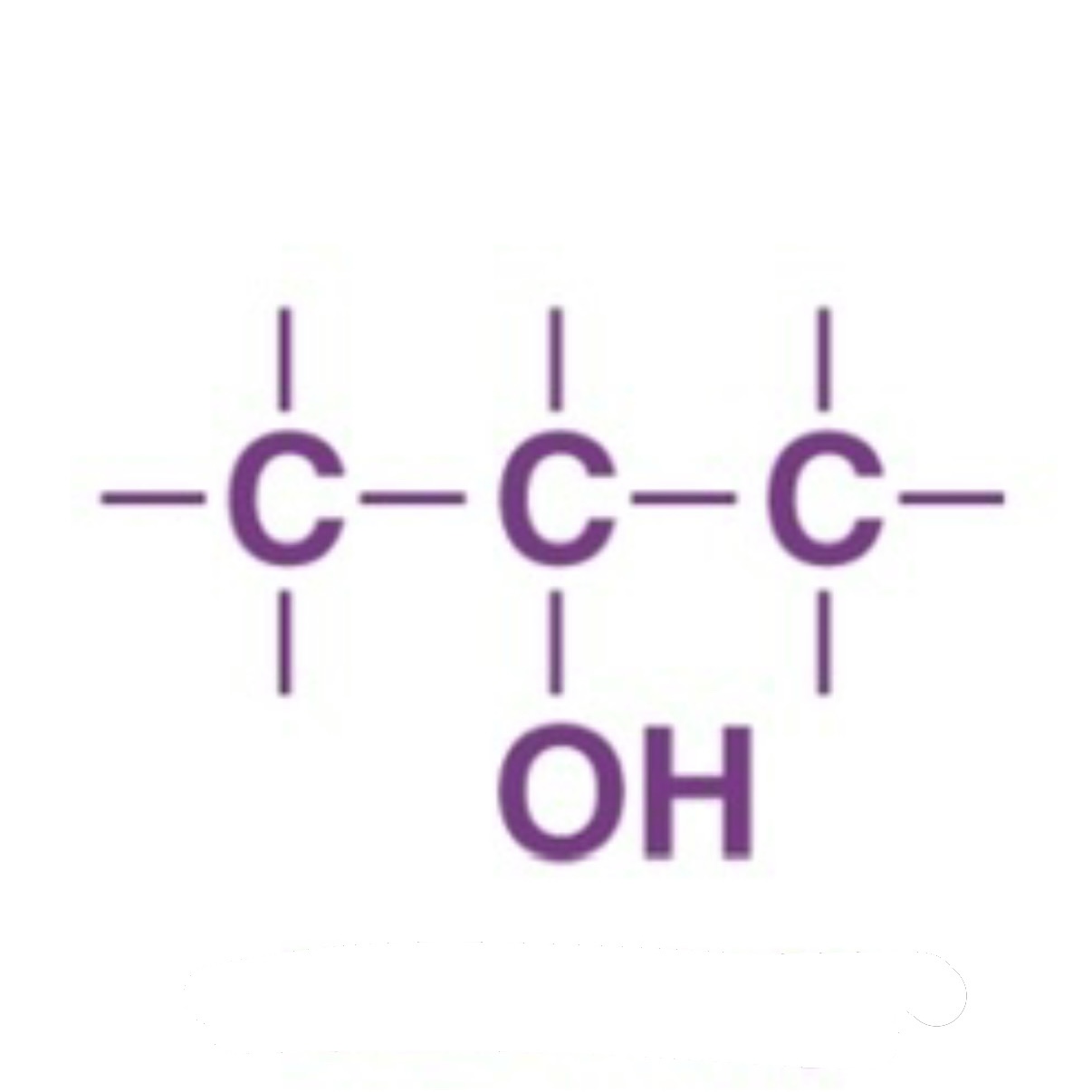
Secondary Alcohol
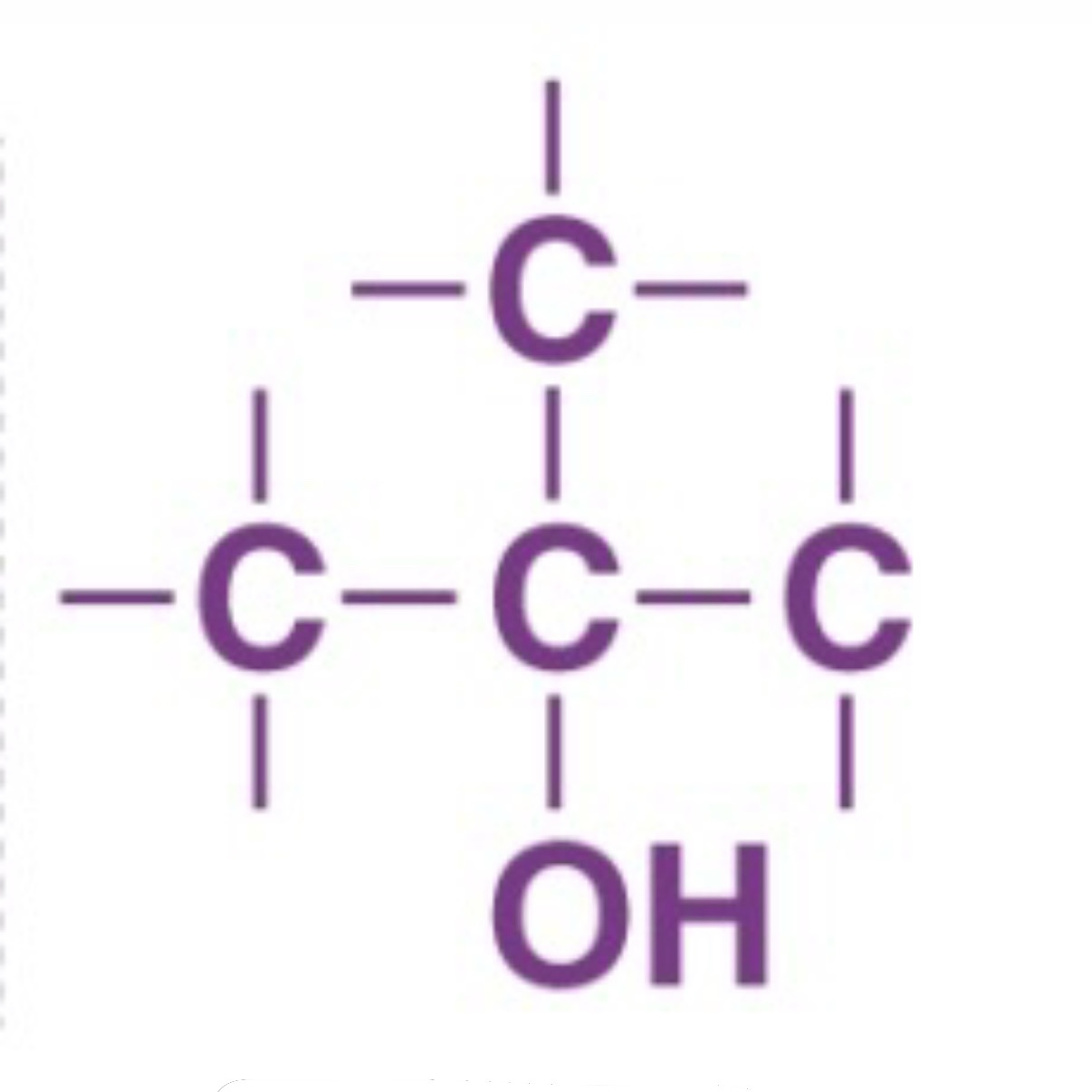
Tertiary Alcohol
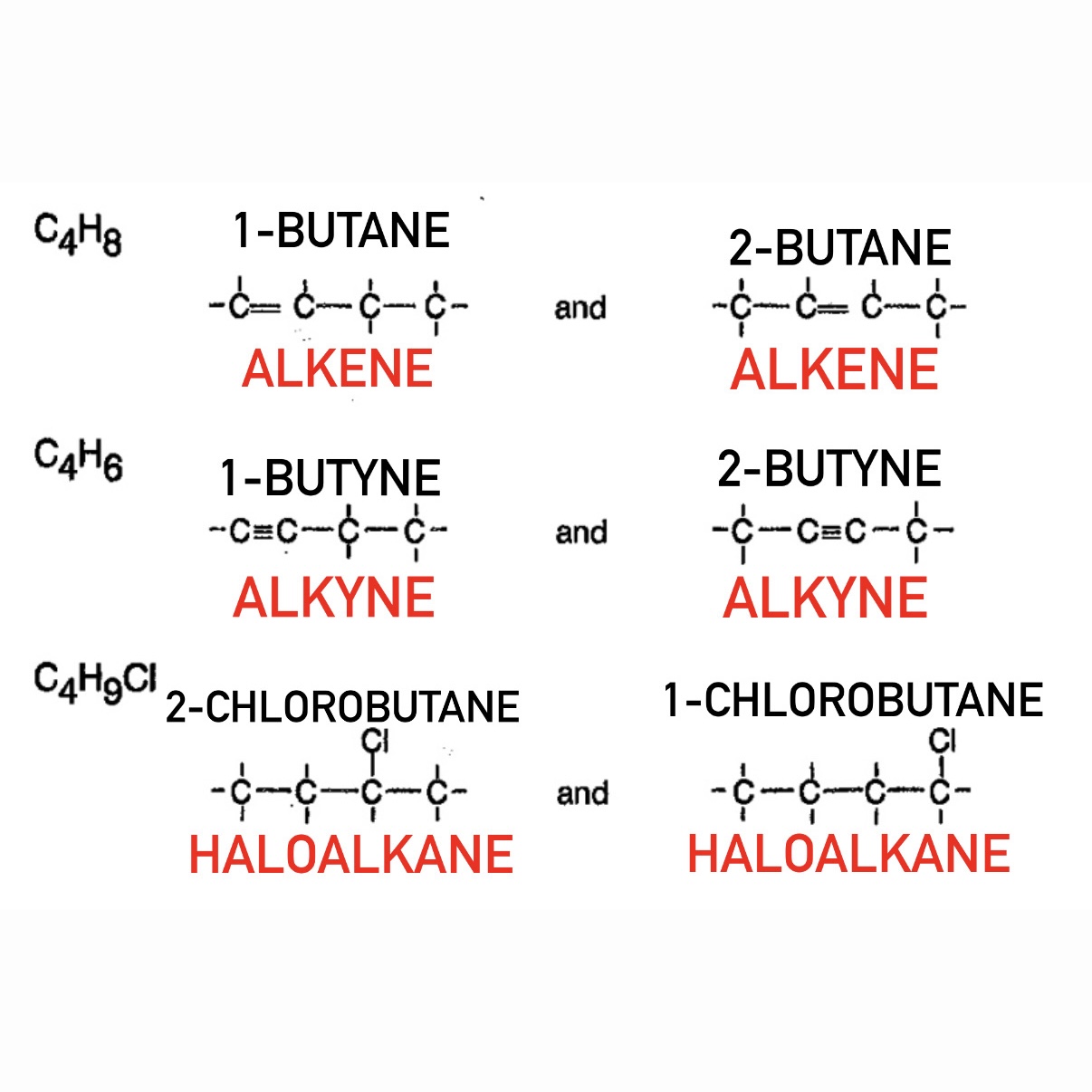
Skeletal Isomers
Positional Isomers
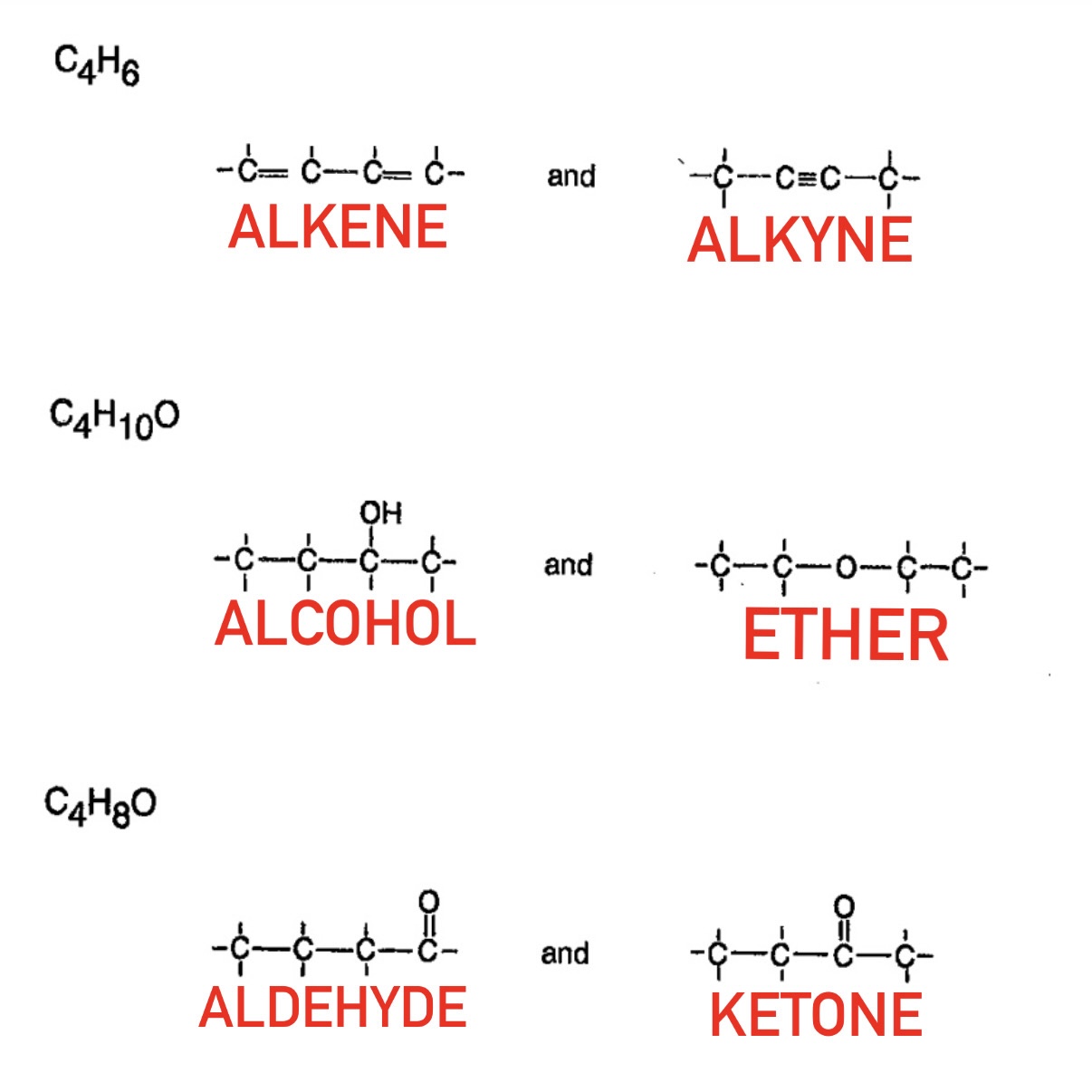
Functional Isomers
Geometrical Isomers
Optical Isomers
What are Optical Isomers?
Mirror-image molecules caused by a chiral carbon
What are Geometric Isomers?
Molecules that differ around a double bond (Cis/Trans)
What are Positional Isomers?
Same functional group in different positions
What are Functional Isomers?
Same molecular formula but different functional groups
What are Skeletal Isomers?
Same formula but different carbon chain arrangement
What is a Substitution Reaction?
A reaction where one atom or group is replaced by another atom or group
What Happens when a Primary Alcohol is Oxidized?
Primary alcohol → aldehyde
What Happens when a Secondary Alcohol is Oxidized?
Secondary alcohol → ketone
What Happens when a Tertiary Alcohol is Oxidized?
Usually no reaction
What is Ionization?
A reaction where a molecule forms ions
What is Esterification?
A reaction where an alcohol and carboxylic acid form an ester + water
What are the Groups that Help Dissolve in Water?
OH (Alcohol)
NH2 (Amine)
COOH (Carboxylic Acid)
What is the Hydrolysis Pattern for Ethers?
R–O–R + H2O → R–OH + HO–R
What is the Hydrolysis Pattern for Esters?
RCOOR + H2O → RCOOH + ROH
How are Substituents listed in an IUPAC Name?
In alphabetical order
Rank the Most Water-Soluble Groups
Carboxylic acid (-COOH)
Amine (-NH2)
Alcohol (-OH)