intro to thermochemistry
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
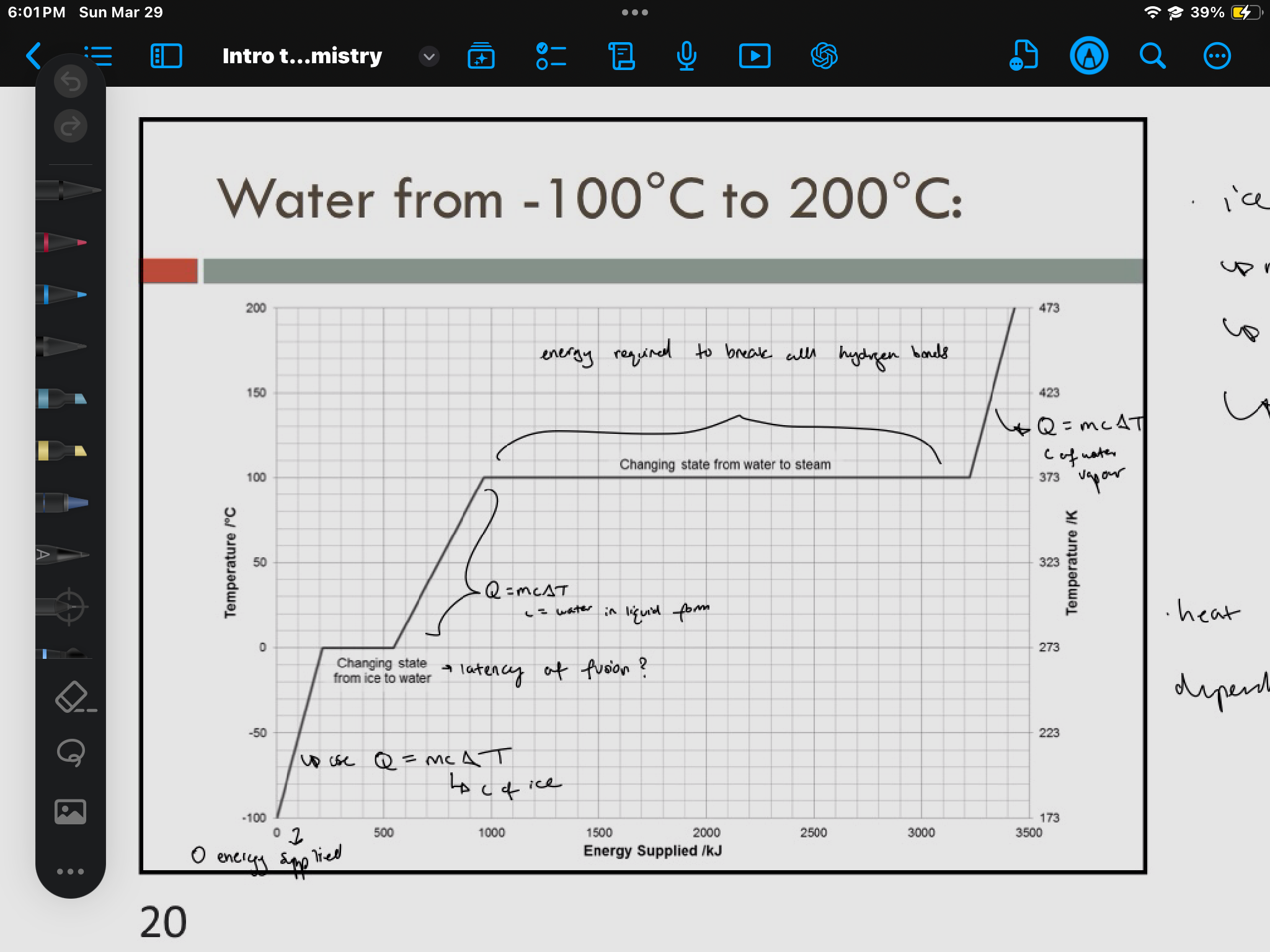
systems
where a physical or chemical change occurs
surroundings
the rest of the universe outside the system
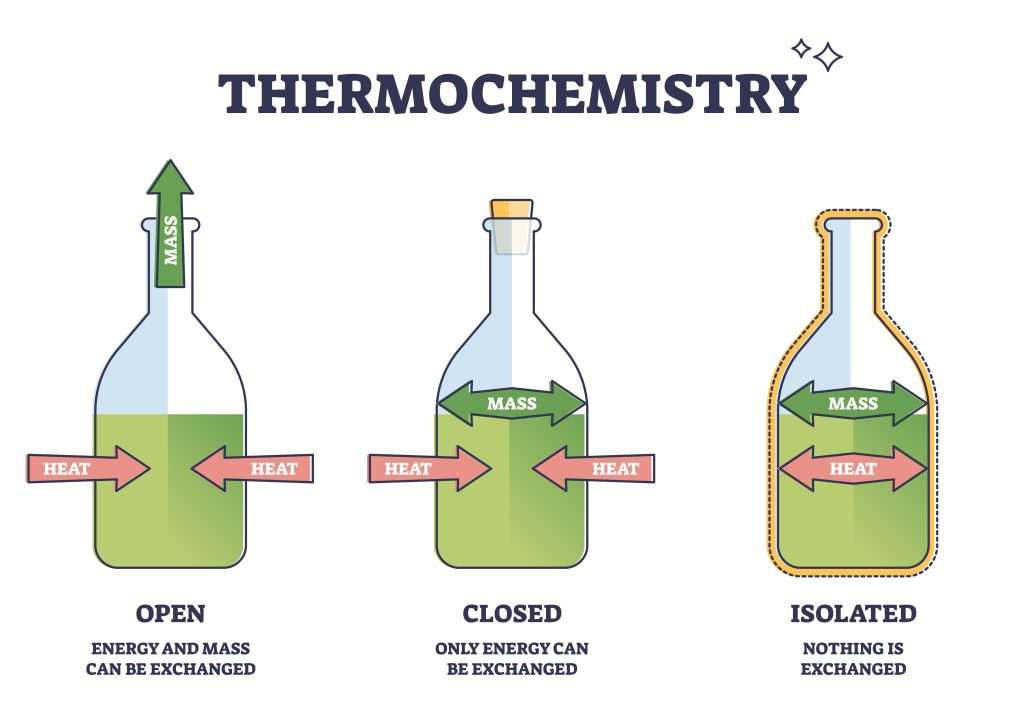
open system
both matter and energy can freely flow into the surroundings
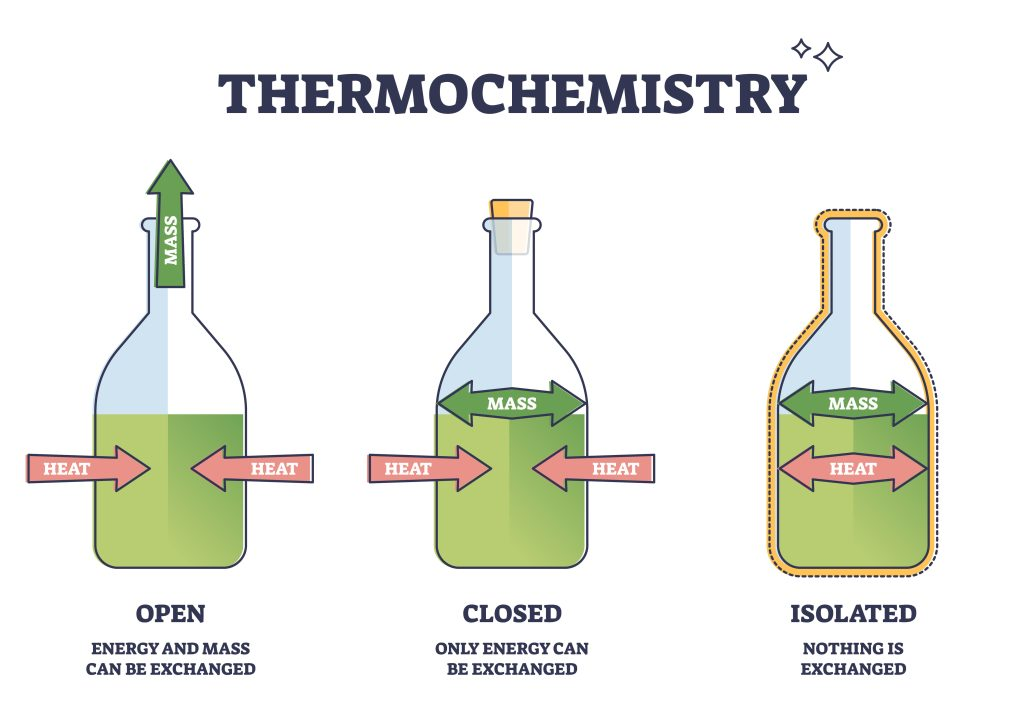
closed system
energy can flow to surroundings but matter cannot
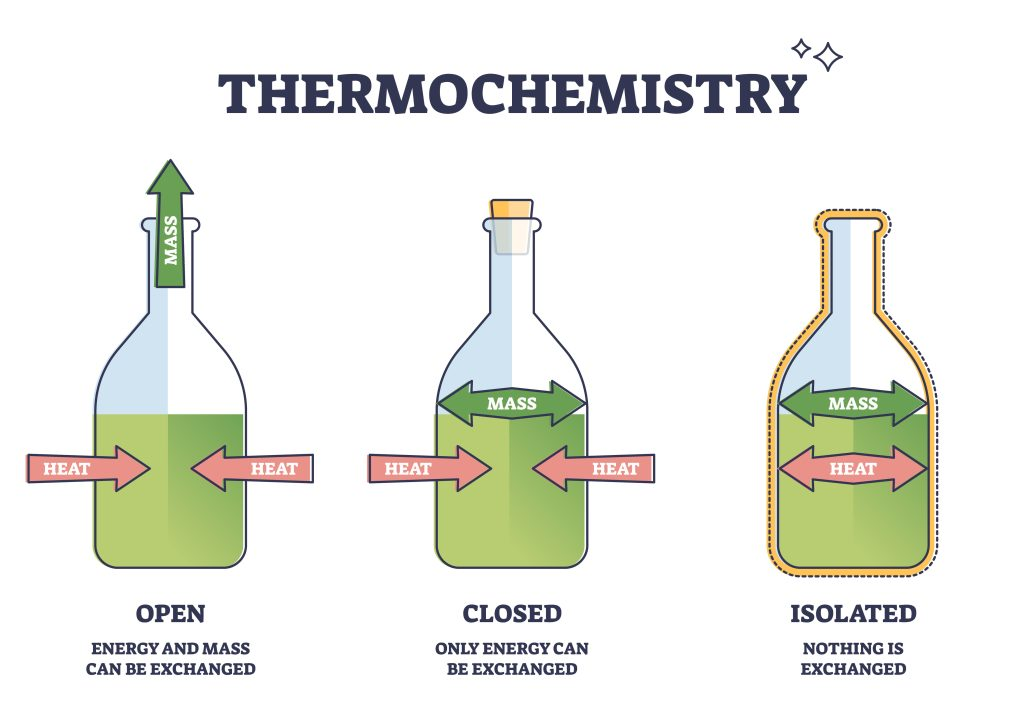
isolated system
neither energy nor matter can flow to the surroundings
cannot have perfect isolated system for a long period of time
energy transfer will occur overtime
delete this card
hallo
what is thermochemistry?
the study of the energy changes that accompany physical or chemical changes in matter
changes that occur in matter: physical changes. chemical changes, nuclear changes (changing the nucleus)
all accompanied by a change in energy that can be measured and quantified
thermal energy
energy available from a substance as a result of the motion of its molecules
stored in particles through translational, rotational, and vibrational motion
translational motion
move from place to place along a linear path
rotational movement
rotate about a bond axis through the center of mass; particles spin
vibrational movement
vibrate or oscillate back and forth
phase and movement type: gas
free translation
free rotation
free vibration
particles far apart and moving freely, mainly lots of fast translational motion
weak intermolecular forces
phase and movement type: liquid
restricted translation
restricted rotation
free vibration
particles close together but may move past each other
moderate amount of intermolecular forces
phase and movement type: solid
absent translation
very restricted rotation
free vibration
particles locked in place, can only vibrate
very strong intermolecular forces
heat
Q
amount of energy transferred between substances
measured in Joules (J)
Q = mcΔT
temperature
T
the average kinetic energy of the molecules in a sample
measured in C or K
relativity of temperature and heat
feeling heat is truly feeling rate at which heat is conducted away or towards you
cold: when surrounding particles hit you, you transfer some heat to them, you feel cold
hot: faster moving particles collide with yours, heat is transferred to you, you feel hot
energy flow
energy flows between substances because of their difference in temperature
depends on ability of substance to conduct heat towards or away from itself
NOT relative
why do metals feel cooler?
a network of delocalized electrons, always moving, transfer electricity and heat quickly
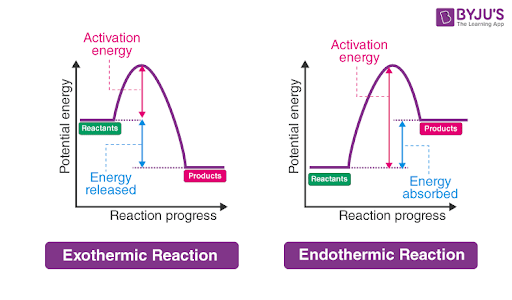
exothermic reactions
exo = exit
release of thermal energy
heat flows from system to surroundings
causes an increase in the temperature of the surroundings
negative Q value, Q < 0
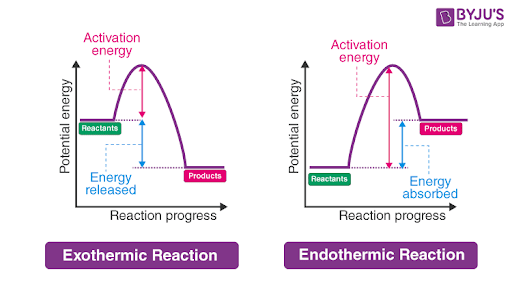
endothermic reaction
endo = enter
absorption of thermal energy
heat flows from surroundings into system
causing a decrease in temperature of surroundings
positive Q value, Q > 0
relation of heat for system and surroundings
Qsystem = -Qsurroundings
what is the internal energy of a system equal to?
Internal energy is the total energy inside a system, coming from all the particles in it
Internal energy = kinetic energy of particles + potential energy of all particles
kinetic energy
stored in
moving electrons in atoms
vibration, rotation and translation of atoms and molecules
chemical potential energy
stored in
nuclear potential energy of protons and neutrons
bond energy
intra and intermolecular forces
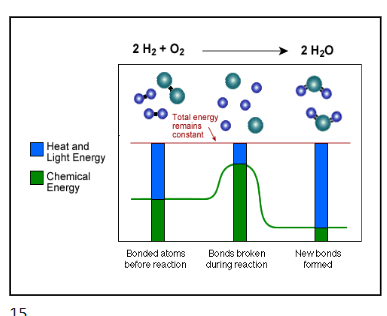
explain this diagram
total energy remains constant
reactions usually require activation energy
covalent bonds broken
atoms rearrange to have electrons in most stable state
form water molecules
specific heat capacity
c
the amount of energy required to raise the temperature of one gram of a substance one °C or one K
dependent on the three ways its molecules, atoms or ions can store thermal energy
more atoms, stringer bonds = higher heat capacity
heat formula
Q = mcΔT
the amount of heat transferred (Q) depends on the mass of the sample measured in grams (m), temperature change in C or K (ΔT), and specific heat capacity measured in J/gC or J/gK (c )
temperature readings explanation on a thermometer
temperature is a measure of the average translational energy of the molecules striking the thermometer
must have collision for transfer of energy
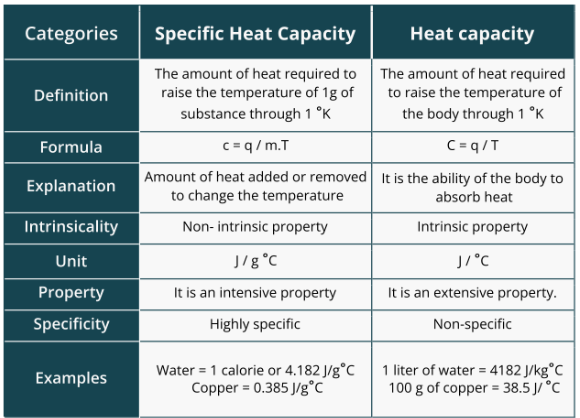
specific heat vs. heat capacity
specific heat capacity: amount of heat required to raise the temperature of 1g of substance through 1 kelvin
heat capacity: amount of the heat required to raise the temperature of the body through 1 K
heat capacity is additionally dependent on how much of the substance is present
energy for change of state: heat of fusion
heat of fusion, Lf: amount of energy required to change 1 gram of pure substance from solid to liquid at its melting point
NOT a change in temperature
Q = mLf
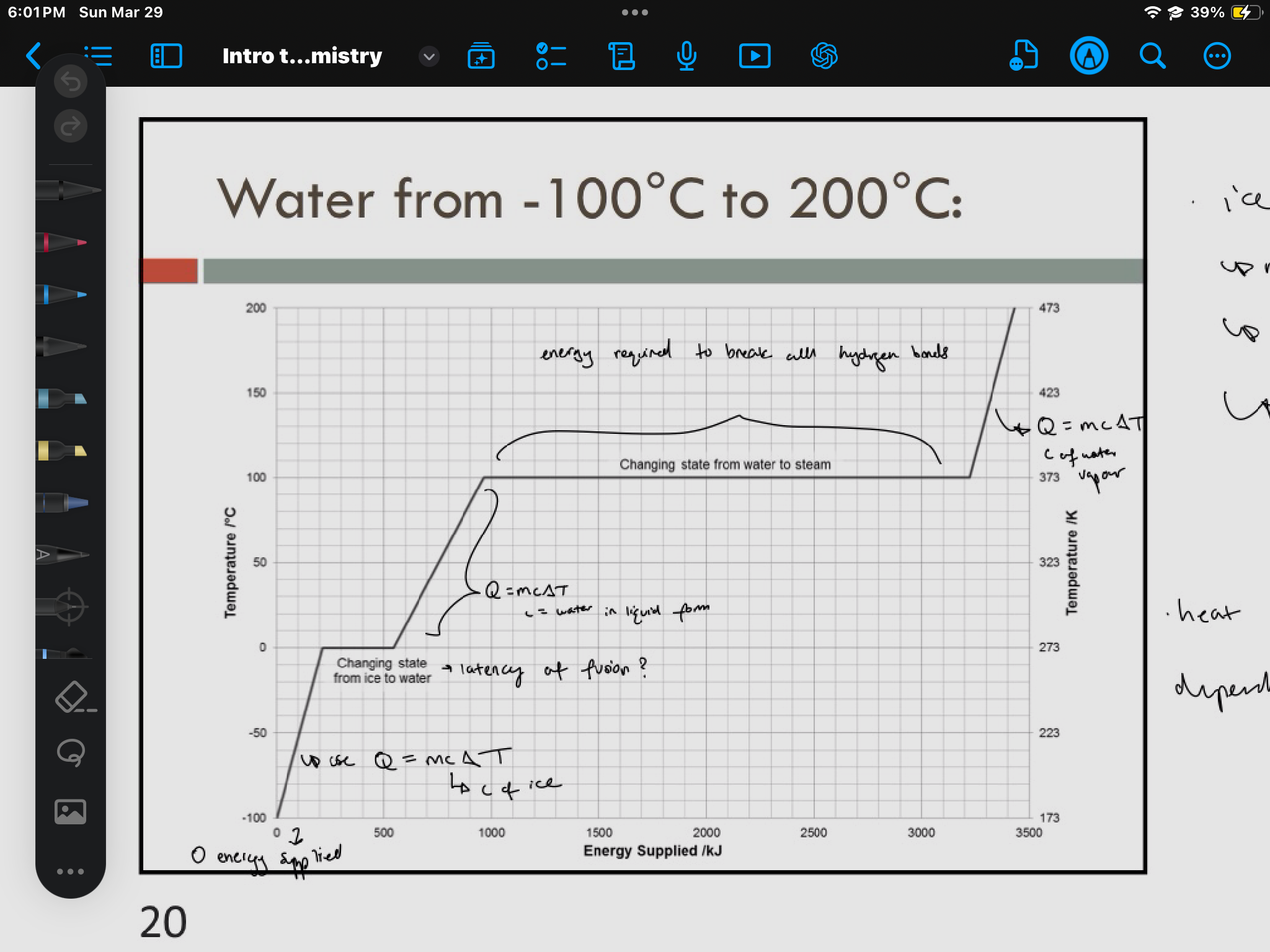
energy for change of state: heat of vapourization
the amount energy required to convert 1 gram of pure substance from liquid to gas at its boiling point
NOT a change in temperature
Q = mLv