Liquid dosage forms: suspensions and emulsions 3
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What is cracking?
Complete coalescence of the internal phase, resulting in separation of emulsion into 2 layers
Why does cracking occur?
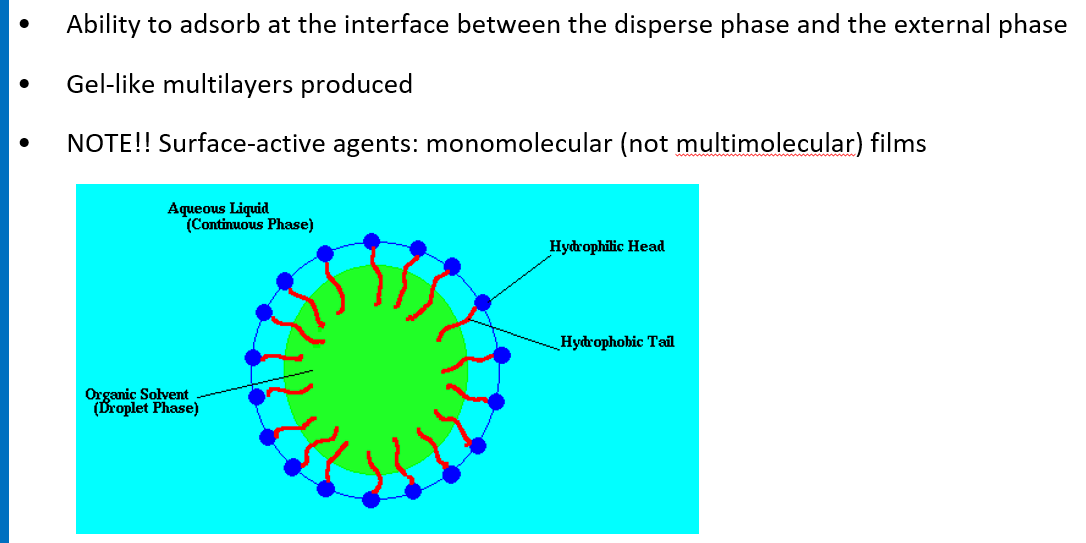
Due to the destruction of the mono/multilayer film at the interface between the droplet and external phase
If emulsion has cracked can it be reversed?
No once it has cracked it can’t be recovered
What are the 4 reasons as to why it cant be?
1) Incorrect selection of emulsifying agents
2) Presence of incompatible excipients
3) Temperature
4) Microbial spoilage
What occurs in a flocculated state?
Secondary interactions (van der waals forces) maintain droplets at a defined distance of seperation (within the secondary minimum)
Putting this into application of shaking what occurs?
Shaking will redisperse these droplets to form a homogenous formulation

What is creaming a result of?
Density difference between the oil and water phases; involves either sedimentation or elevation of droplets of internal phase, producing a layer of concentrated emulsion either at top or bottom of container
What is a problem with creaming?
Aesthetic problem (resulting emulsion is unsightly); upon shaking emulsion is rendered homogenous
Rate of creaming links to what equation?
Stoke’s equation
How can it be prevented?
If density difference between the 2 phases is 0 (in practice, cannot be easily achieved)
What are 2 methods with the rate of creaming can be reduced by?
Reduce the average particle size of the disperse phase
Increase the viscosity of the emulsion
Phase inversion

Emulsion instability: Hydrophilic polymers
Emulsion instability: Adsorbed particles



Emulsifying agents