chem midterm 3 flashcards
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
alpha decay
alpha particle released is 4 2 He
reduces element mass by 4
reduces element protons/atomic # by 2
beta - decay
neutron → proton
eject beta particle
add 1 to atomic number
Beta + decay
proton → neutron
mass stays same
atomic # decreases by 1
releases positron and neutrino
gamma decay
ejects photon (excess E)
mass and atomic number doesn’t change, and element goes from excited to stable.
fission
nuclei split into two or more nuclei. nuclear bomb, power plants
fusion
two small particle combine. clean nuclear power
magnitude of bonding power
nuclear energies = millions-billions
chemical bond energies = 100s-1000s
intermolecular forces = 10s
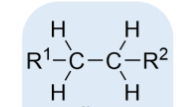
alkane
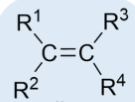
alkene
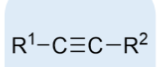
alkyne
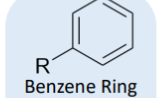
benzene ring
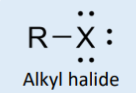
alkyl halide (F, Cl, Br, I)
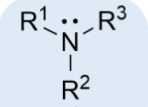
amine
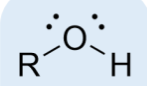
alcohol
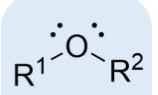
ether
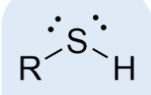
thiol
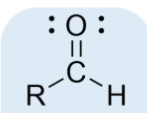
aldehyde
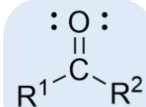
ketone
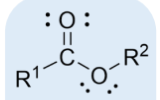
ester
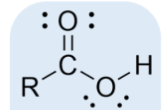
carboxylic acid
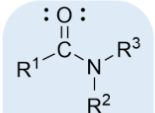
amide
ozone layer decomposition
homogenous catalyst Cl radical
formed when UV breaks down CFCs, which are used in refridgeration and aerosal
destroys ozone layer
2O3→3O2
catalyst converter
automobile, improve air quality
heterogenous Pt/Pd metal catalyst
reduces N2
oxidizes rxns
NO + CO + CH → N2 + CO2 + H2O
haber process
N2 → ammonia and fertilizer
exothermic heterogenous slightly oxidized iron powder
N2+3H2 → 2NH