DAT OChem Reactions
1/147
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
148 Terms
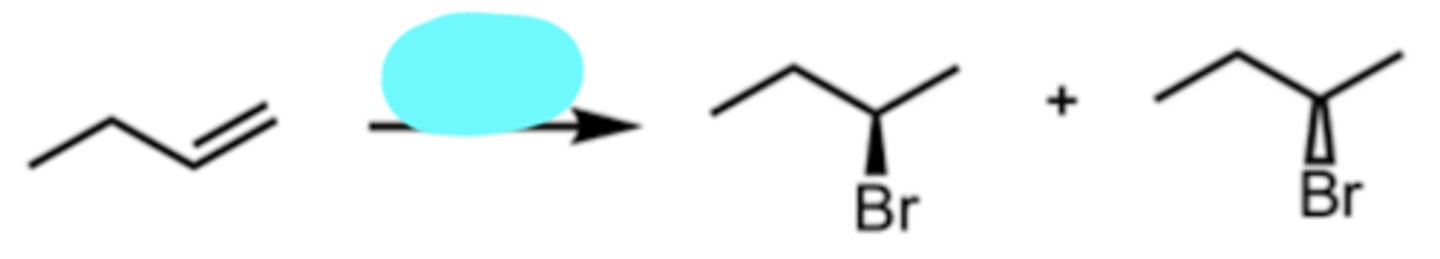
HBr
Hydrohalogenation
markovnikov addition (Br attaches to the less substituted side of alkene)
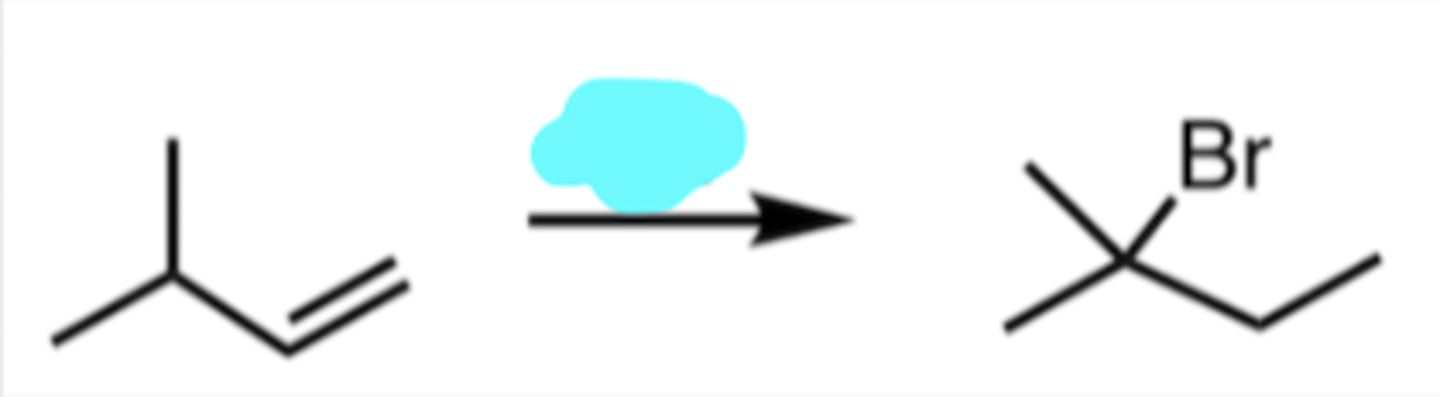
HBr
Hydrohalogenation (with Rearrangement)
2° Carbocation near 3° Carbon → Hydride Shift
HBr indicates Markovnikov addition (the intermediate that’s going to form is going to be the most stable, most substituted intermediate possible)
initial intermediate would be a 2° carbocation right next to a 3° carbon
intermediate undergoes a hydride shift so the carbocation can then be more highly substituted as a 3° carbocation
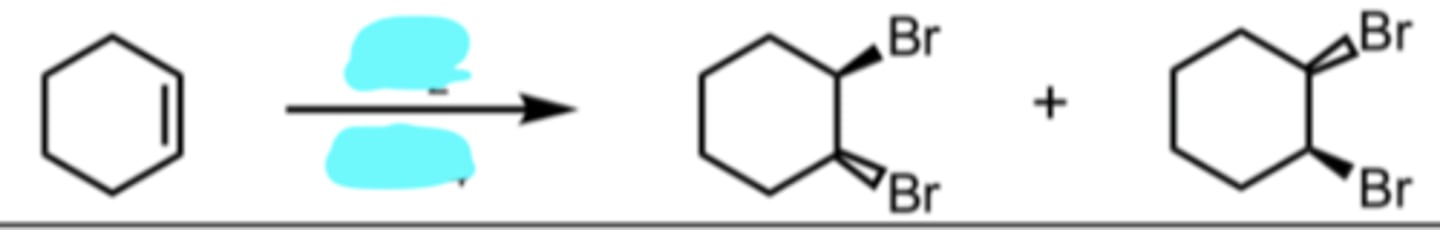
Br2
CCl4
Halogenation
anti addition
products are enantiomers
ignore CCl4 it’s just a solvent
HBr
ROOR
Hydrobromination with Peroxide
anti-mark addition (Br goes on less substituted side of alkene)
ROOR (peroxide) causes it to be anti-mark
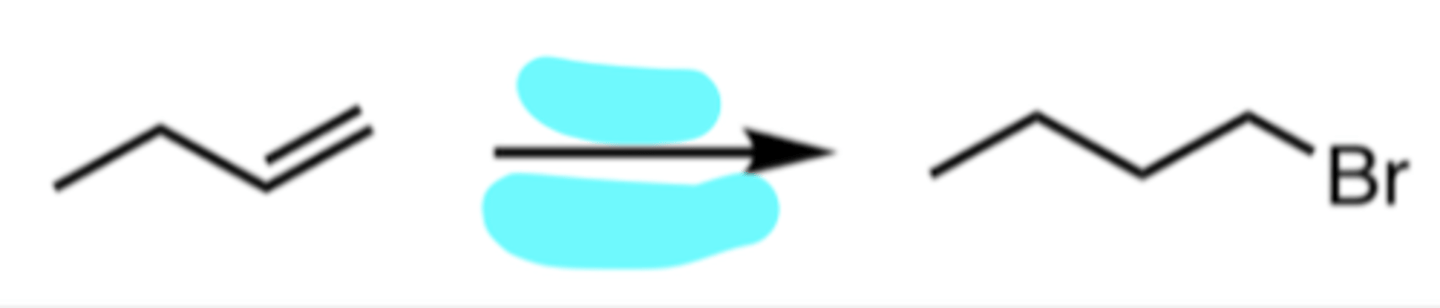
H3O+ *
*any acid catalyst (H2SO4, H3PO4, etc.) and water
Hydration
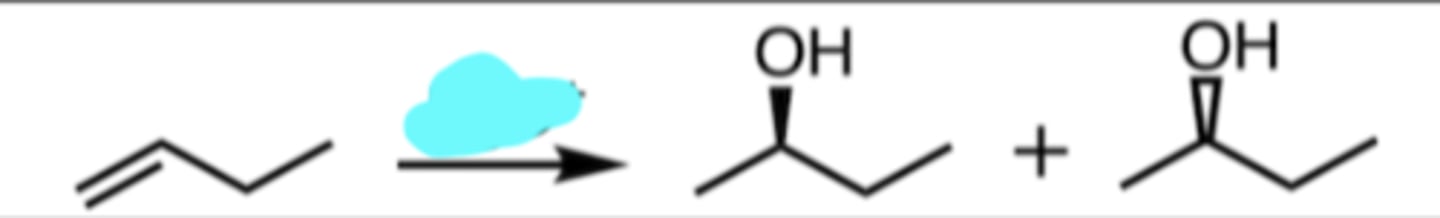
H3O+
Hydration (with Rearrangement)
markovnikov addition (add to more substituted)
intermediate creates 2° carbocation right next to 3° carbon → hydride shift for carbocation to be in 3° position (more stable)
3° carbocation formed and 3° alcohol is product
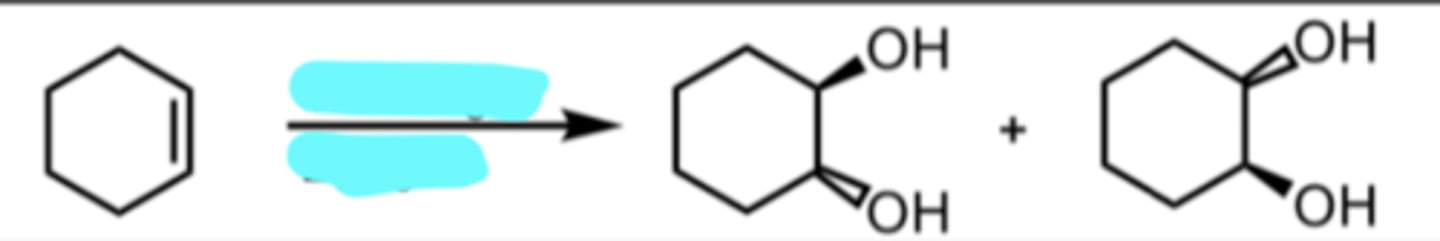
Br2
H2O
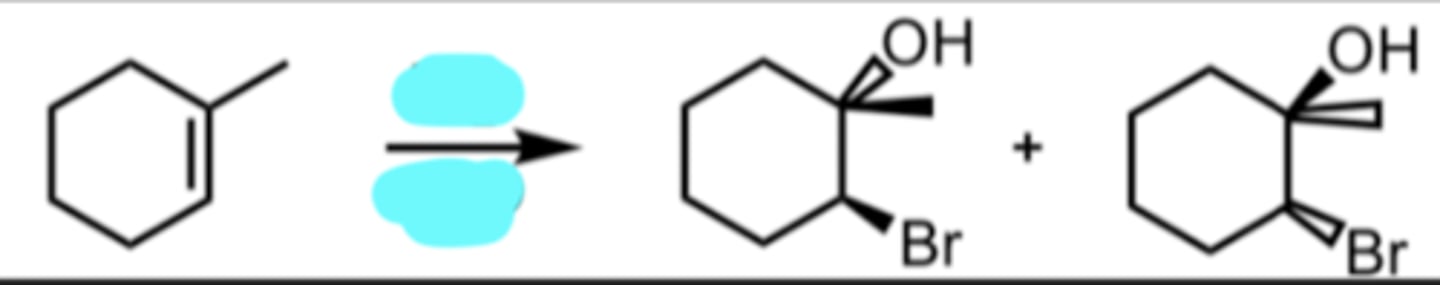
Bromination in H2O
OH and Br added anti to each other
when we have a protonated epoxide looking intermediate, H2O will always attack the more substituted side and result in anti addition
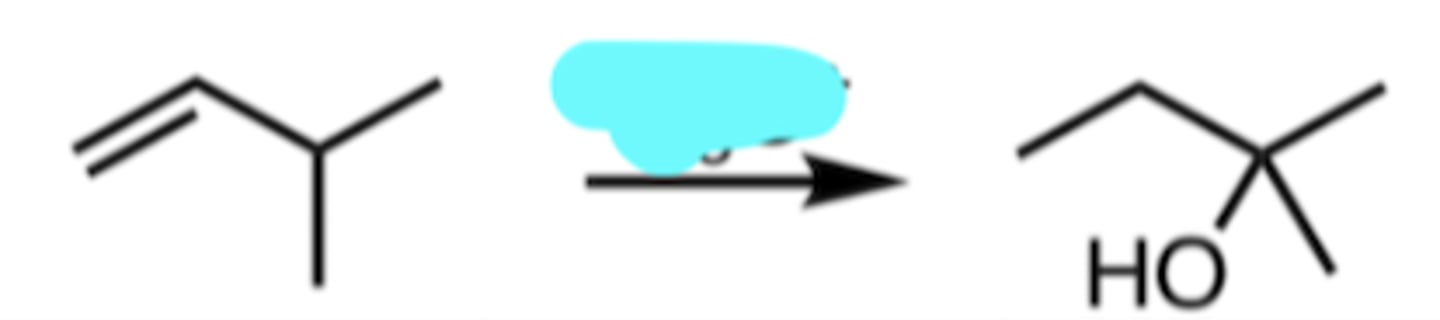
Hg(OAc)2, H2O
NaBH4
Oxymercuration-Demurcuration
produces the more highly substituted alcohol
NO rearrangements possible (no carbocations)

BH3, THF
H2O2, OH-, H2O
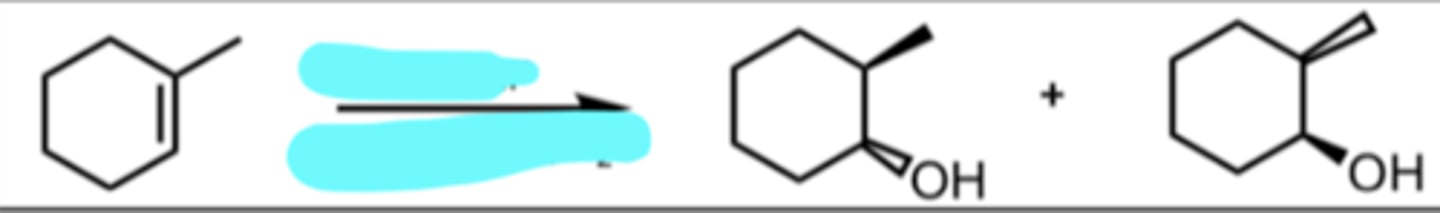
Hydroboration-Oxidation
anti-markov product (add to less substituted side of alkene)
H is added to more substituted side of alkene
OH is added to less substituted side of alkene
since both groups are added at the same time, it is SYN addition
OH and H are both placed on a wedge and the methyl is pushed back on a dash (or vice versa)
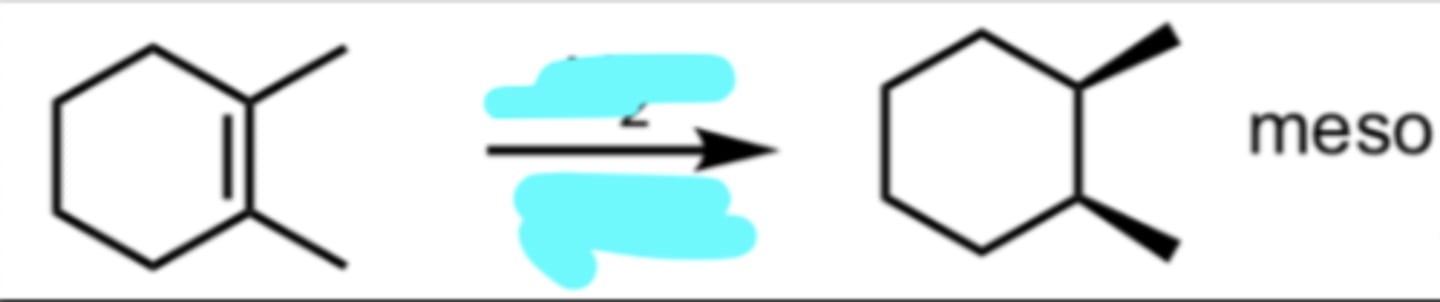
OsO4
H2O2
Syn-Dihydroxilation of Alkenes
OsO4 adds 2 hydroxy groups onto alkene in SYN addition
meso compound = no enantiomer it would just be the same compound
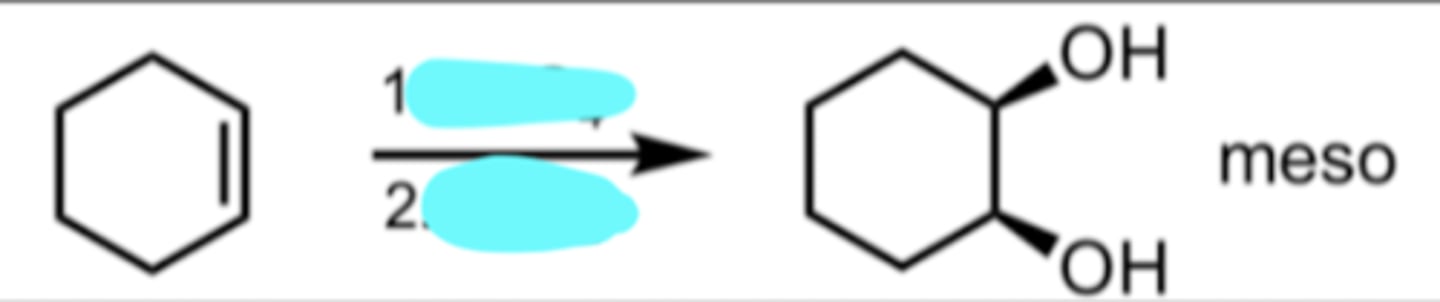
KMnO4 (cold, dilute)
OH-
Syn-Dihydroxilation of Alkenes
KMnO4 is cold and dilute so it won’t be doing ozonolysis (cleaving/splitting through alkene bond) unlike if it were hot and acidic
KMnO4 adds 2 hydroxy groups onto alkene in SYN addition
meso compound = no enantiomer it would just be the same compound

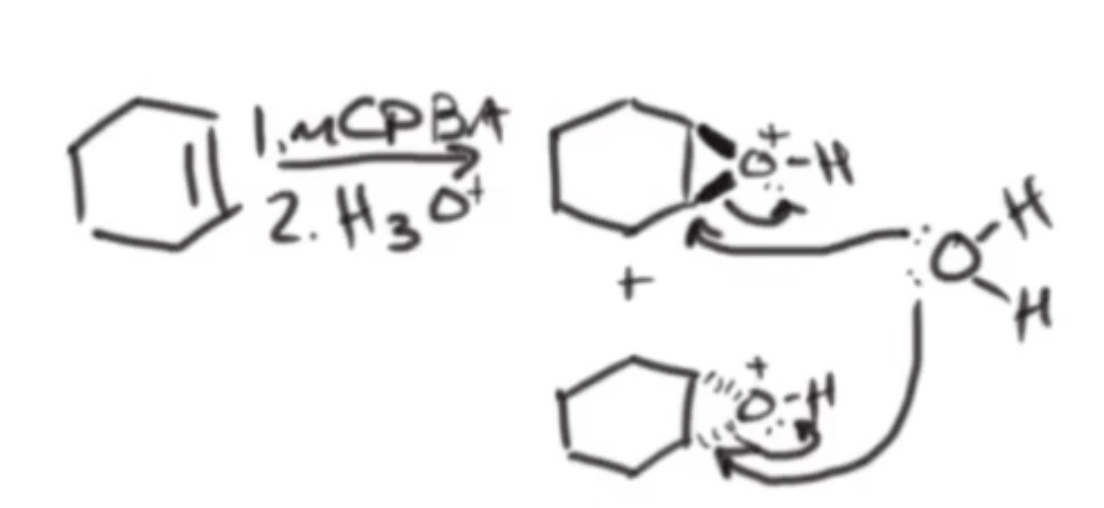
mCPBA
H3O+
Anti-Dihydroxylation
hydroxyl groups added anti to each other
step 1: mCPBA → forms epoxide intermediates
step 2: aqueous acid H3O+ (or aqueous base OH-)
protonates the epoxide O
rule: whenever epoxide has + charge, you are going to attack the more substituted side (none here)
second OH is added anti to first
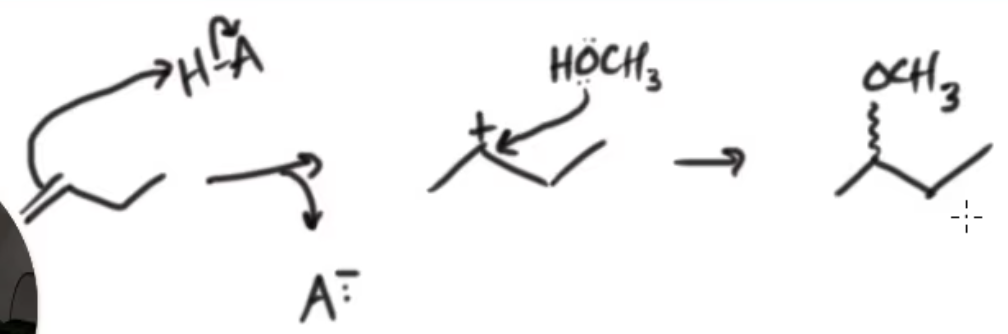
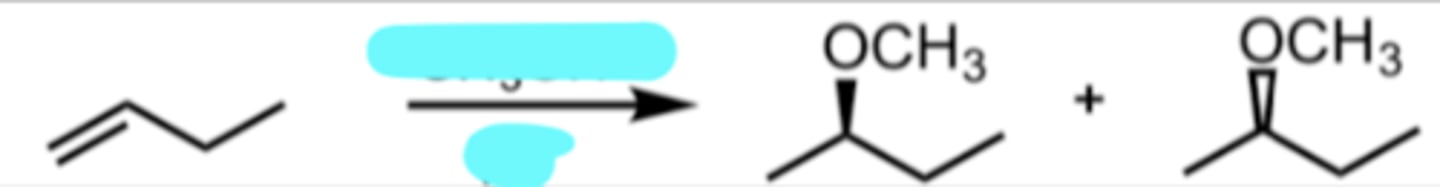
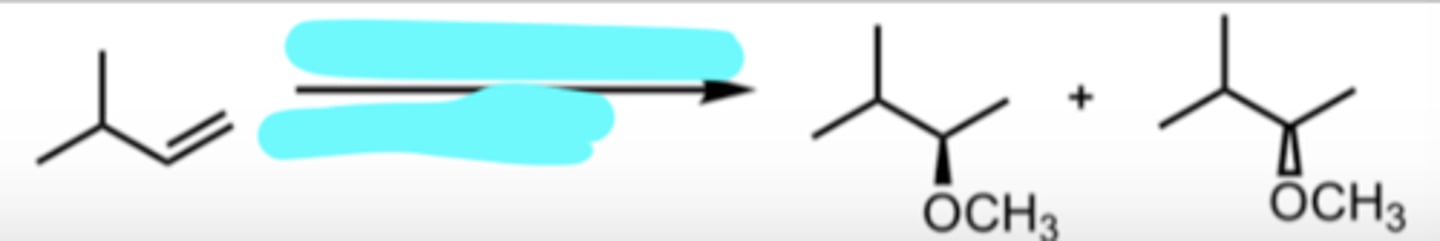
CH3OH
H+
Addition of an Alcohol to Produce an Ether
markovnikov addition
ether is produced
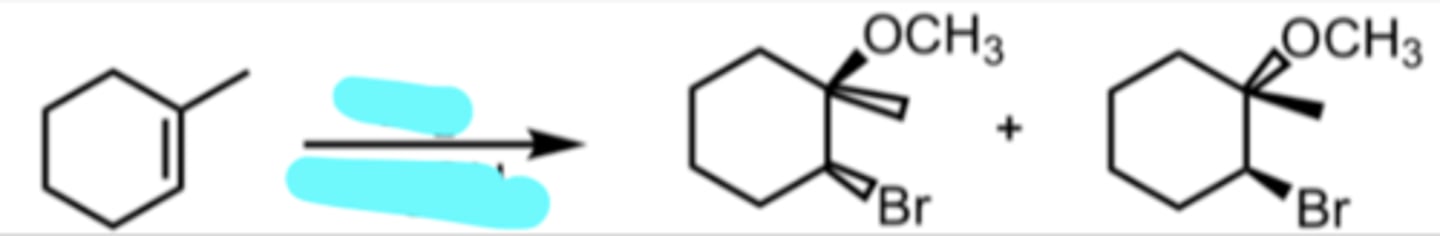
Br2
CH3OH
Bromination in Alcohol
“epoxide” with Br formed with + charge → means that next thing added goes to more substituted side
anti addition (Br and OCH3 are opposite)
ether formed
Hg(OAc)2, CH3OH
NaBH4
Alkoxymercuration-Demercuration
NO rearrangements possible (no carbocation formed)
be careful: reagents look very similar to oxymercuration-demercuration conditions. if it were oxymercuration-demercuration, it would use water instead (resulting in OH added)
this however has methanol (CH3OH) which ends up being added as an alkoxy group (R-O-R), forming an ester
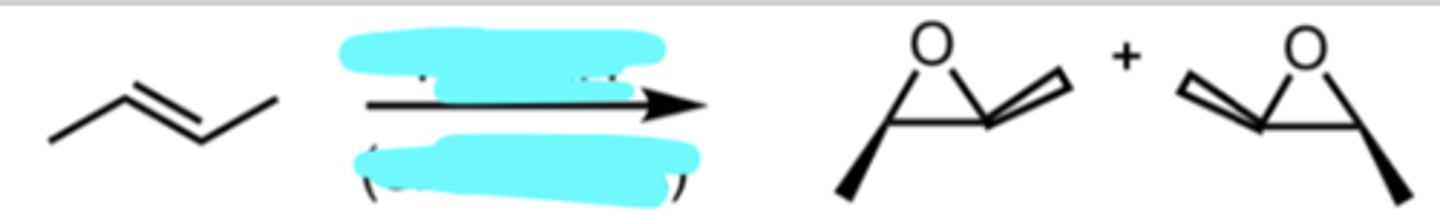
mCPBA (or RCO3H)
Epoxidation
mCPBA is a peroxy acid that converts alkene → epoxide
need to form both enantiomers
mirror image but NOT superimposable (if they were: identical and can be placed directly on top of one another so that all parts align perfectly)
H2, Pd-C (or Pt)
Catalytic Hydrogenation (alkene → alkane)
Pd-C is a palladium catalyst that helps the H’s reduce the alkenes
Need either Pd-C as catalyst or Pt (platinum) catalyst

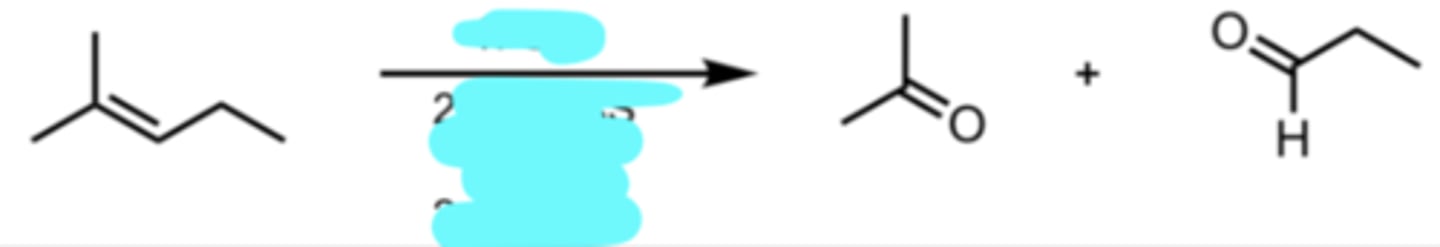
O3
(CH3)2S (aka DMS) or Zn/H2O
Ozonolysis (Reducing Conditions)
cleave double bond and form 2 carbonyl products
the carbonyl on the right side keeps its H, making it an aldehyde
O3 + DMS → ketone + aldehyde

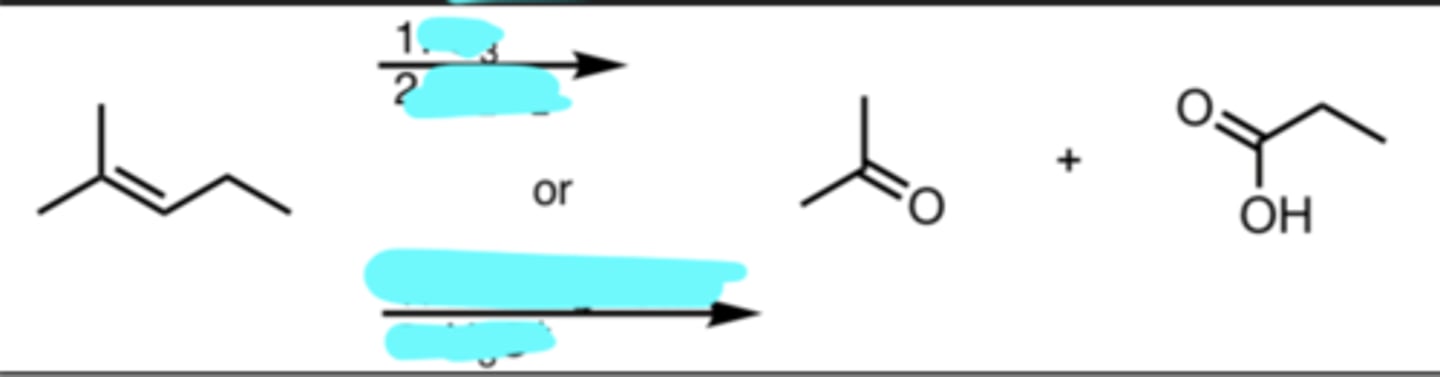
O3
H2O2
or
KMnO4/heat (acidic or basic doesn’t matter)
H3O+
Ozonolysis (Oxidizing Conditions)/Oxidative Cleavage
HOT KMnO4 → double bond is cleaved and forms two carbonyls
carbonyl on the right is originally bonded to an H. it is then replaced by an OH, so either H2O2 or H3O+
cold, dilute KMnO4 → would NOT cleave double bond. would instead just add an OH on either side of where the alkene was originally

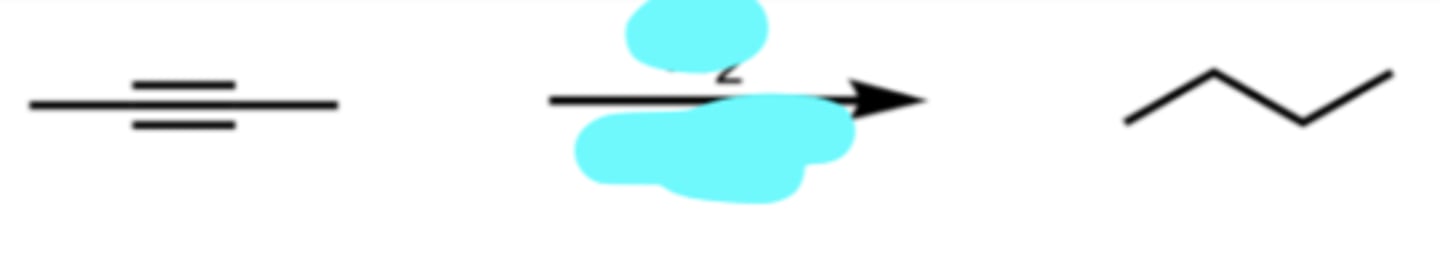
H2, Pd-C
Catalytic Hydrogenation (Catalytic Reduction)
alkyne → alkane
H2 alone will not react, need Pd-C catalyst
H2 + Pd-C = Lebron, can’t stop it, takes it all the way to alkane
Pd-C: P Diddy doesn’t stop he goes all the way
H2, Lindlar's catalyst
Reduction to Cis-Alkene (alkyne → cis alkene)
Lindlar’s Catalyst:
H2/Lindlar’s: alkyne → cis alkene “linda says sisss but only gets halfway into story”
poisoned (ineffective) metal catalyst. ineffective and slow so only reduces down to alkene
we know product is cis because metal tends to grab the pi bond from one face

Na or Li, NH3 (liquid)
Reduction (alkyne → trans alkene)
Na or Li/NH3: alkyne → trans alkene (Naaa u Lied ur trans - NoHate(NH3)
Na / Li are neutral, meaning they are radicals → electrons in intermediate want to be as far apart as possible → H’s go as far apart as possible → TRANS configuration

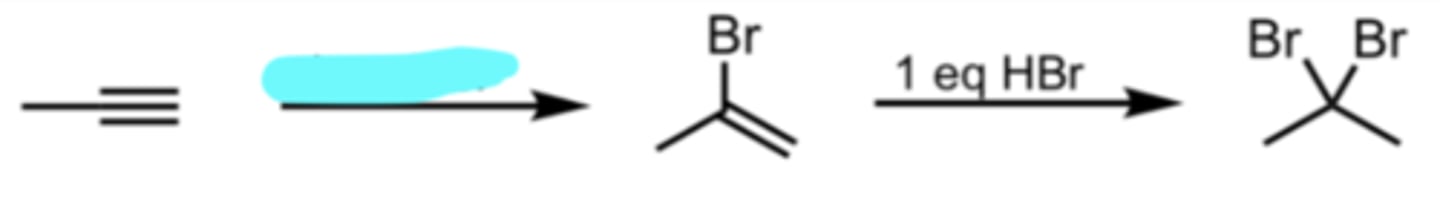
1 eq HBr
Hydrohalogenation with HBr (Terminal Alkyne)
1 eq HBr → 2nd eq HBr
Hydrohalogenation with HBr (Internal Alkyne)

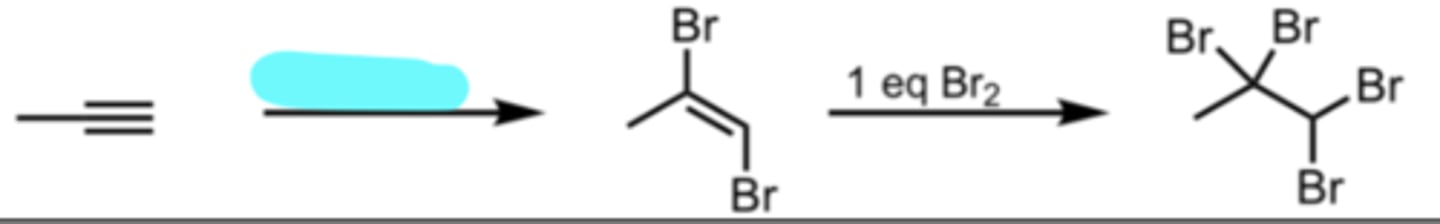
1 eq Br2
Halogenation with Br2
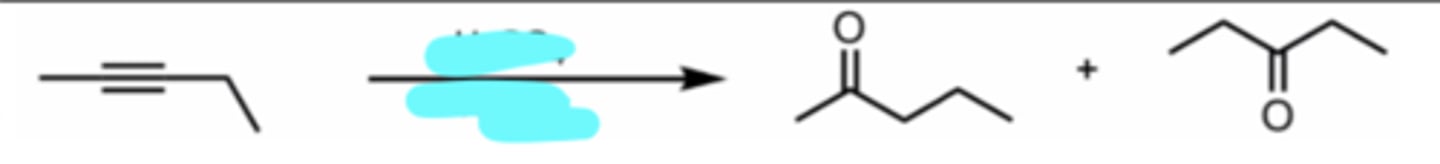
HgSO4, H2SO4
Hydration of an Internal Alkyne
HgSO4, H2SO4
Hydration of a Terminal Alkyne (Markovnikov)
Oxygen is added Markovnikov
HgSO4 is the catalyst because the reaction is so slow

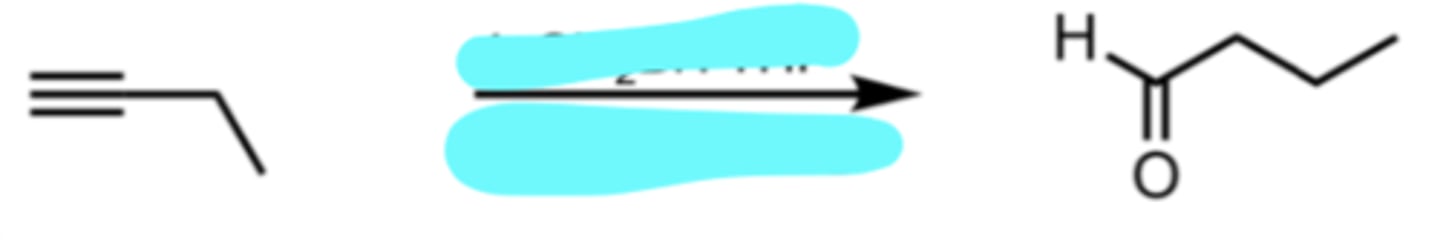
Sia2BH, THF
H2O2, OH-, H2O
Hydration of a Terminal Alkyne (Anti-Markovnikov)
NaNH2
SN2 Addition of an Acetylide Ion to an Alkyl Halide
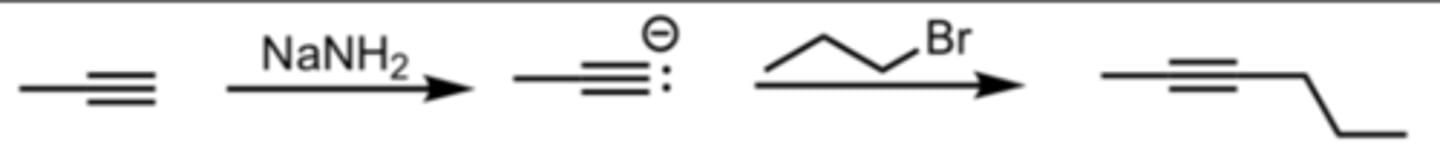
NaNH2
SN2 Addition of an Acetylide Ion to a Ketone
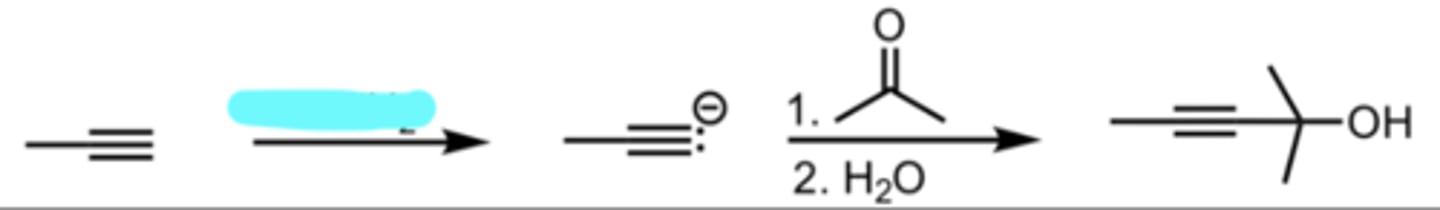
NaNH2
SN2 Addition of an Acetylide Ion to an Epoxide
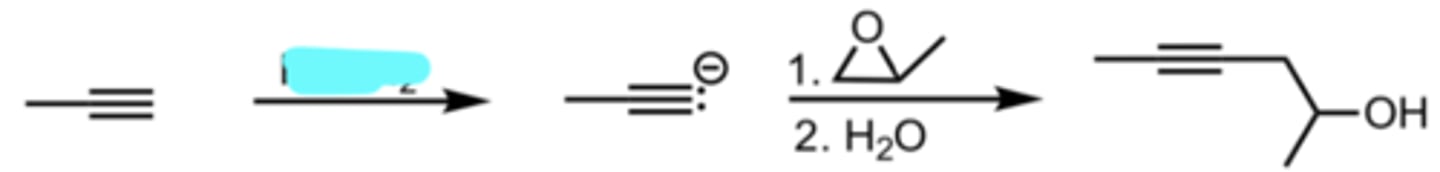
O3
H2O2
or
KMnO4/heat
H3O+
Ozonolysis/Oxidative Cleavage on an Internal Alkyne
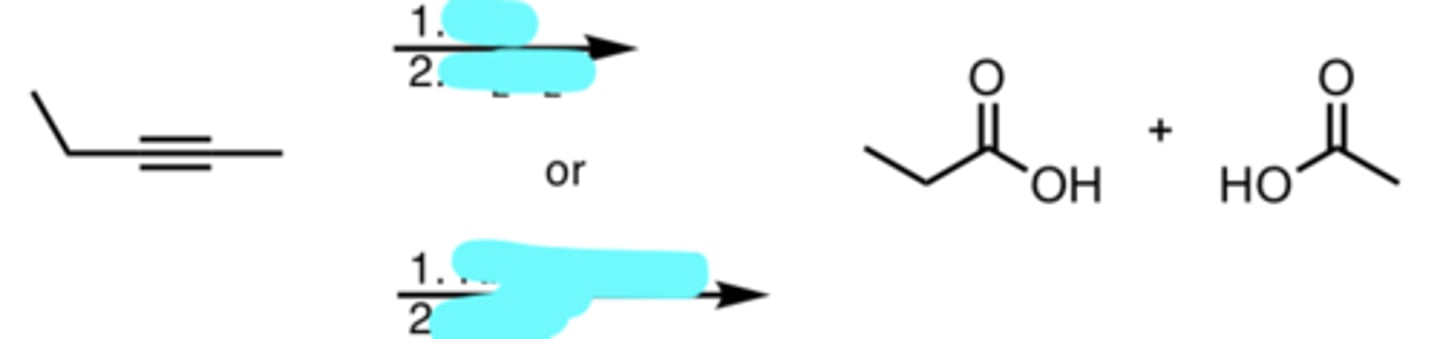
O3
H2O2
or
KMnO4/heat
H3O+
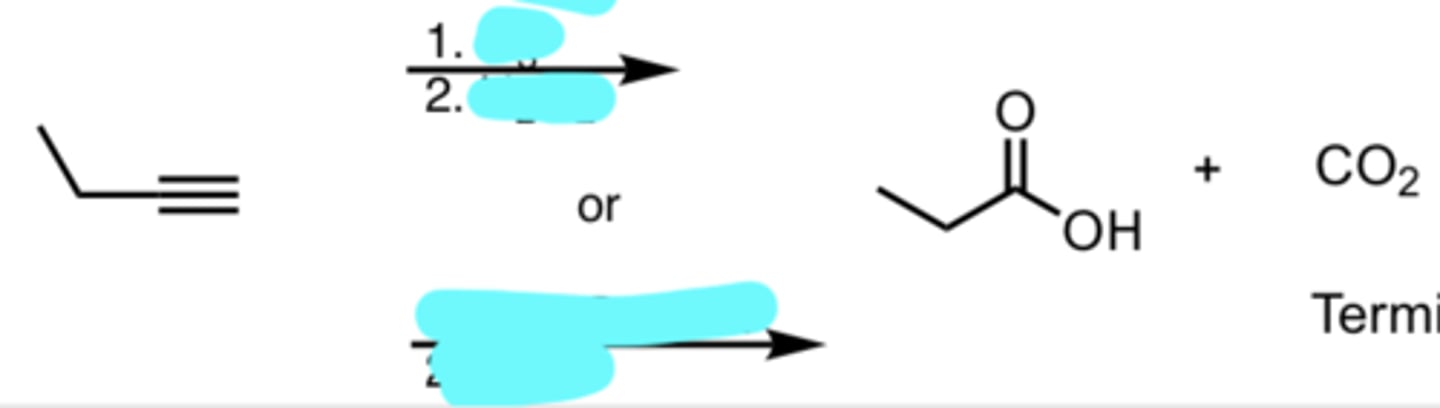
Ozonolysis/Oxidative Cleavage on a Terminal Alkyne
Terminal carbon becomes CO2
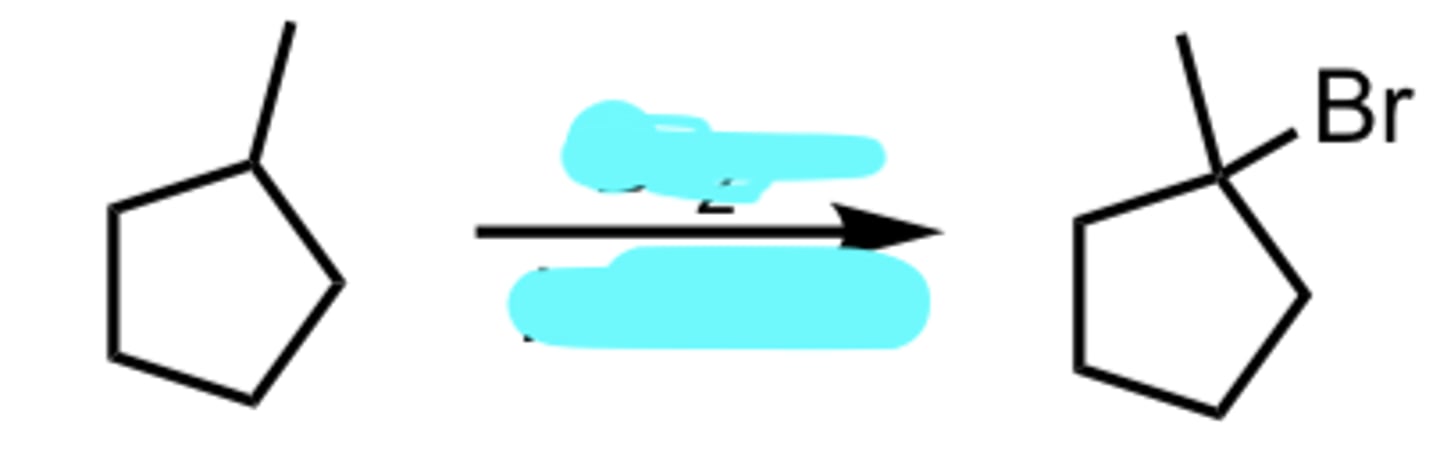
Br2, hv (light) or Δ (heat)
Free Radical Halogenation using Bromine (more selective)
Markovnikov
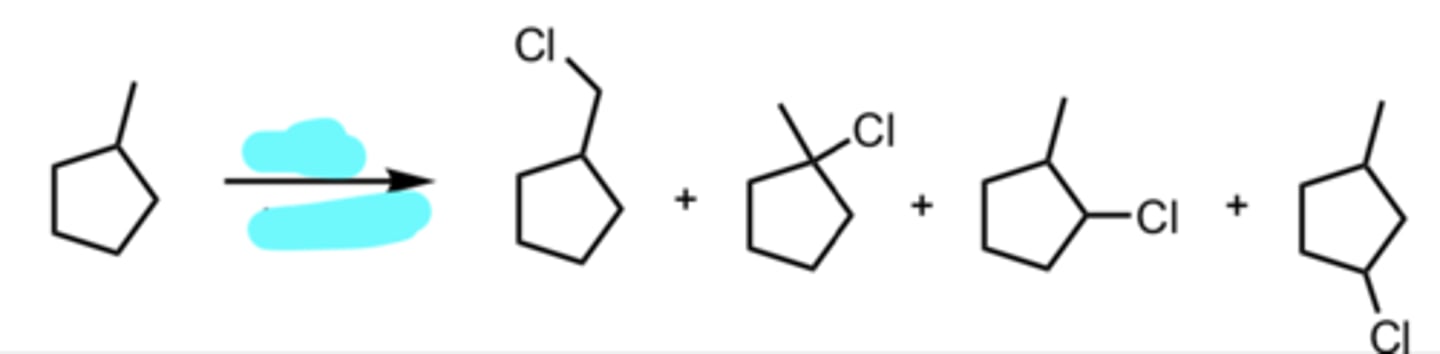
Cl2, hv (light) or Δ (heat)
Free Radical Halogenation using Chlorine (less selective)
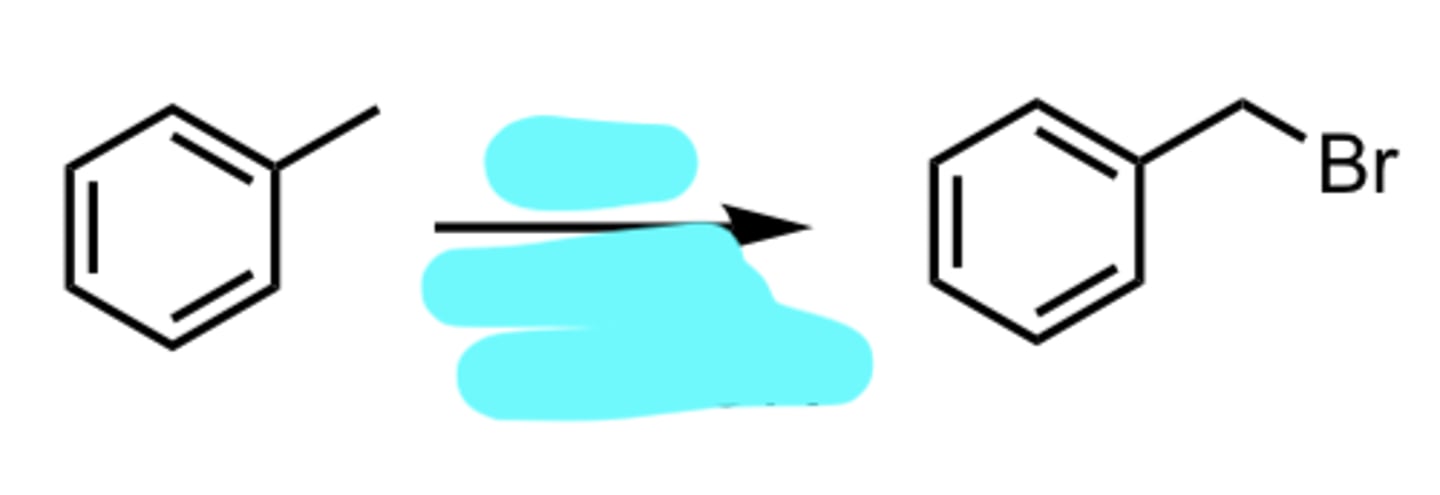
NBS, hv or Δ (heat) or ROOR
Benzylic Bromination
NBS, hv or Δ (heat) or ROOR
Allylic Bromination
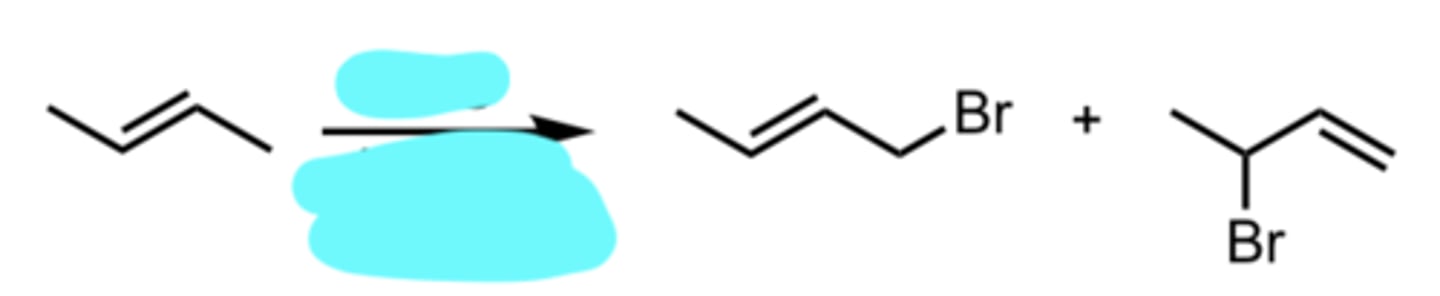
hv or Δ (heat)
Diene Addition to a Dienophile (Alkene)

hv or Δ (heat)
Diene Addition to a Dienophile (Alkyne)

hv or Δ (heat)
Diene Addition to a cis Dienophile
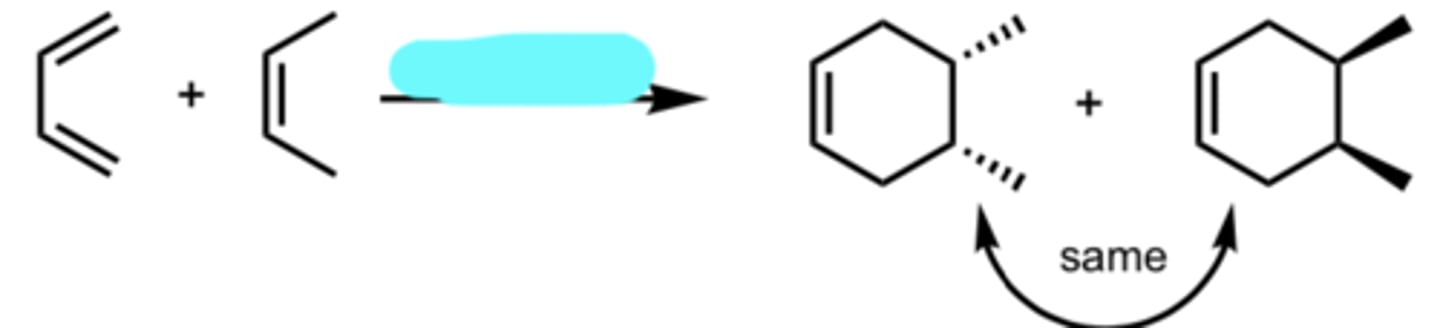
hv or Δ (heat)
Diene Addition to a trans Dienophile
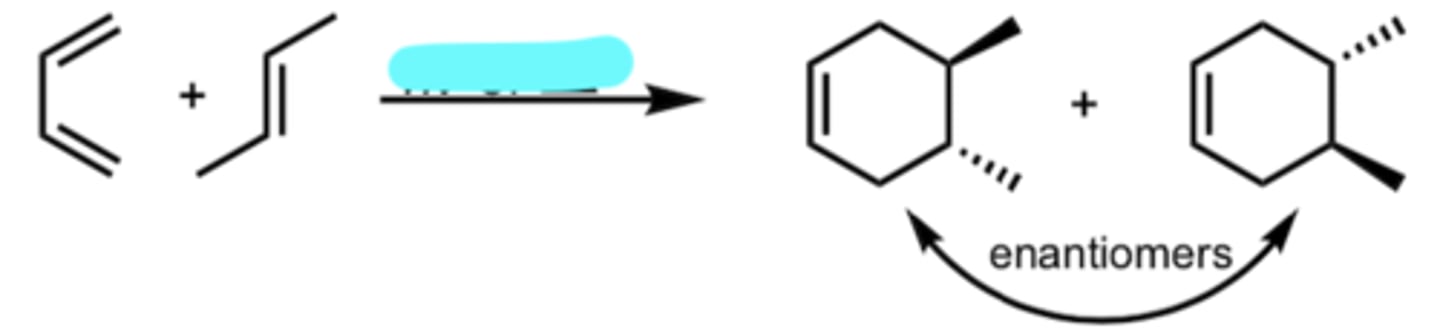
hv or Δ (heat)
Diene Addition to a substituted Dienophile
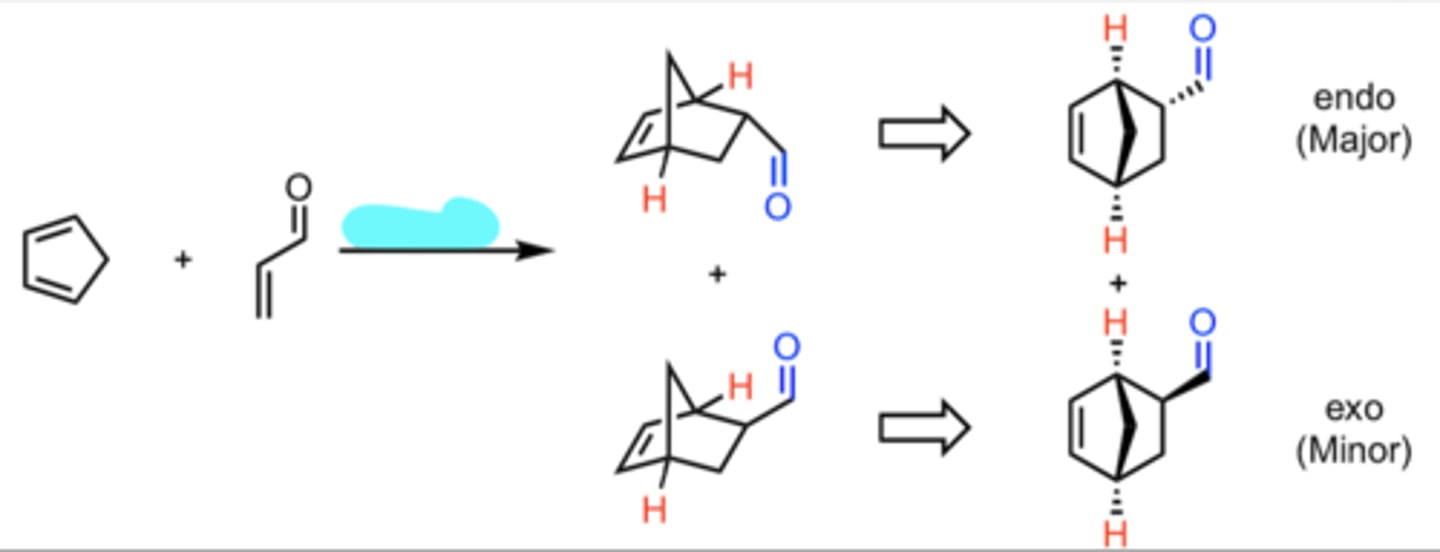
CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to an Aldehyde
produces 2° alcohol
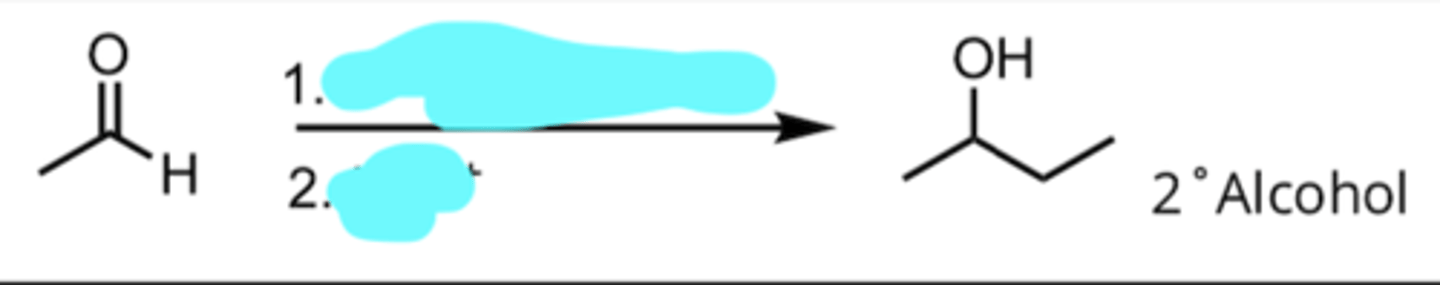
CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to a Ketone
produces 3° alcohol
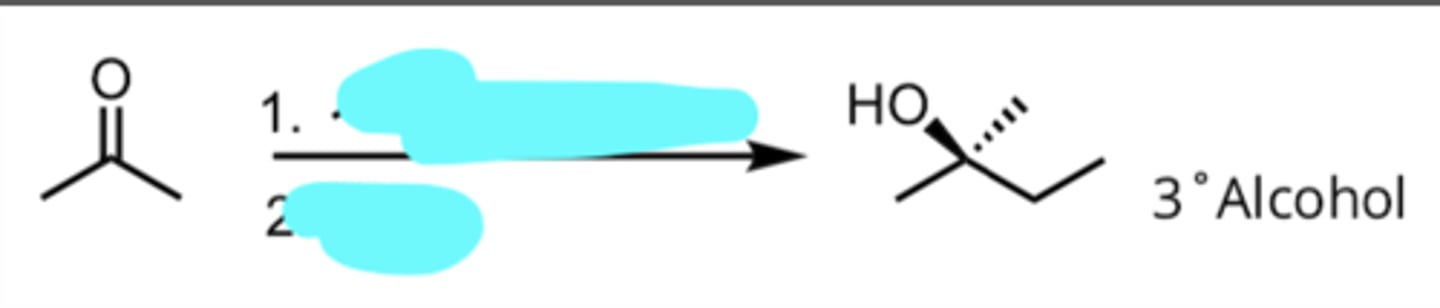
2 eq. CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to an Ester
produces 3° alcohol
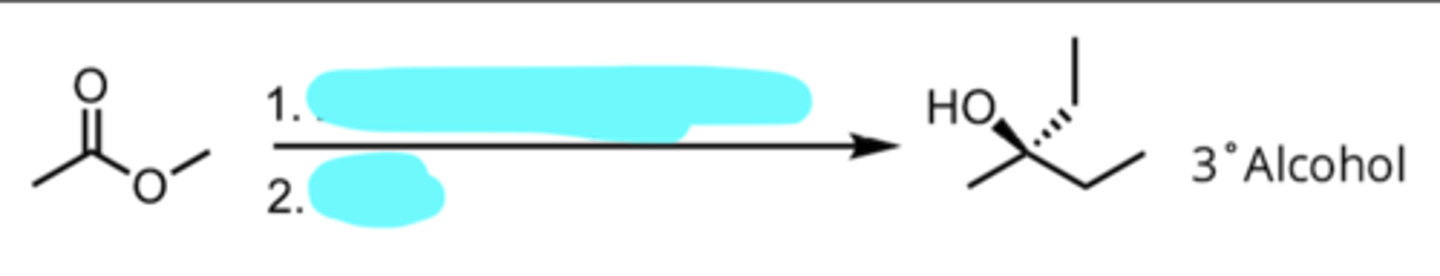
2 eq. CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to an Acyl Chloride
produces 3° alcohol
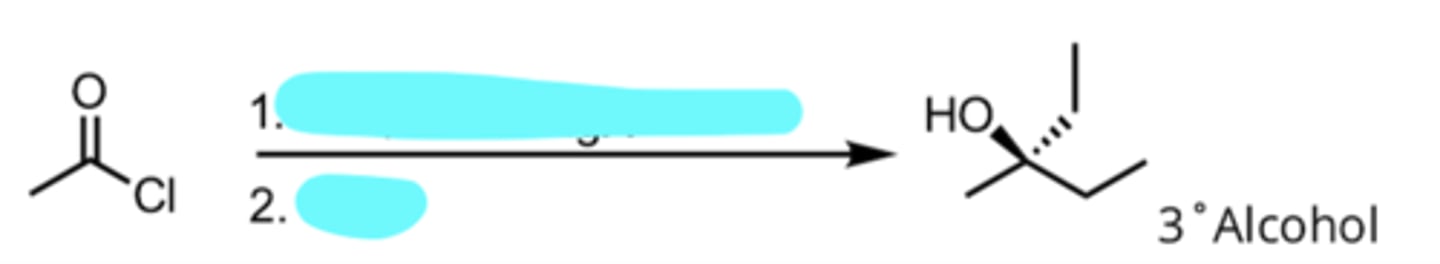
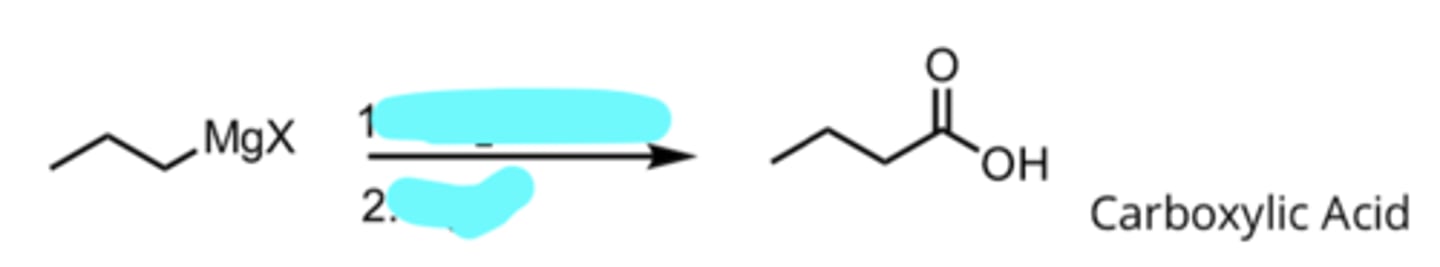
CO2, Ether
H3O+
Addition of a Grignard Reagent to CO2
produces carboxylic acid
CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to an Epoxide
adds to the less substituted side

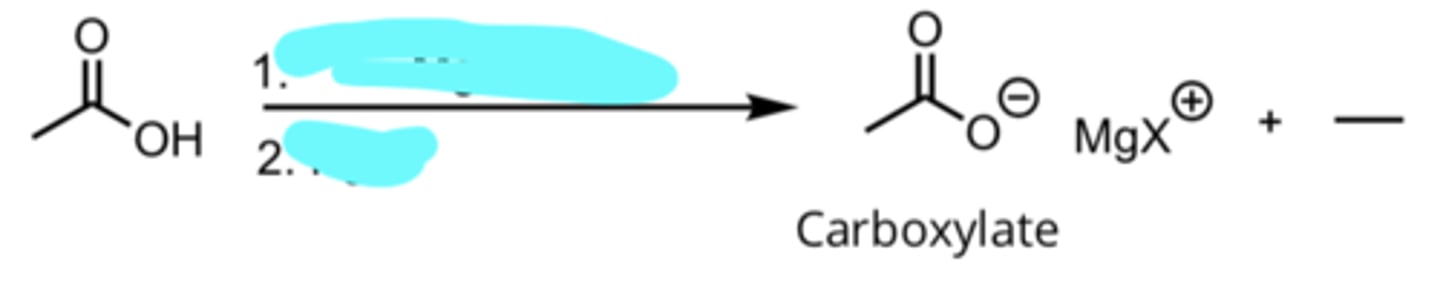
CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to a Carboxylic Acid
produces carboxylate
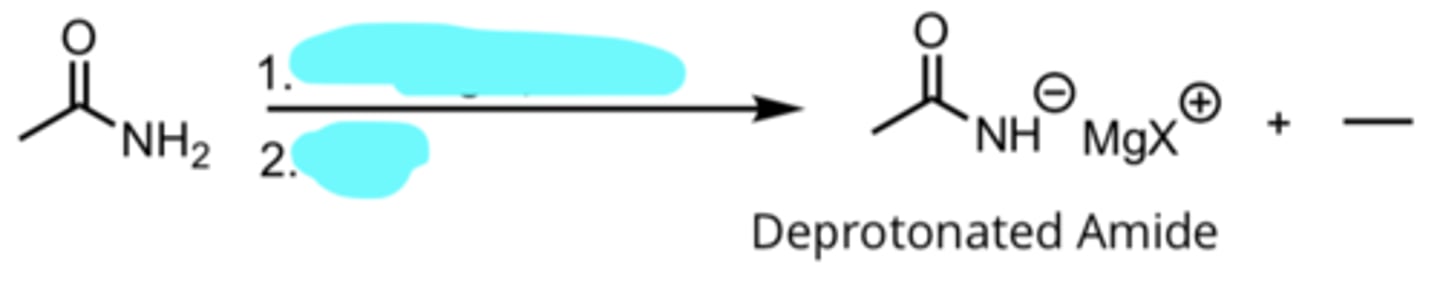
CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to an Amide
produces deprotonated amide
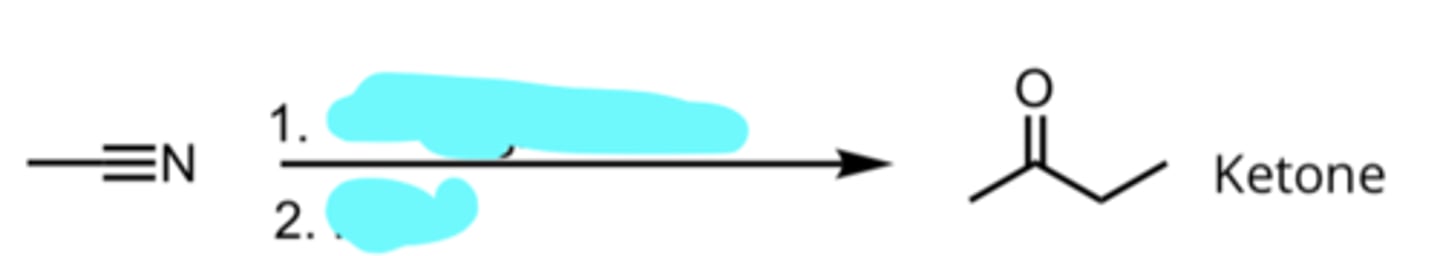
CH3CH2MgX, Ether
H3O+
Addition of a Grignard Reagent to a Nitrile
produces ketone
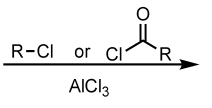
CH3CH2Cl, AlCl3
Friedel-Crafts Alkylation (Rearrangement Possible)
(Electrophilic Aromatic Substitution (EAS) Reaction)
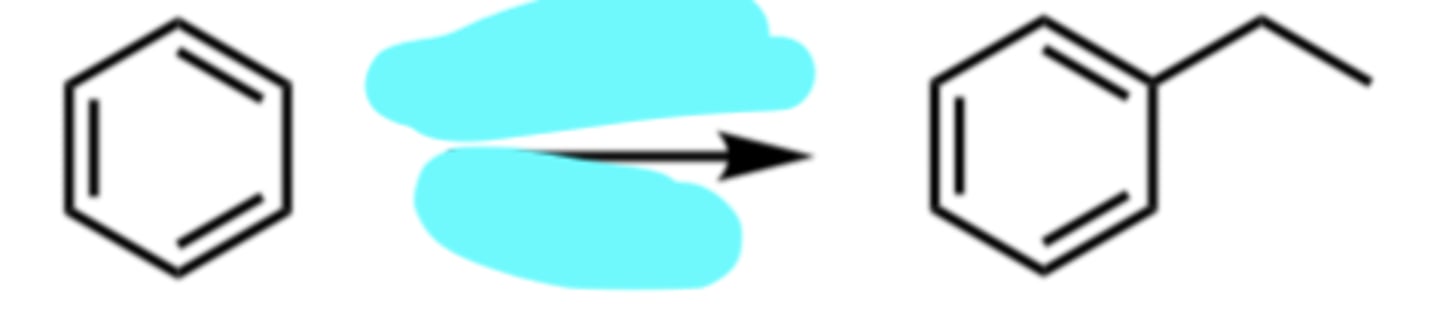
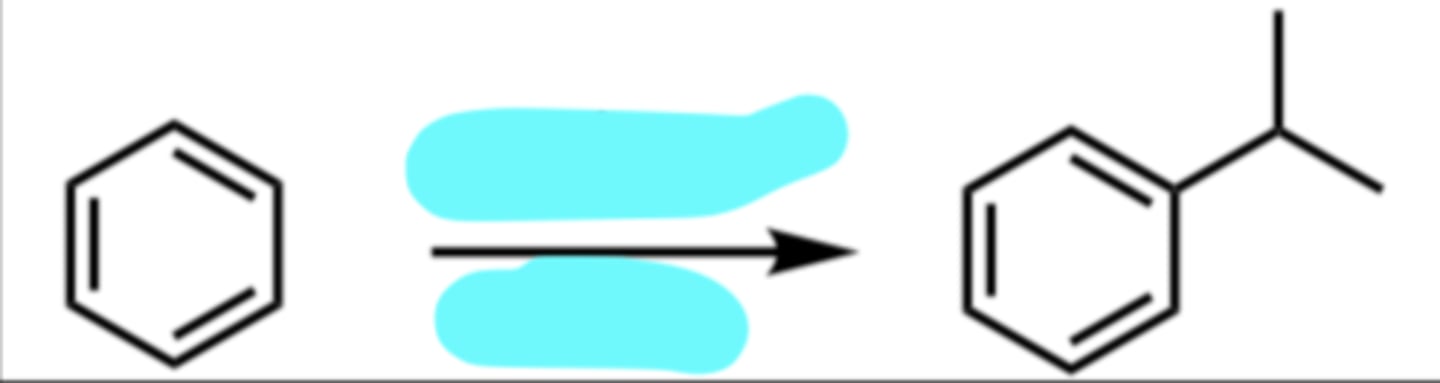
CH3CH2Cl, AlCl3
Friedel-Crafts Alkylation (Rearrangement Possible)
(Electrophilic Aromatic Substitution (EAS) Reaction)
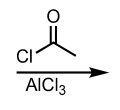
Friedel-Crafts Acylation (No Rearrangement Possible)
(Electrophilic Aromatic Substitution (EAS) Reaction)
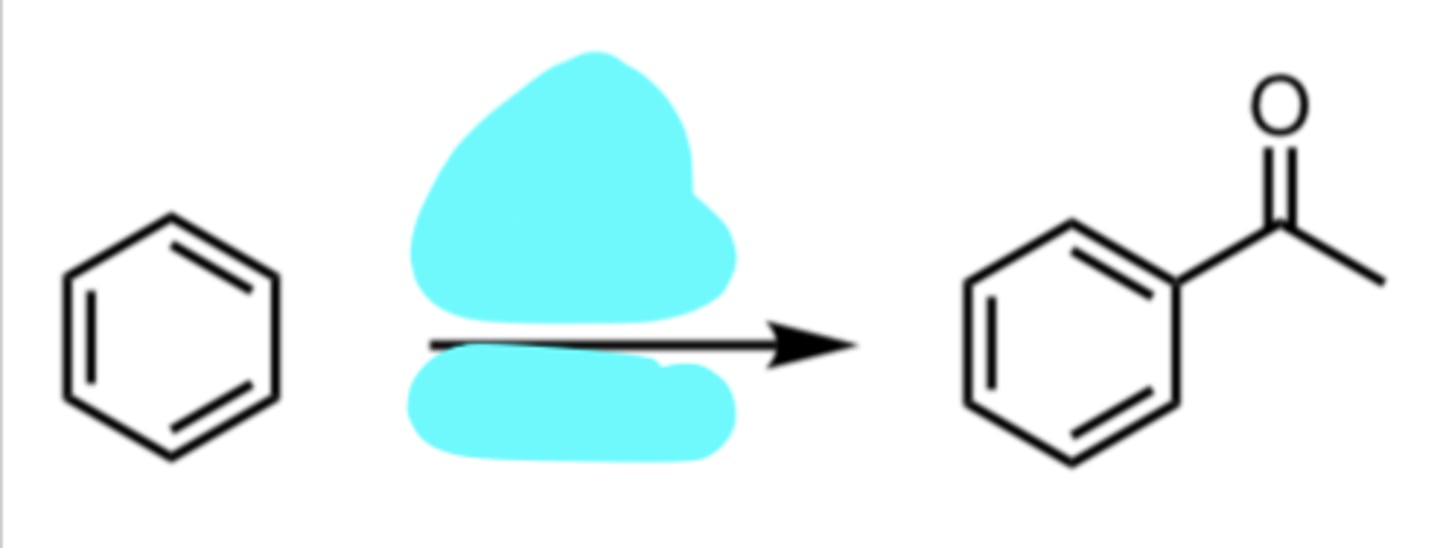
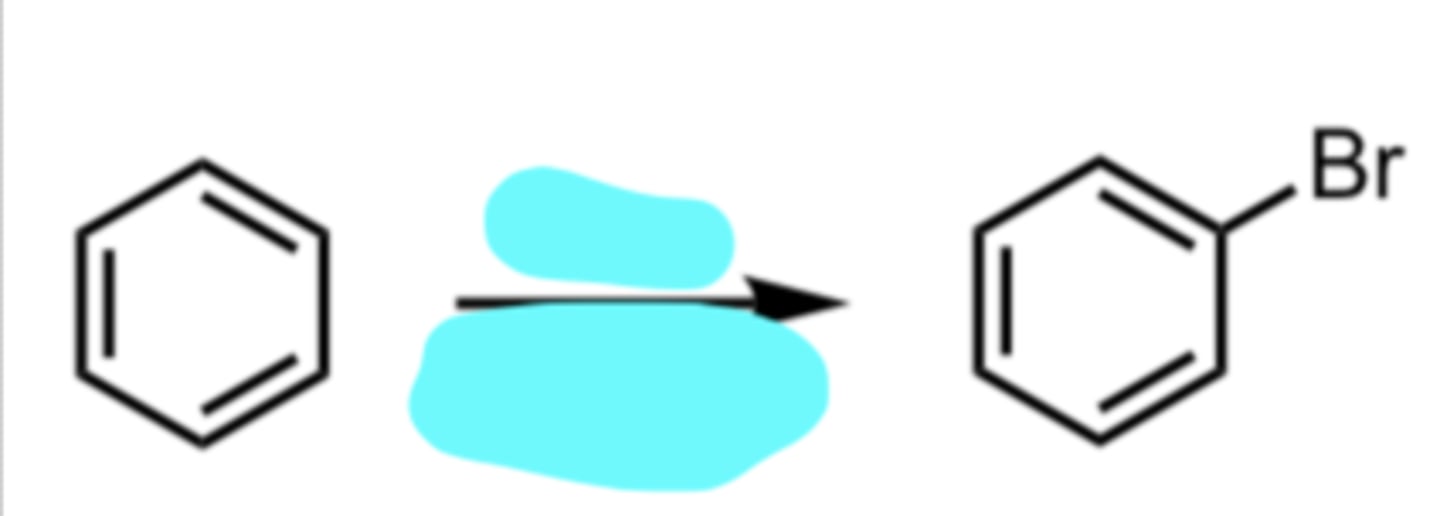
Br2, FeBr3
Bromination
(Electrophilic Aromatic Substitution (EAS) Reaction)
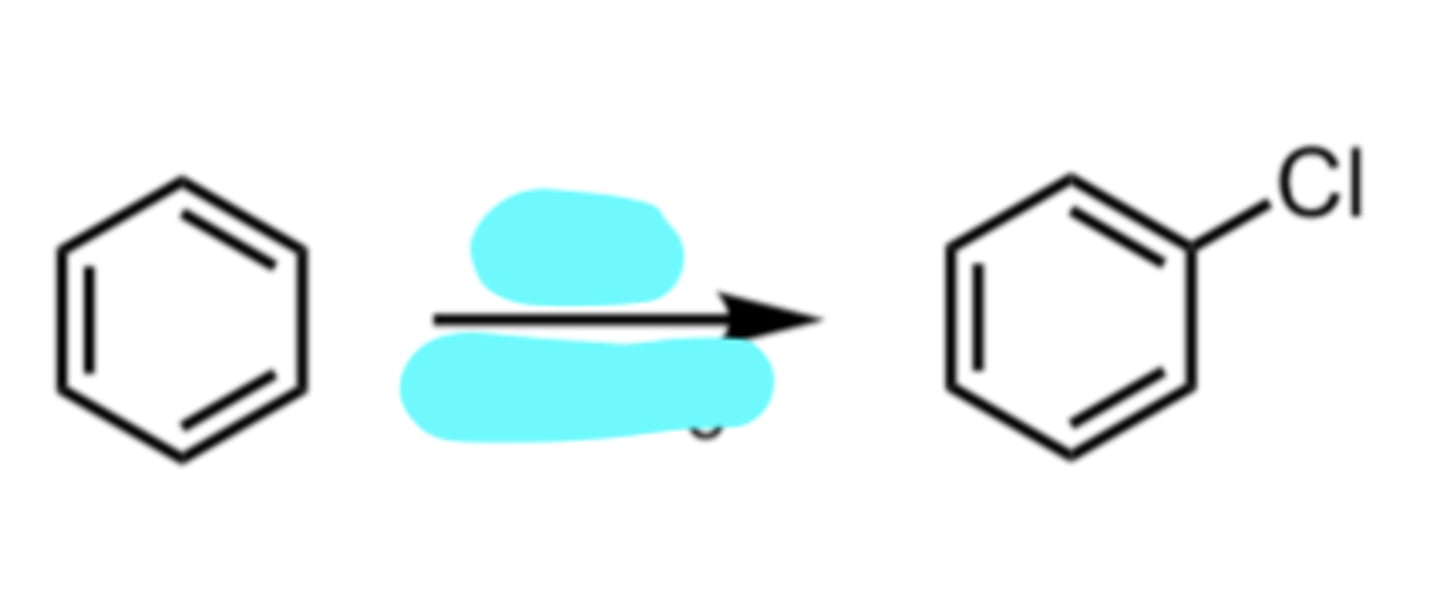
Cl2, FeCl3
Chlorination
(Electrophilic Aromatic Substitution (EAS) Reaction)
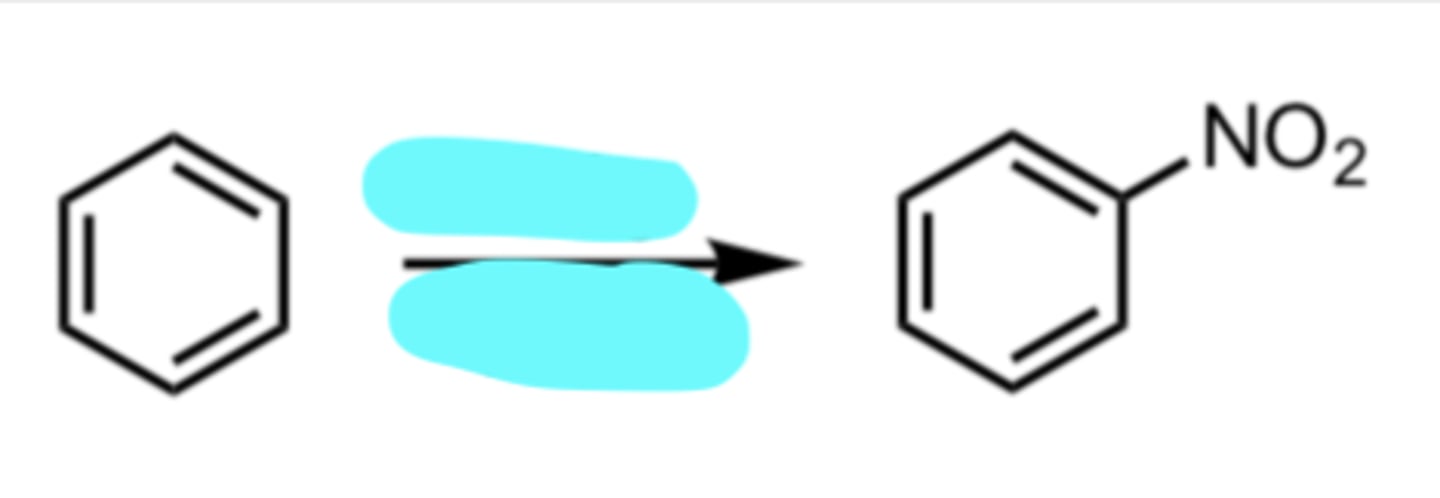
HNO3, H2SO4
Nitration
(Electrophilic Aromatic Substitution (EAS) Reaction)
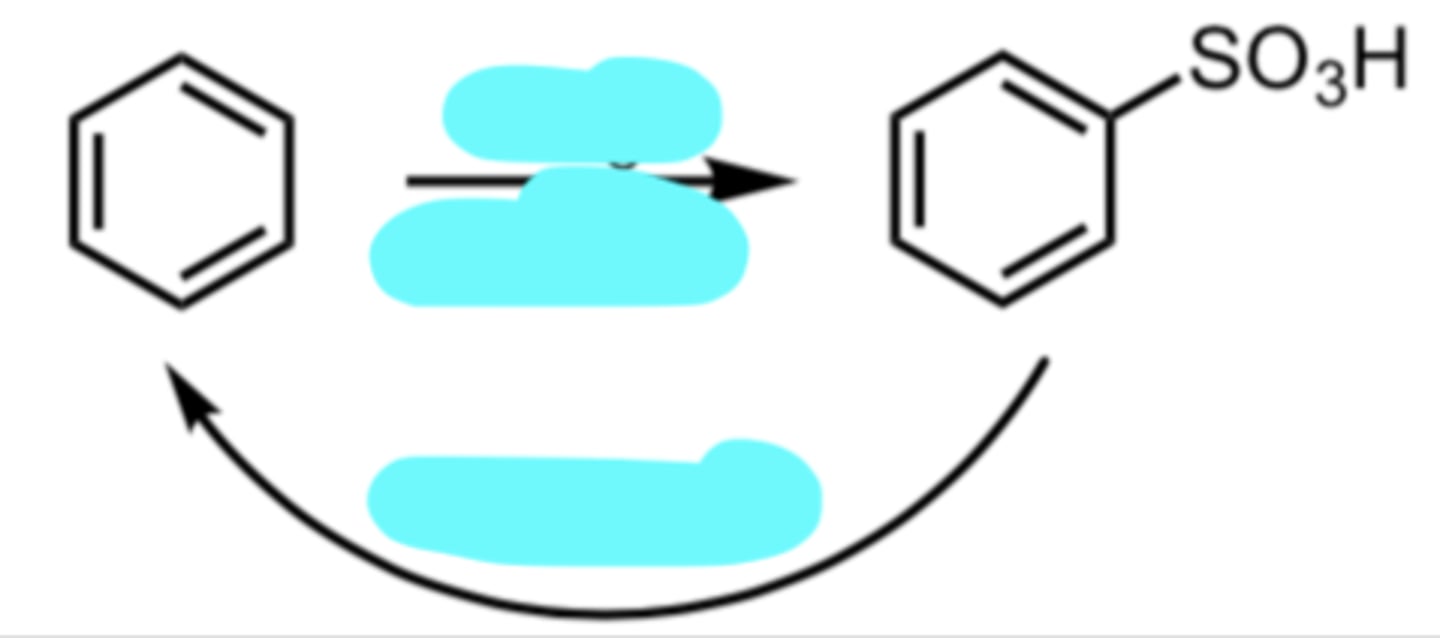
→ SO3, H2SO4
← H2SO4/heat
Sulfonation
(Electrophilic Aromatic Substitution (EAS) Reaction)
CO, HCl
AlCl3
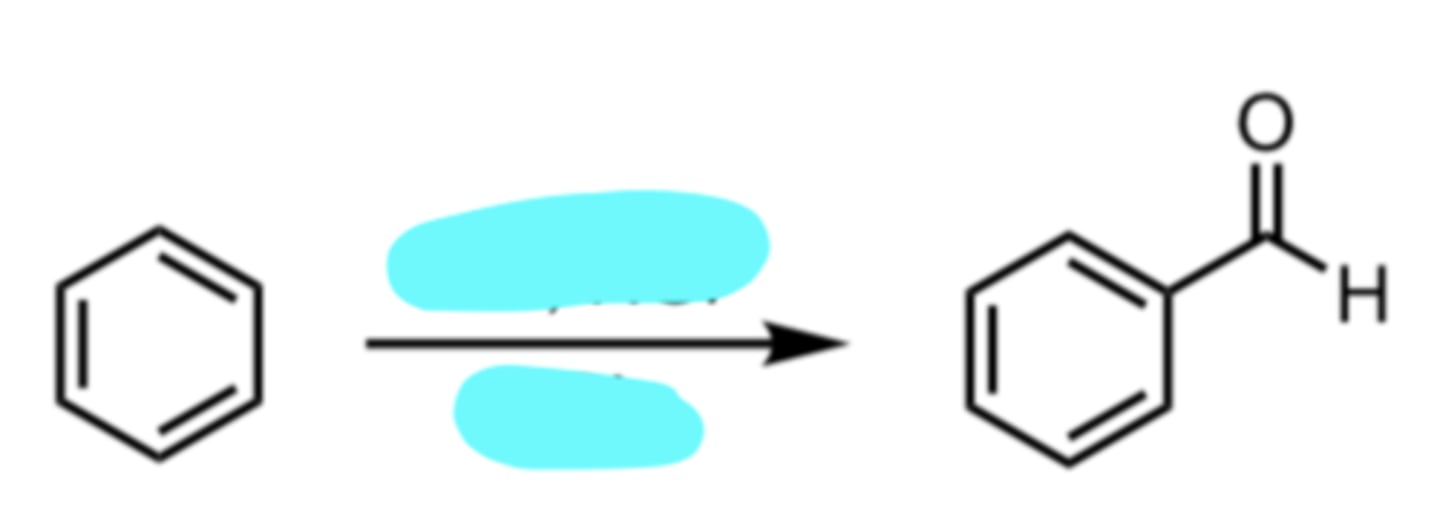
Formylation
(Electrophilic Aromatic Substitution (EAS) Reaction)
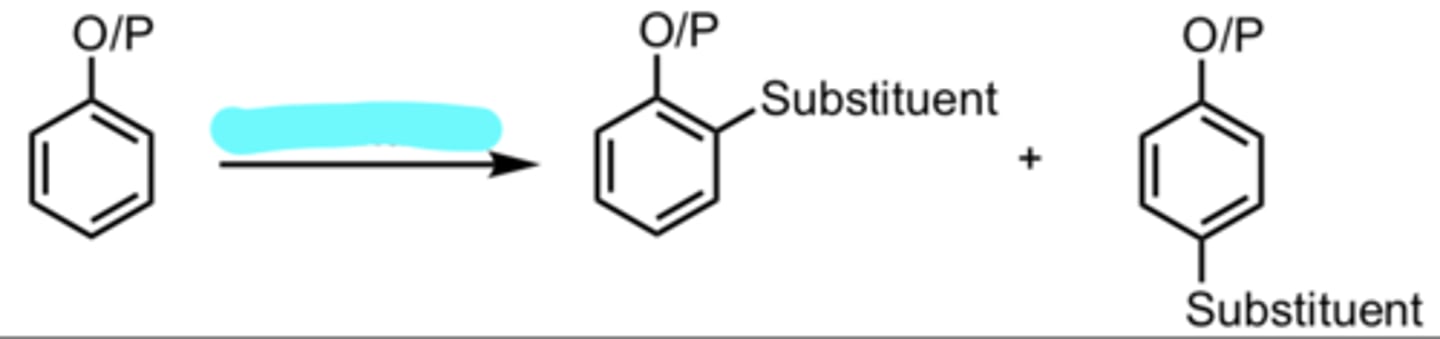
substituent
EAS with an ortho/para-directing group on Benzene
ortho/para director = electron donating group
(Electrophilic Aromatic Substitution (EAS) Reaction)
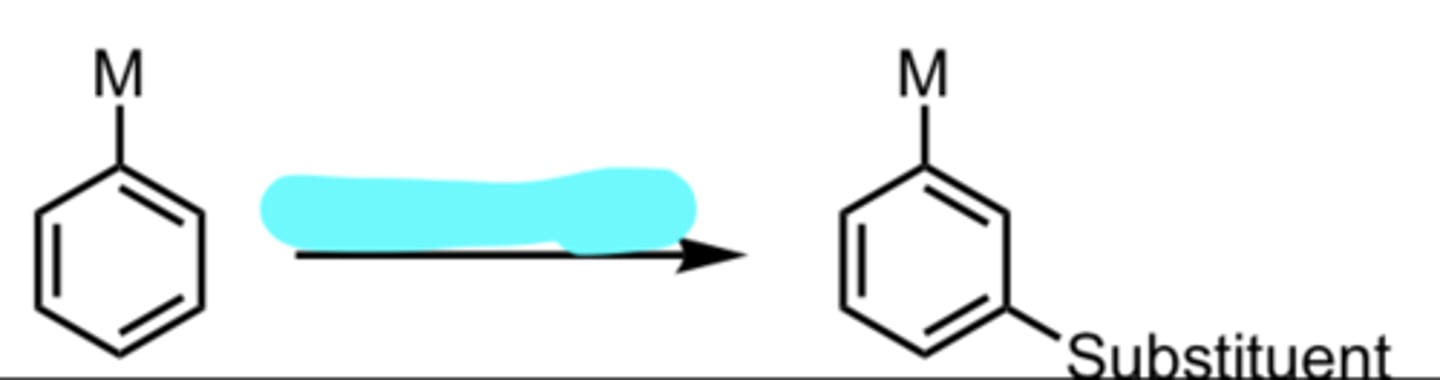
substituent
EAS with a meta-directing group on Benzene
M = electron-withdrawing group (because those code for meta)
(Electrophilic Aromatic Substitution (EAS) Reaction)
Friedel-Crafts Alkylation/Acylation with a meta-directing group or an amine on Benzene
no reaction
(Electrophilic Aromatic Substitution (EAS) Reaction)

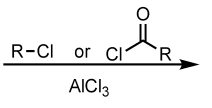
Friedel-Crafts Alkylation/Acylation with a meta-directing group or an amine on Benzene
no reaction
(Electrophilic Aromatic Substitution (EAS) Reaction)

KMnO4, -OH
H3O+, heat
or
Na2Cr2O7, H2SO4
Side-Chain Oxidation of Benzene to form Benzoic Acid
(Benzene Side-Chain Reactions)
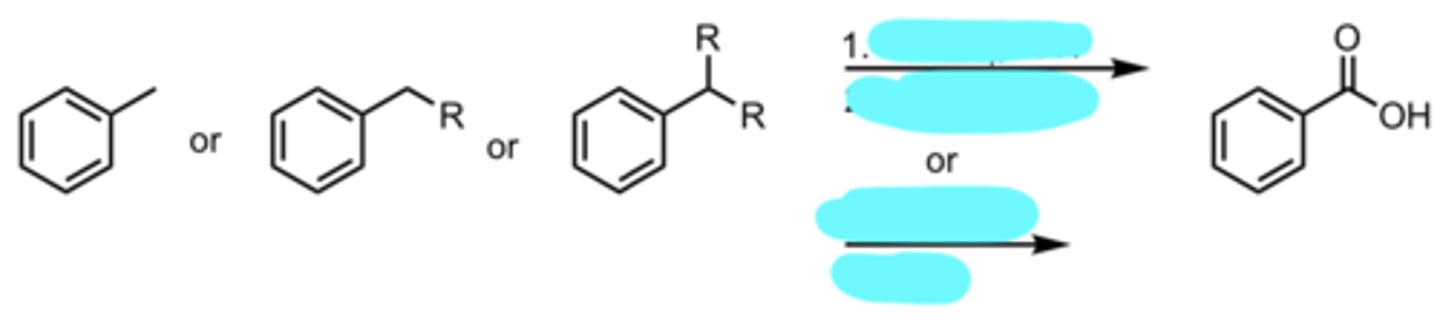
KMnO4, -OH
H3O+, heat
or
Na2Cr2O7, H2SO4
Side-Chain Oxidation of Benzene to form Benzoic Acid
no reaction (requires free hydrogen at benzylic position)
(Benzene Side-Chain Reactions)
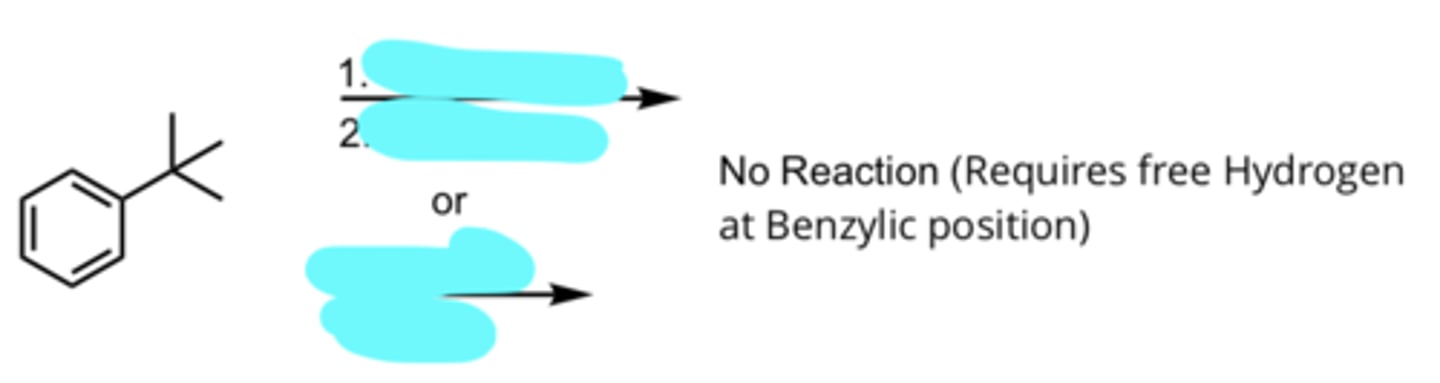
H2NNH2 or N2H4, -OH, heat
Wolff-Kishner Reduction
(Benzene Side-Chain Reactions)

Zn(Hg), HCl, Heat
or
H2, Pd/C
Clemmensen Reduction
(Benzene Side-Chain Reactions)

Zn(Hg), HCl, Heat
or
H2, Pd/C
or
Sn, HCl
Clemmensen Reduction
(Benzene Side-Chain Reactions)

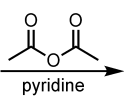
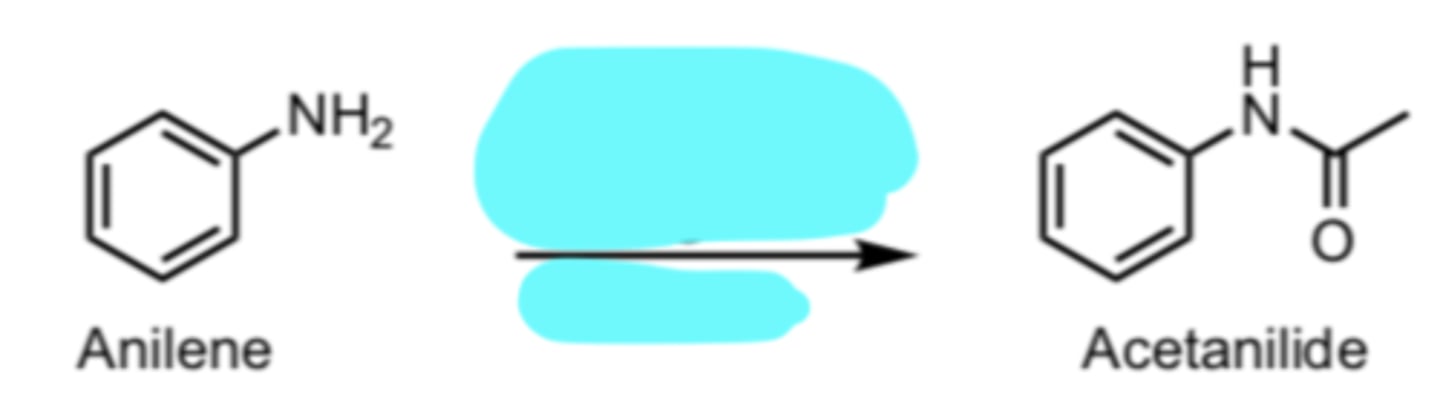
pyridine
Acetylation of Aniline using Acetic Anhydride
(Benzene Side-Chain Reactions)
NaBH4
H3O+
Reduction of an Aldehyde to a 1° Alcohol

LiAlH4
H3O+
Reduction of an Aldehyde to a 1° Alcohol

NaBH4
H3O+
Reduction of a Ketone to a 2° Alcohol

LiAlH4
H3O+
Reduction of a Ketone to a 2° Alcohol

LiAlH4
H3O+
Reduction of a Carboxylic Acid to a 1° Alcohol

LiAlH4
H3O+
Reduction of an Ester to a 1° Alcohol
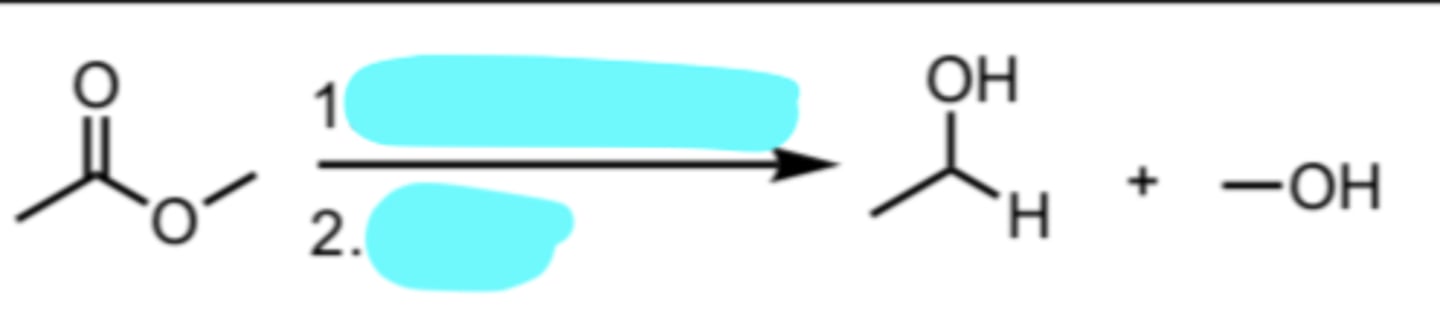
DIBAL-H, -78°C
H2O
Reduction of an Ester to an Aldehyde

LiAlH4
H3O+
or
NaBH4, EtOH
H3O+
Reduction of an Ester to a 1° Alcohol
Reduction of an Acyl Chloride to a 1° Alcohol

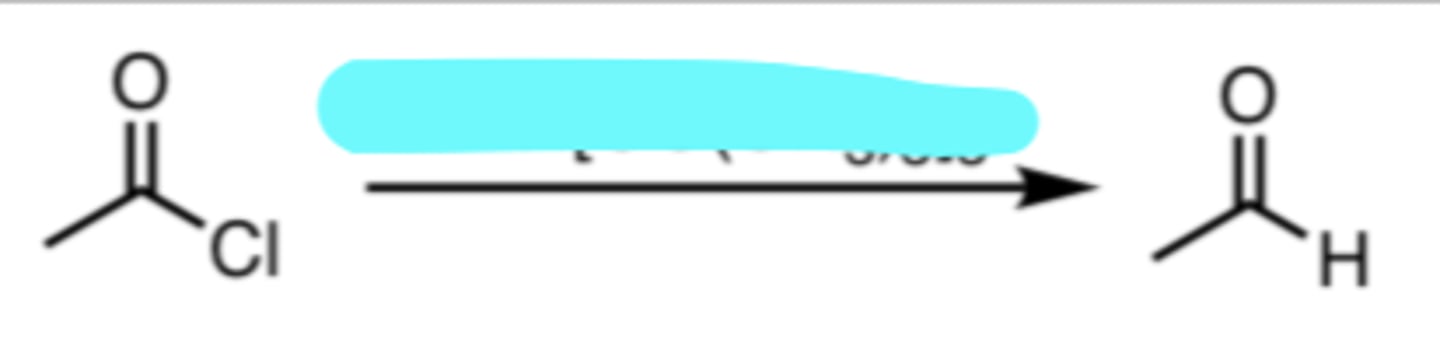
LiAlH[OC(CH3)3]3
Reduction of an Acyl Chloride to an Aldehyde
LiAlH4
H3O+
Reduction of an Amide to an Amine

Br2
NaOH
Hofmann Rearrangement
takes out the carbon with a double-bonded O on it
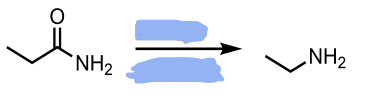
LiAlH4
H3O+
Reduction of a Nitrile to an Amine

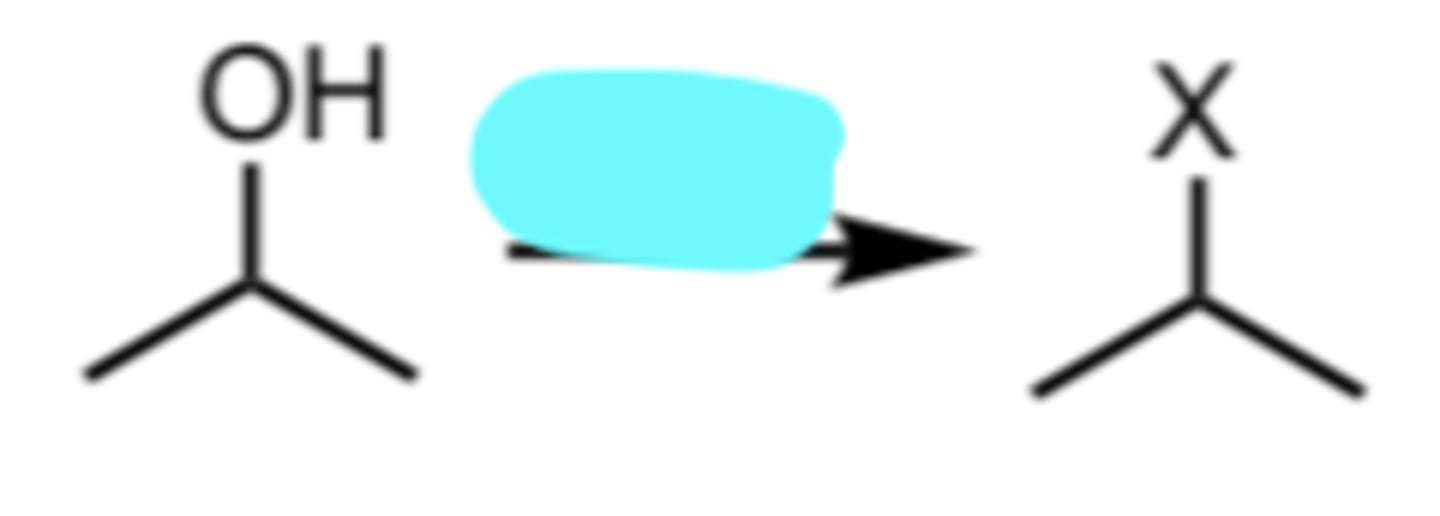
HX
Conversion of a 2°/3° Alcohol to an alkyl halide via SN1
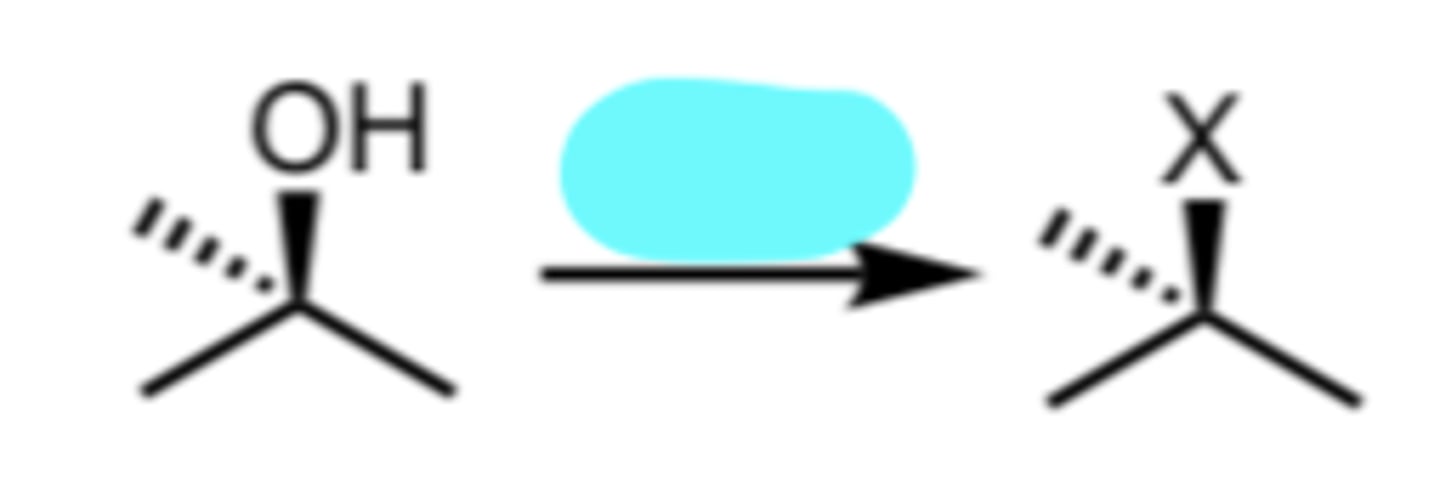
HX
Conversion of a 2°/3° Alcohol to an alkyl halide via SN1
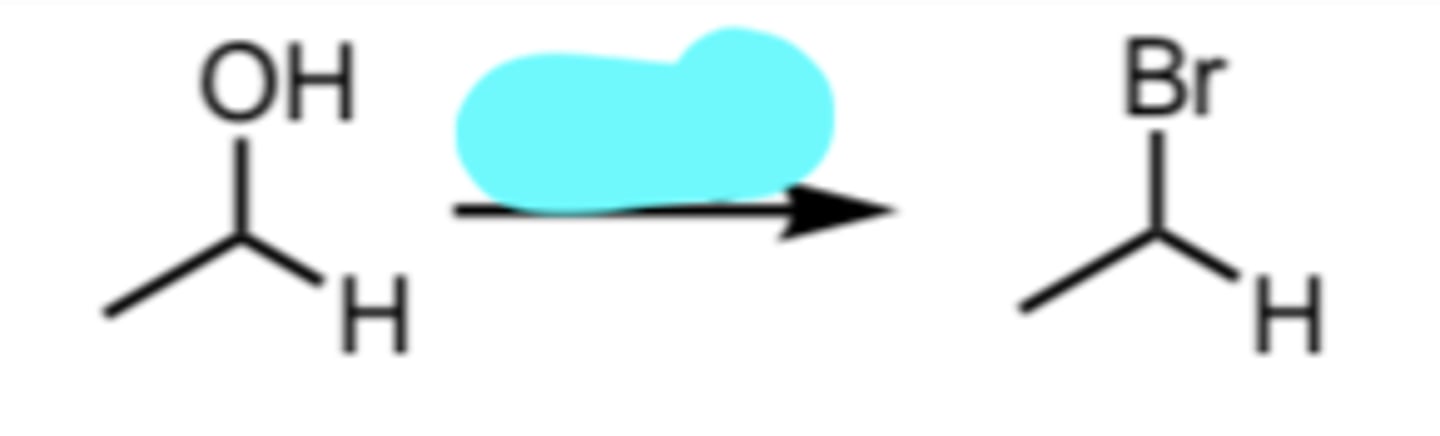
PBr3
Conversion of a 1°/2° Alcohol to an alkyl bromide via SN2
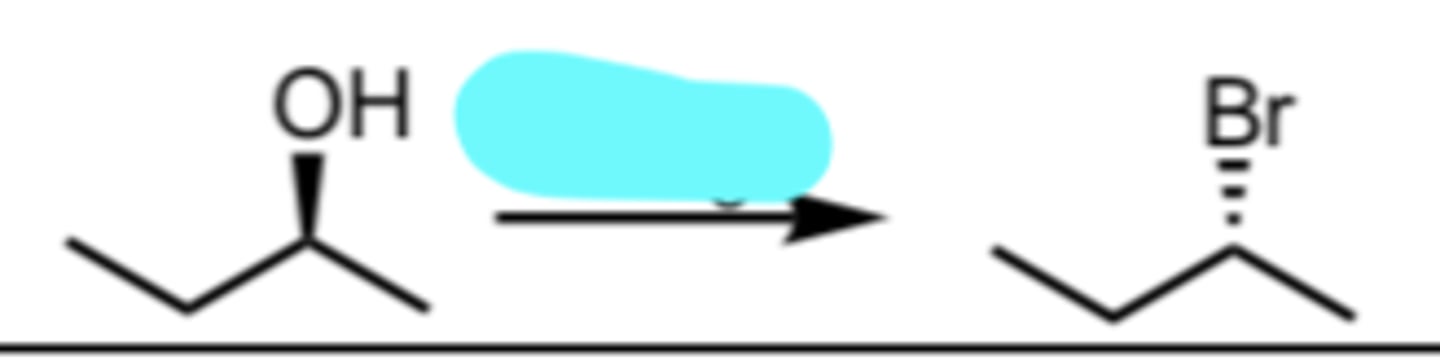
PBr3
Conversion of a 1°/2° Alcohol to an alkyl bromide via SN2
SOCl2, pyridine (or PCl3 or PCl5)
Conversion of a 1°/2° Alcohol to an alkyl chloride via SN2
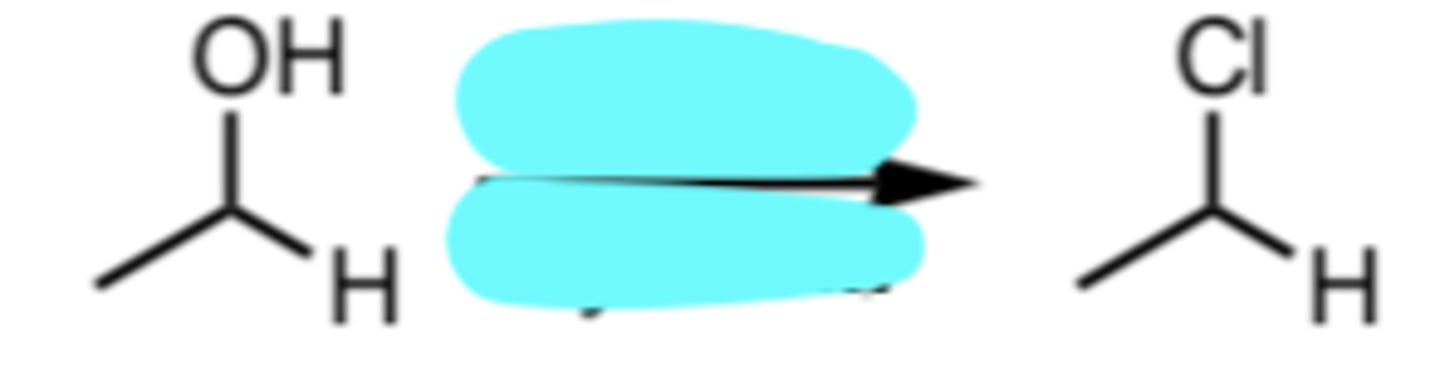
SOCl2, pyridine
Conversion of a 1°/2° Alcohol to an alkyl chloride via SN2

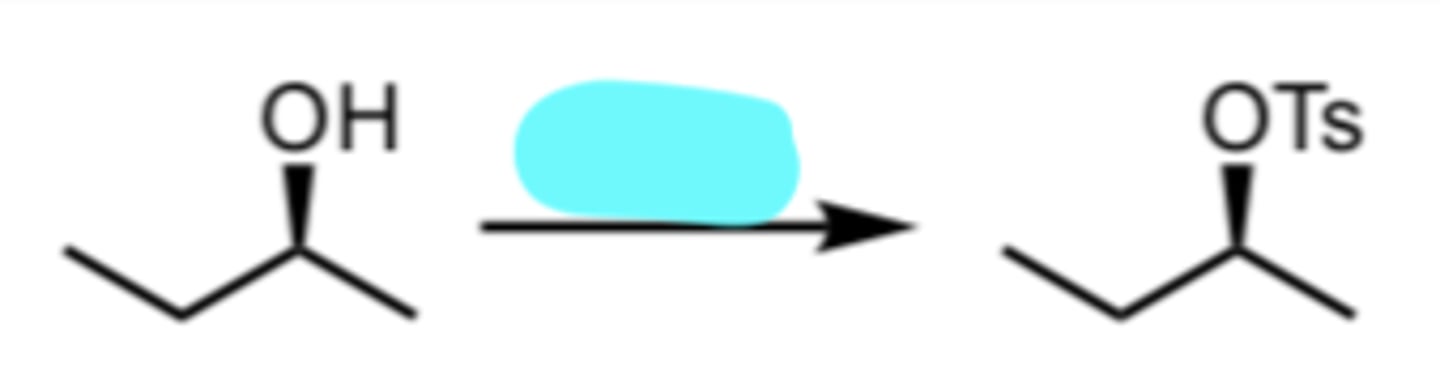
TsCl
Conversion of an Alcohol to a Tosylate Ester
retention of stereochemistry
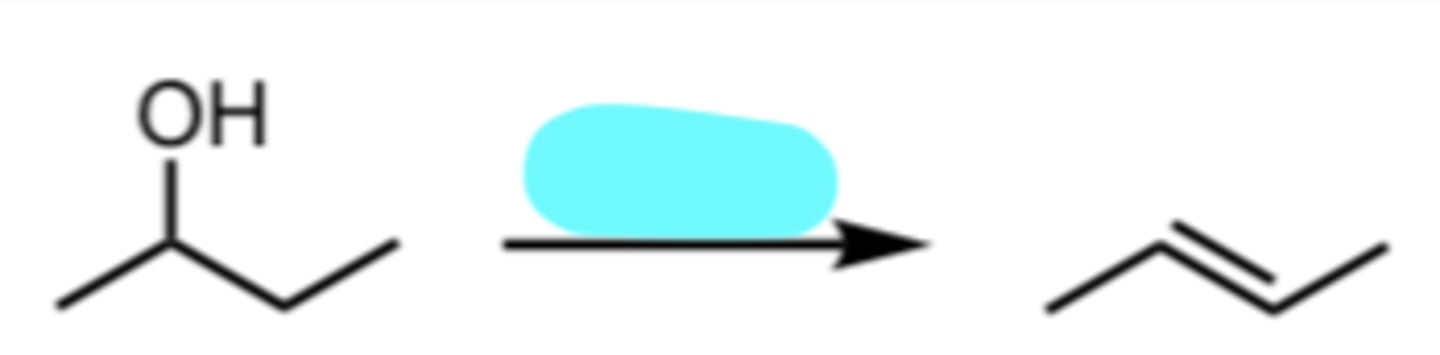
H3O+
Acid-catalyzed Dehydration of an Alcohol
Zaitsev’s Rule: the major product is the most substituted alkene
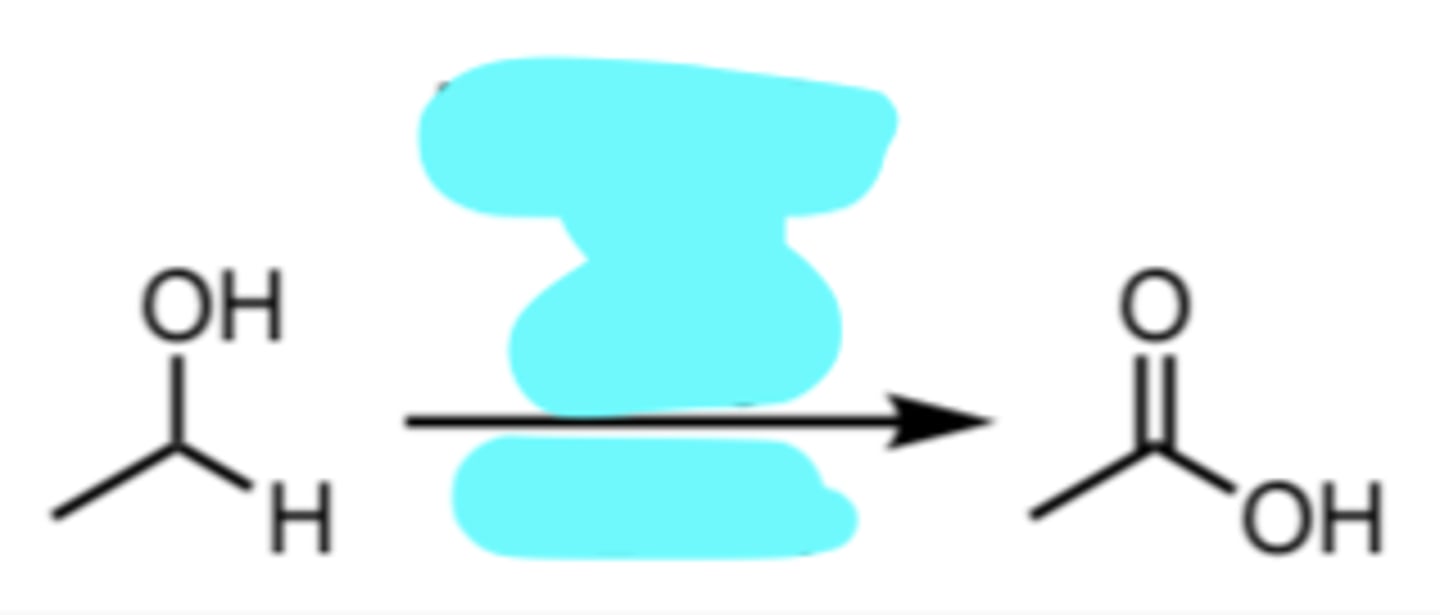
Na2Cr2O7 or CrO3, H2SO4
Chromic Acid Oxidation of a 1° Alcohol to a Carboxylic Acid
Na2Cr2O7 or CrO3, H2SO4
Chromic Acid Oxidation of a 2° Alcohol to a Ketone
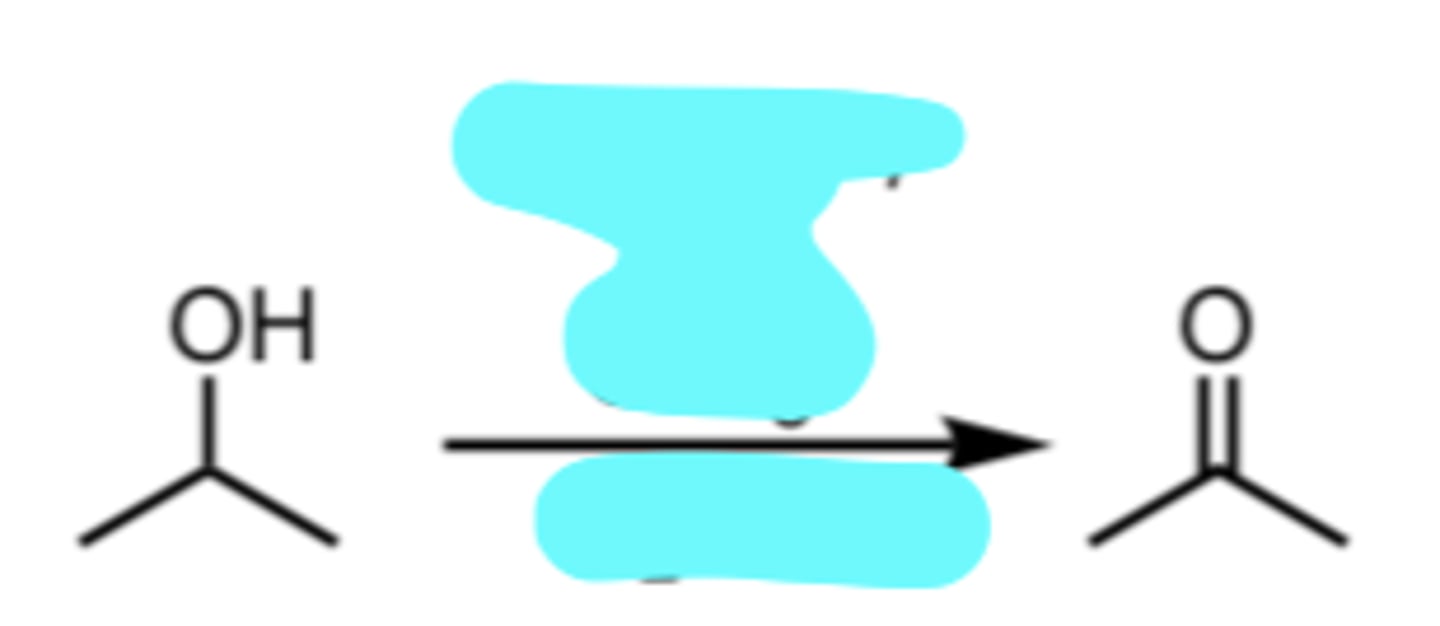
Na2Cr2O7 or CrO3, H2SO4
Chromic Acid Oxidation of an Aldehyde to a Carboxylic Acid
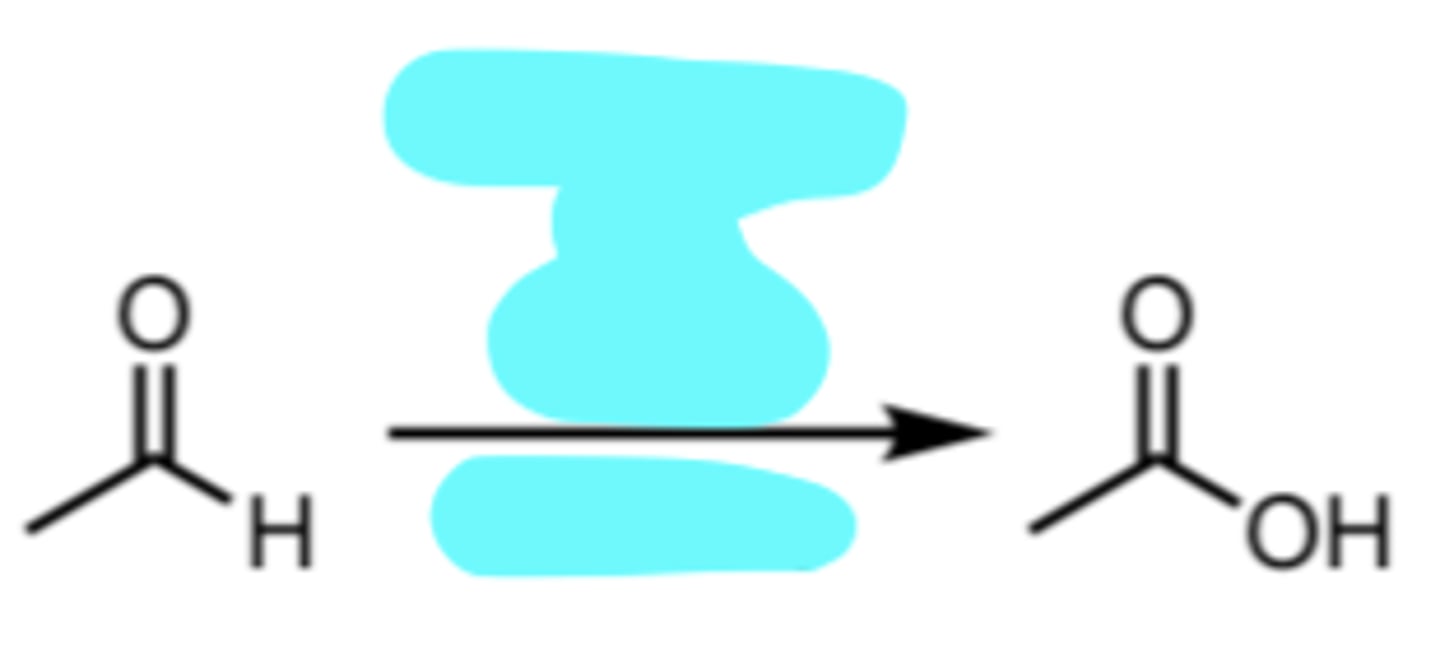
PCC or DMP
PCC or DMP Oxidation of a 1° Alcohol to an Aldehyde

PCC or DMP
PCC or DMP Oxidation of a 2° Alcohol to an Aldehyde

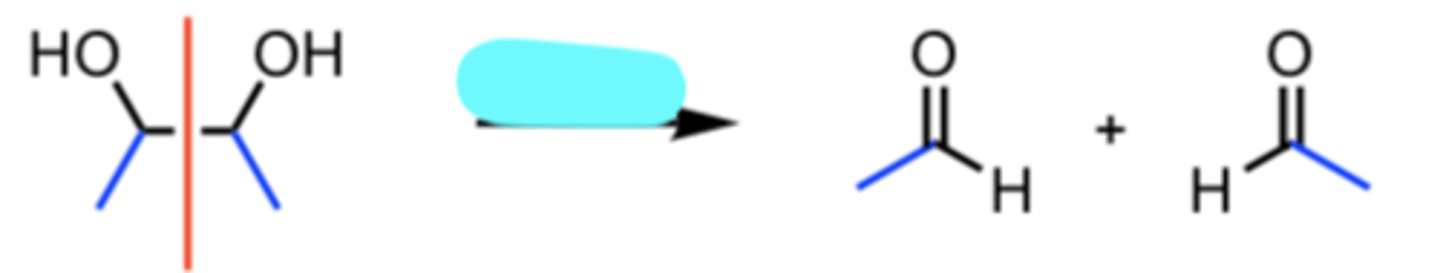
HIO4
Oxidative Cleavage of a 1,2 Diol
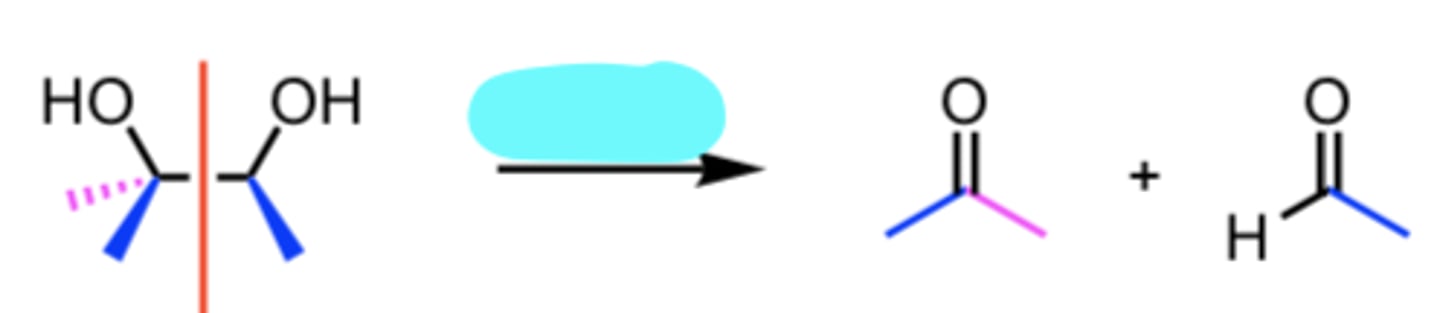
HIO4
Oxidative Cleavage of a 1,2 Diol
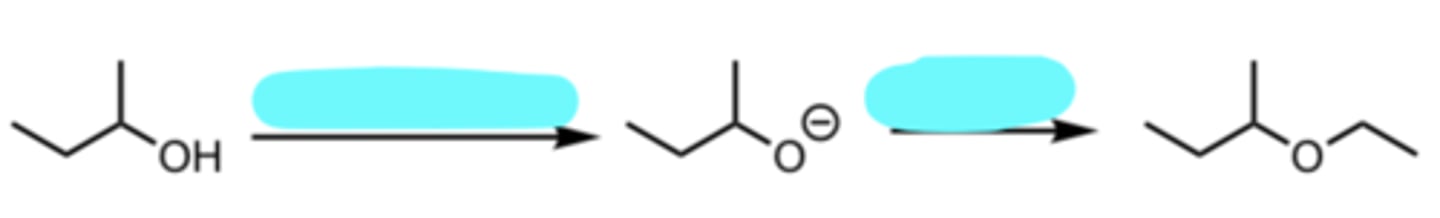
NaH, Na, or K
Williamson Ether Synthesis via SN2
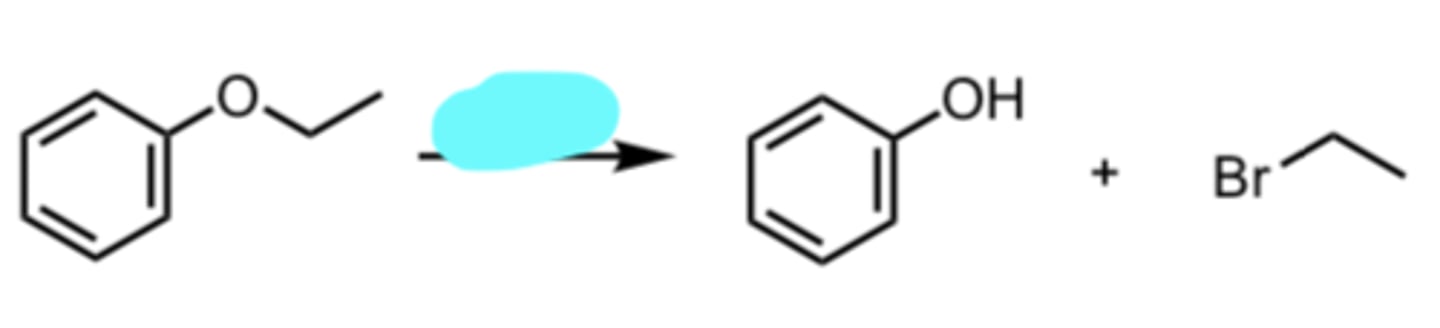
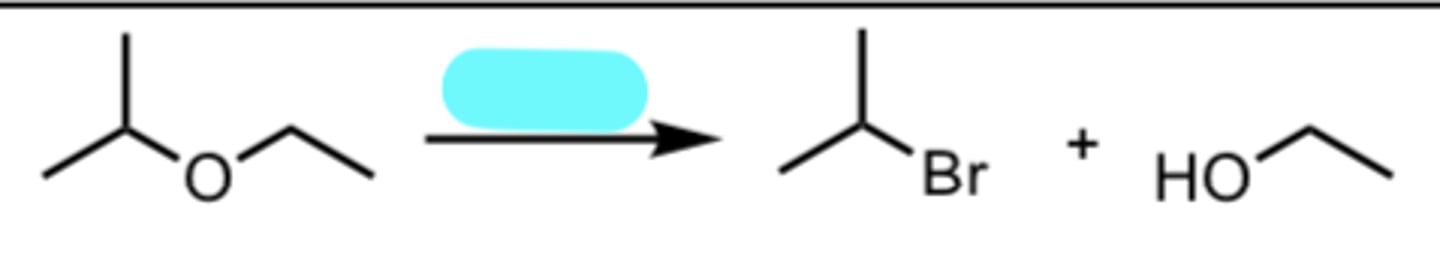
HBr
Acid-catalyzed Cleavage of Ethers when one side is 2°/3°
nucleophile attacks MORE substituted side via SN1
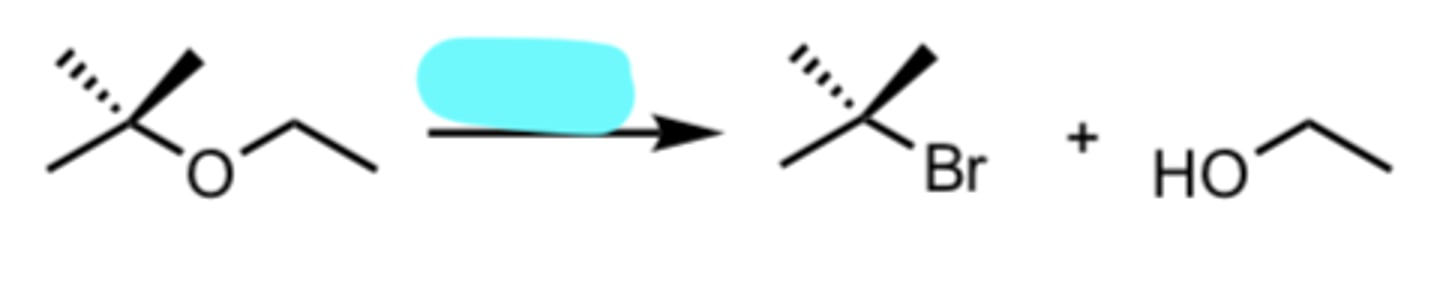
HBr
Acid-catalyzed Cleavage of Ethers when one side is 2°/3°
nucleophile attacks MORE substituted side via SN1
HBr
Acid-catalyzed Cleavage of Ethers when neither side is 2°/3°
nucleophile attacks LESS substituted side via SN2