BIOC 4331 Lecture 40
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
What is the general property that defines lipids?
Lipids are soluble in organic solvents and generally insoluble in water
Why are fatty acids such energy-rich molecules?
They are highly reduced hydrocarbons
Oxidation to CO2 and H2O is highly exergonic
What is the general formula of a fatty acid?
CH3(CH2)nCOOH
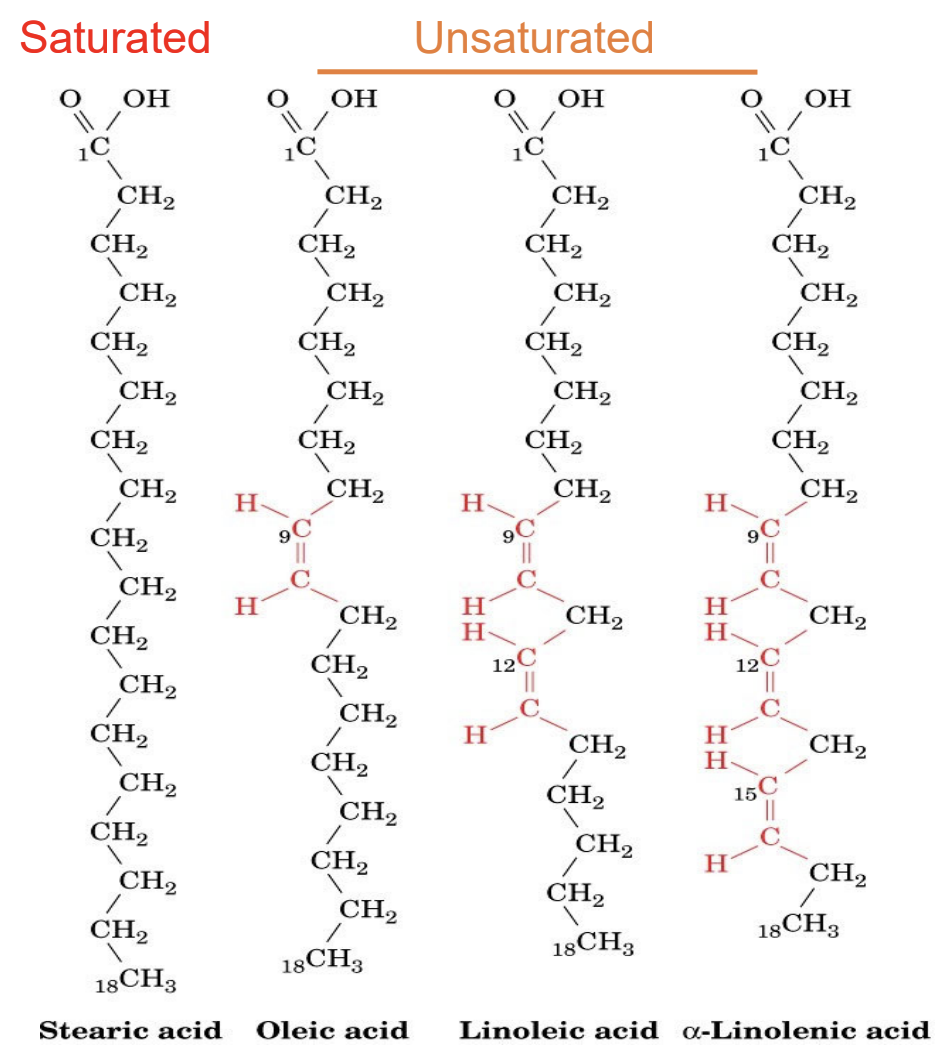
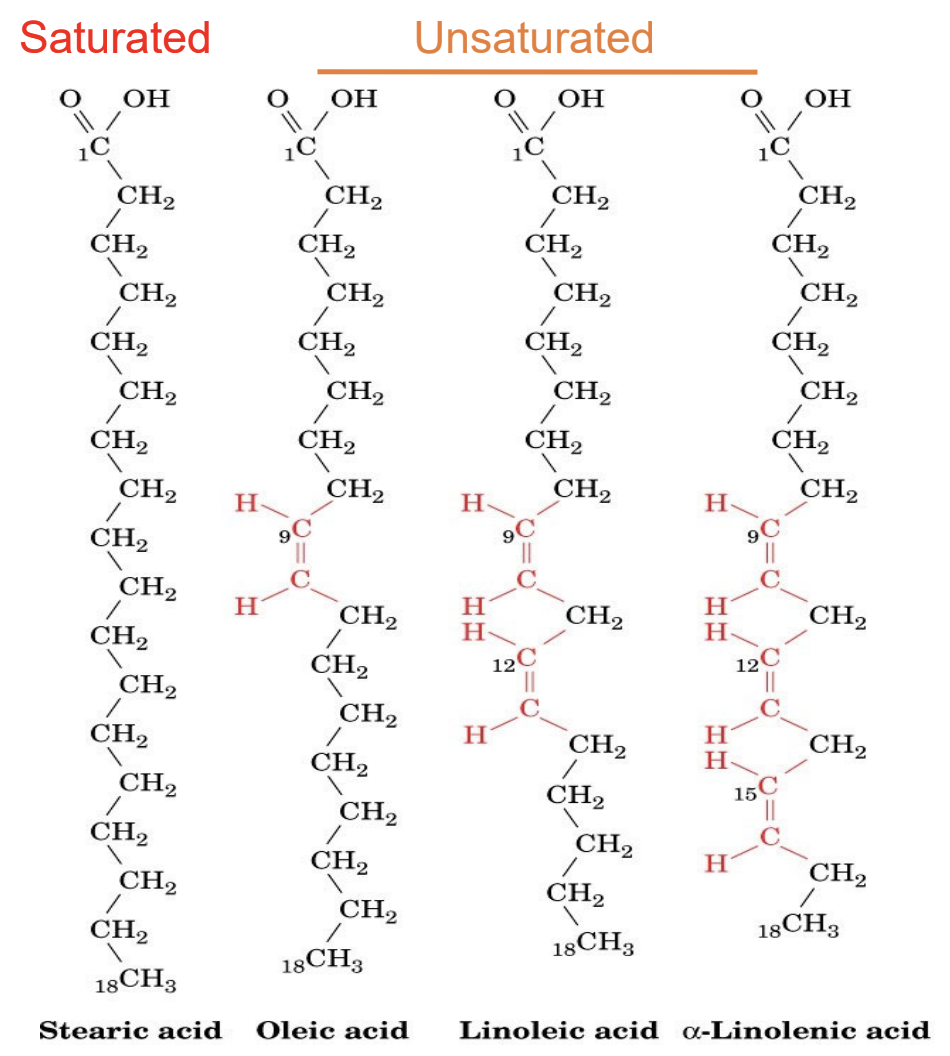
Difference between saturated, unsaturated, and polyunsaturated fatty acids?
Saturated: no double bonds
Unsaturated: ≥1 double bond
Polyunsaturated: multiple double bonds
What fatty acid chain lengths are most common in biology?
C16 and C18 (usually even number of carbons)
What is the usual geometry of fatty acid double bonds?
Cis
What does a cis double bond do to a fatty acid chain?
Introduces a kink (~30°), disrupting packing
How do chain length and unsaturation affect melting point?
Longer chains ↑ melting point
More double bonds ↓ melting point
Why do saturated fatty acids pack tightly?
Straight chains allow tight packing
Cis double bonds disrupt packing
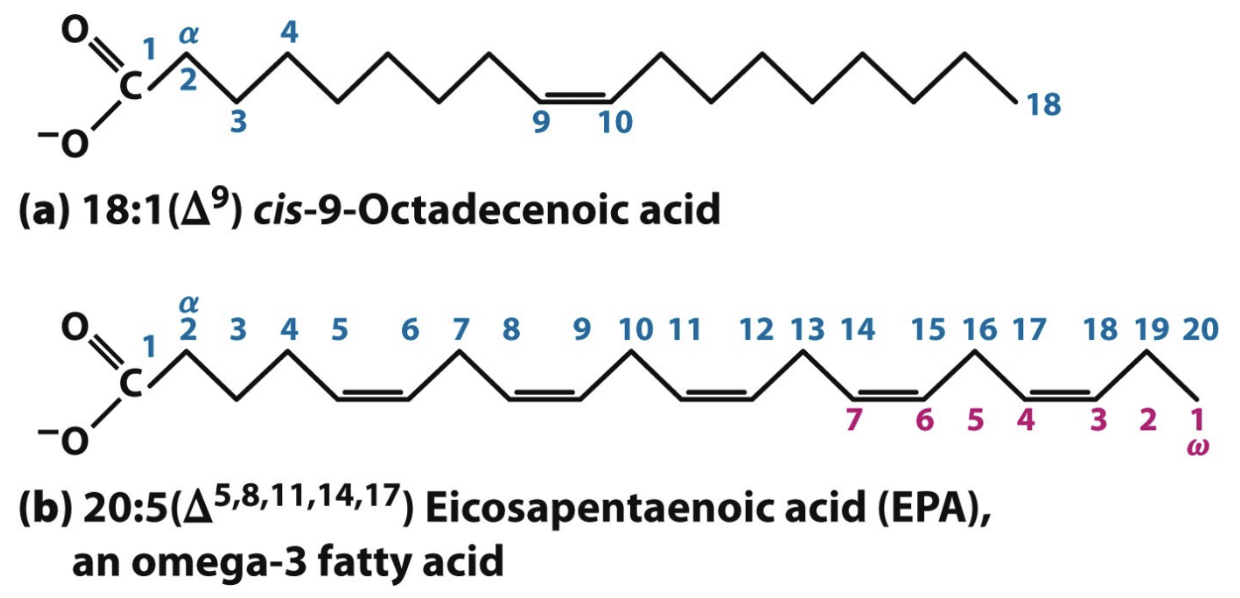
In fatty acid nomenclature, where is carbon 1?
Carboxyl carbon; next is alpha carbon
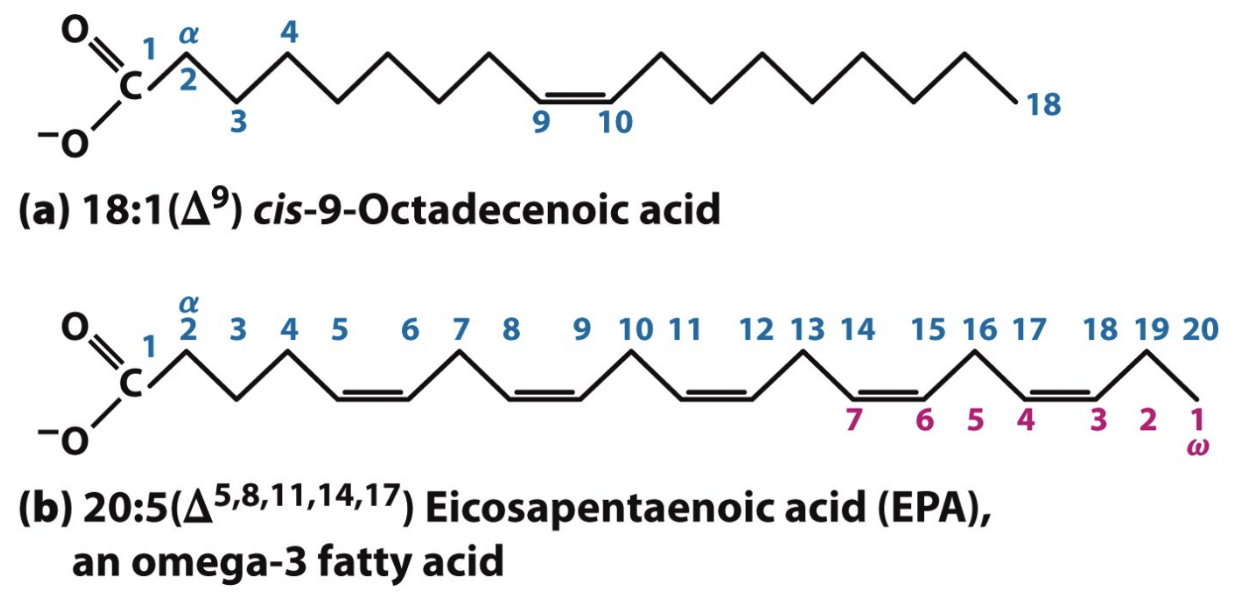
What does 18:1(Δ9) mean?
18 carbons, 1 double bond at carbon 9 (from carboxyl end)
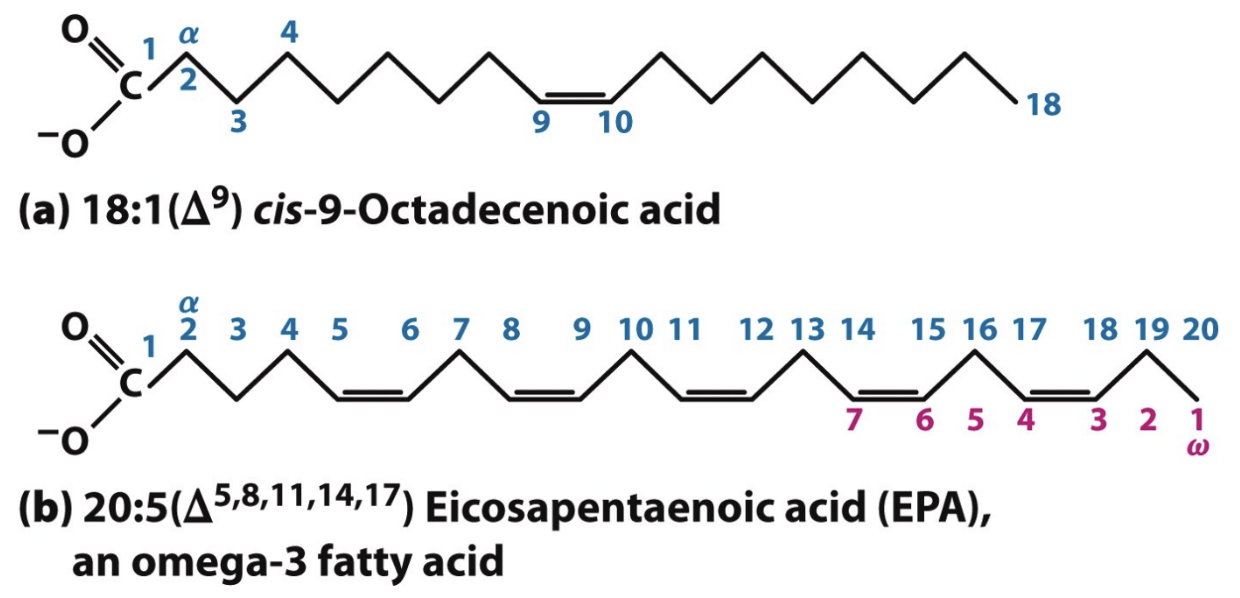
Where do you start counting in PUFAS nomenclature?
From terminal methyl (ω) carbon
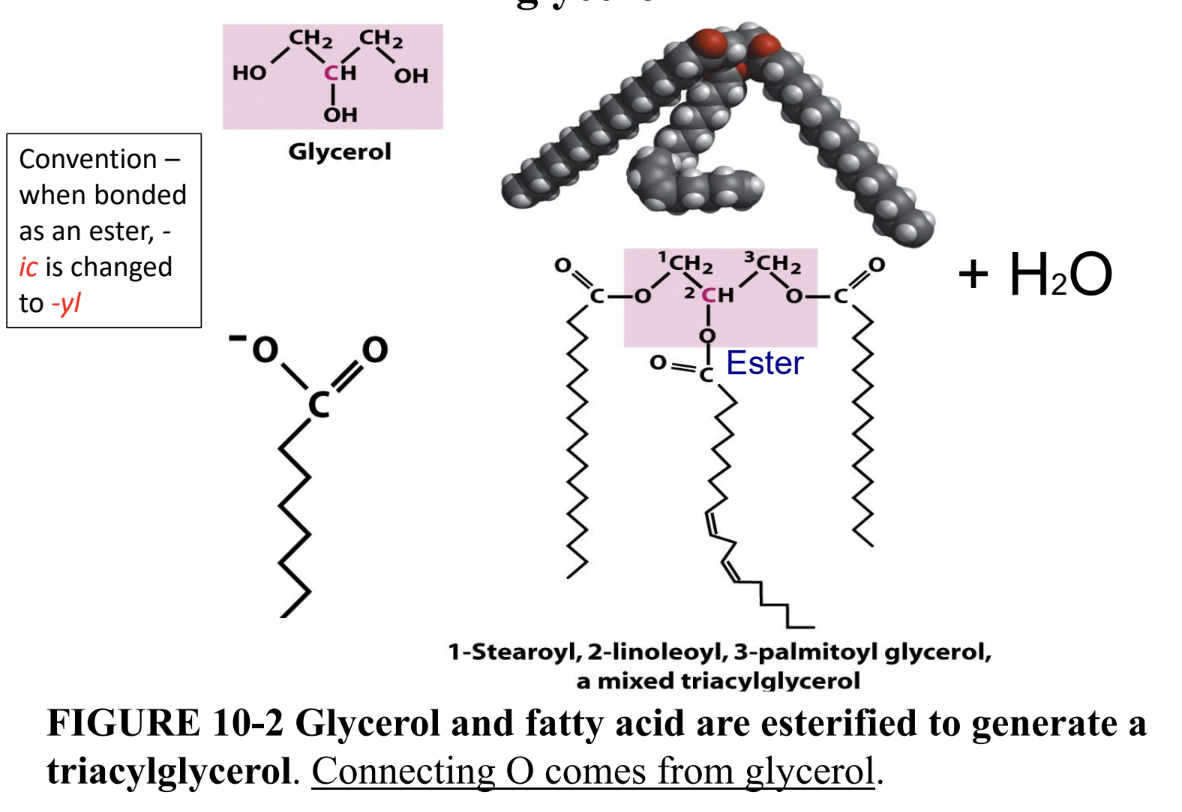
What are triacylglycerols?
Storage lipids: 3 fatty acids esterified (ester bonds) to glycerol
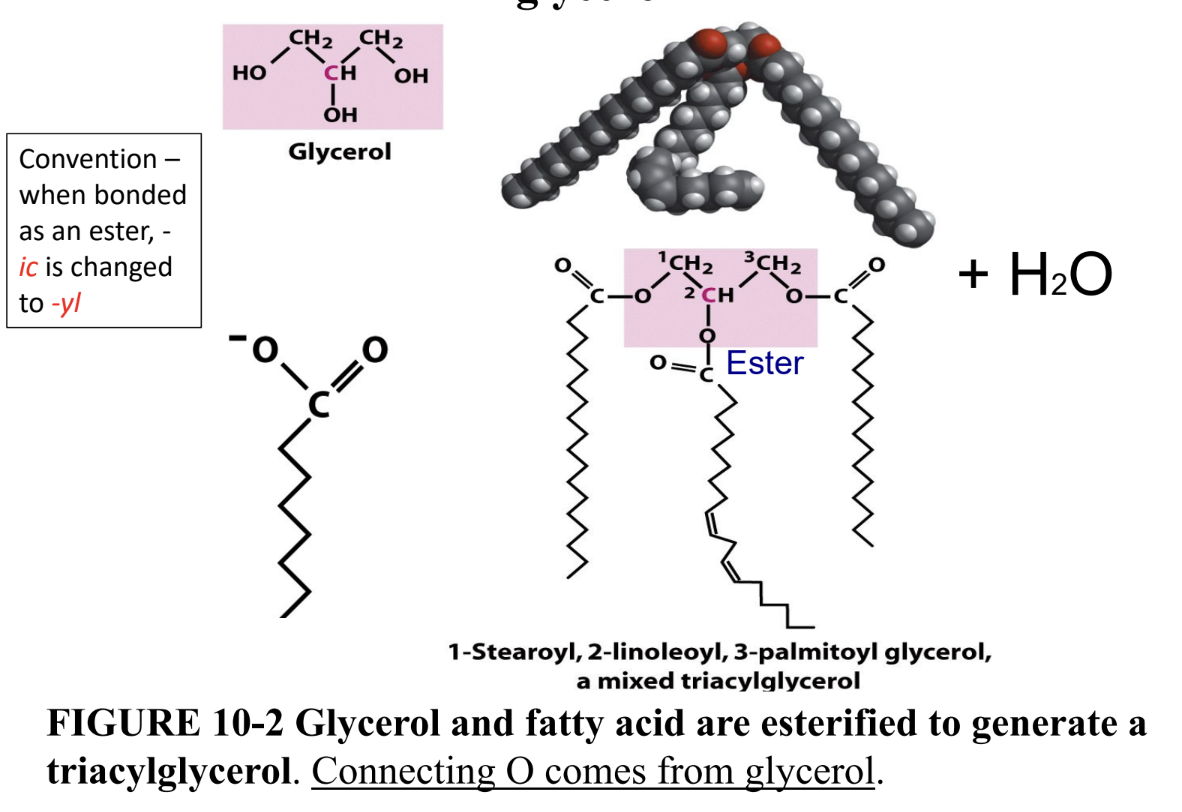
Why are triacylglycerols better energy storage than polysaccharides?
More reduced (more energy) + hydrophobic (no water weight)
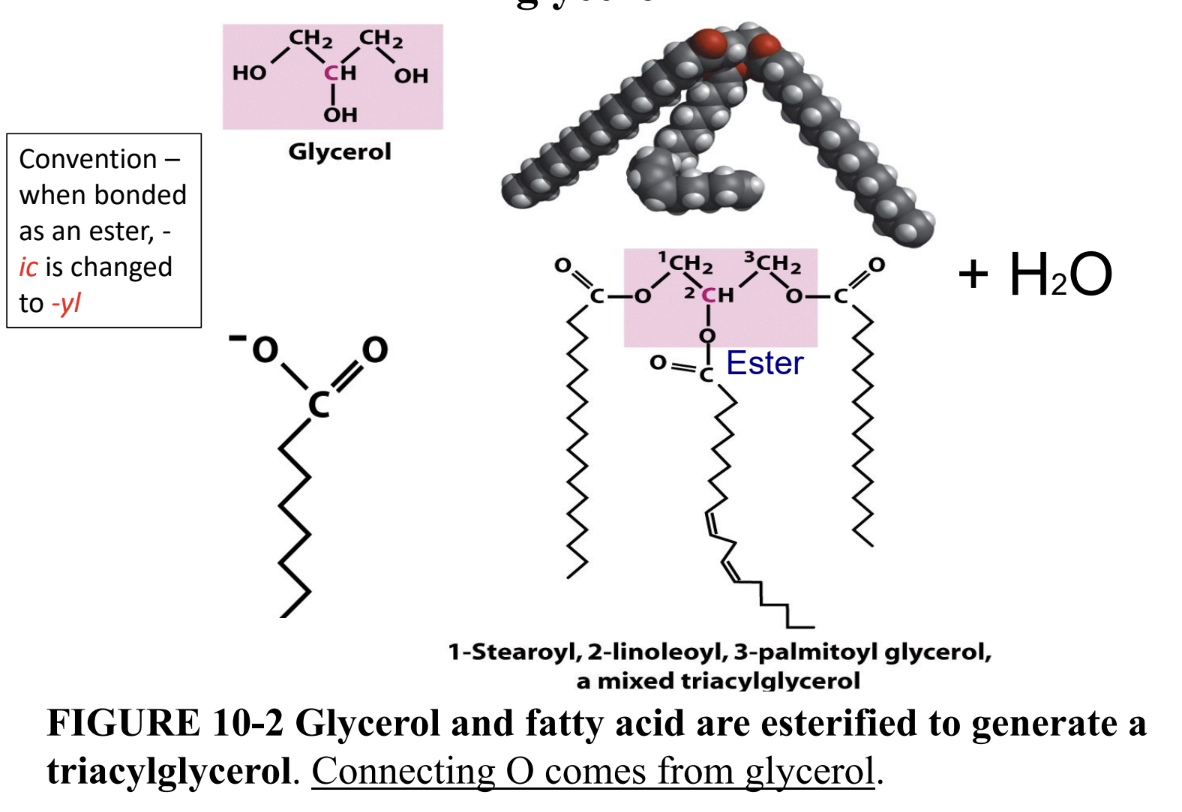
Why aren’t triacylglycerols membrane components?
Fully hydrophobic, not amphipathic
Why do partially hydrogenated oils like Crisco and margarine contain high amounts of trans fat?
Partial hydrogenation of vegetable oils does not fully saturate all double bonds (partially hydrogenated).
Instead, it often causes cis-trans isomerization of unsaturated fatty acids, producing trans fats.
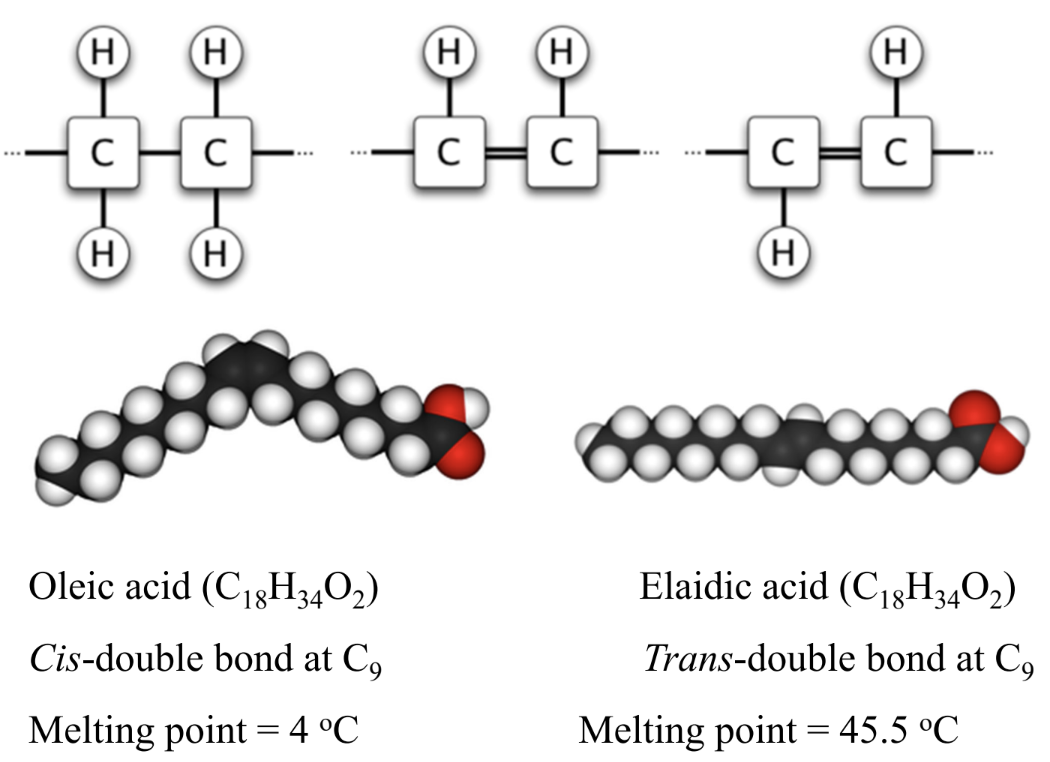
Why do trans fats have higher melting points than cis?
More linear (less cis double bonds) → tighter packing
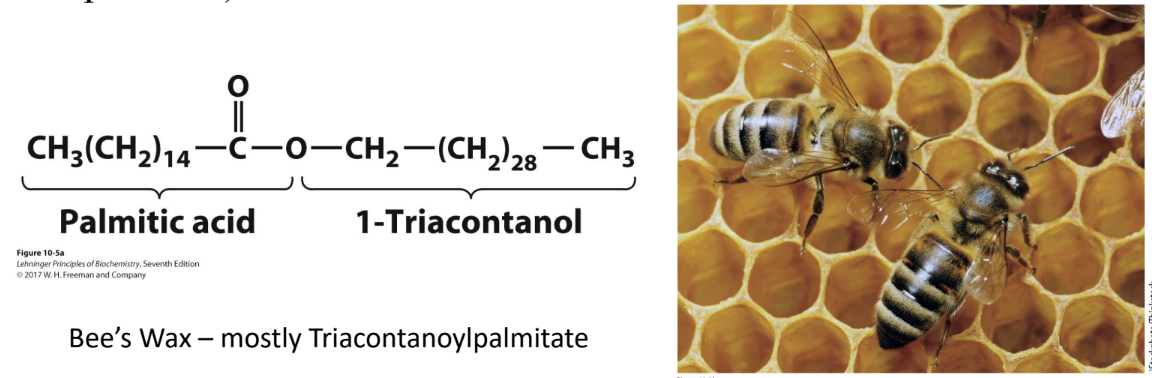
What are biological waxes?
Esters of long-chain fatty acids + long-chain alcohols
Protective, water-repellent
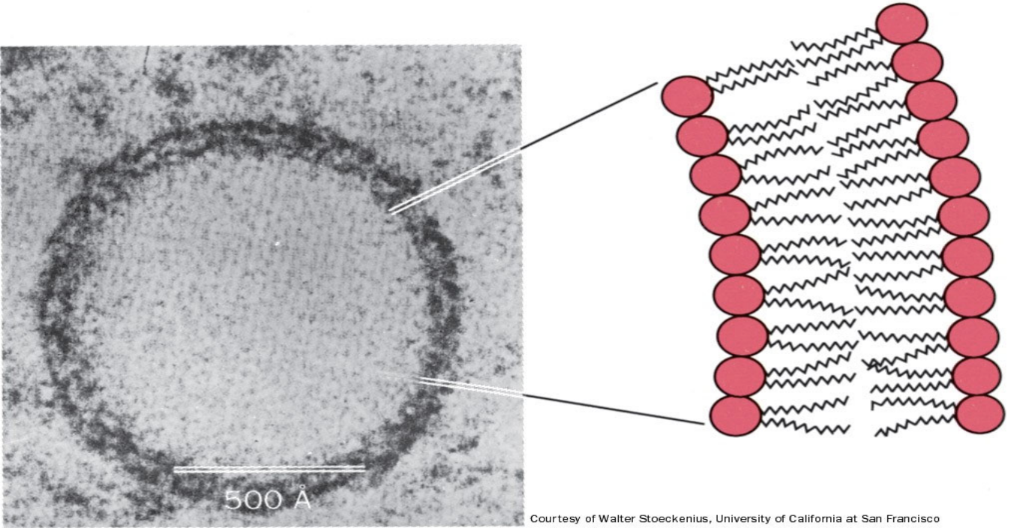
What does amphipathic mean?
Has both hydrophilic and hydrophobic regions
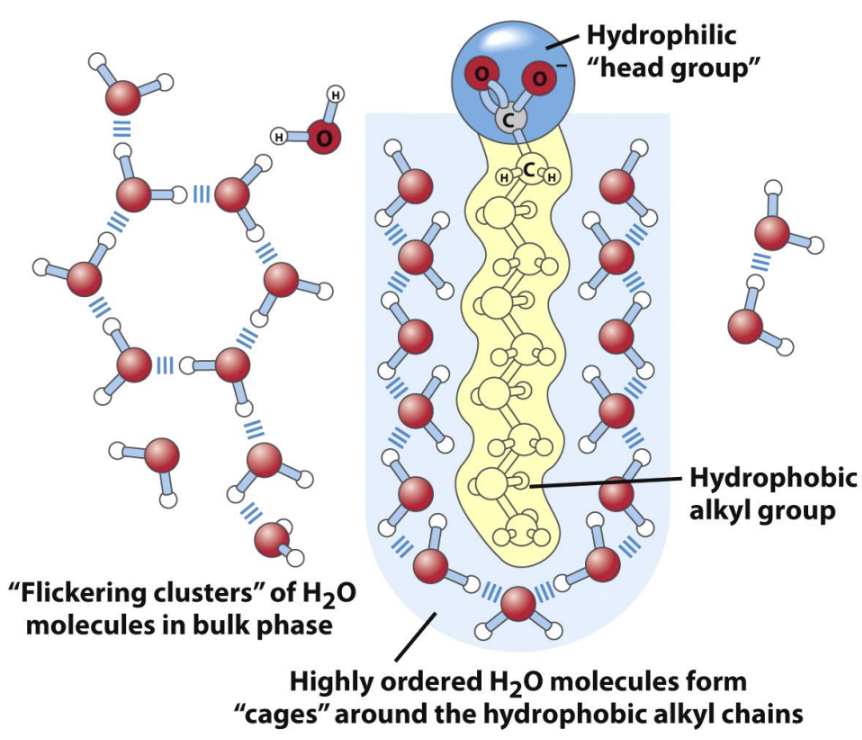
Why do lipid bilayers form in water?
Reduce ordered water shell → increase entropy
What is the hydrophobic effect?
Hydrophobic groups cluster to reduce water ordering
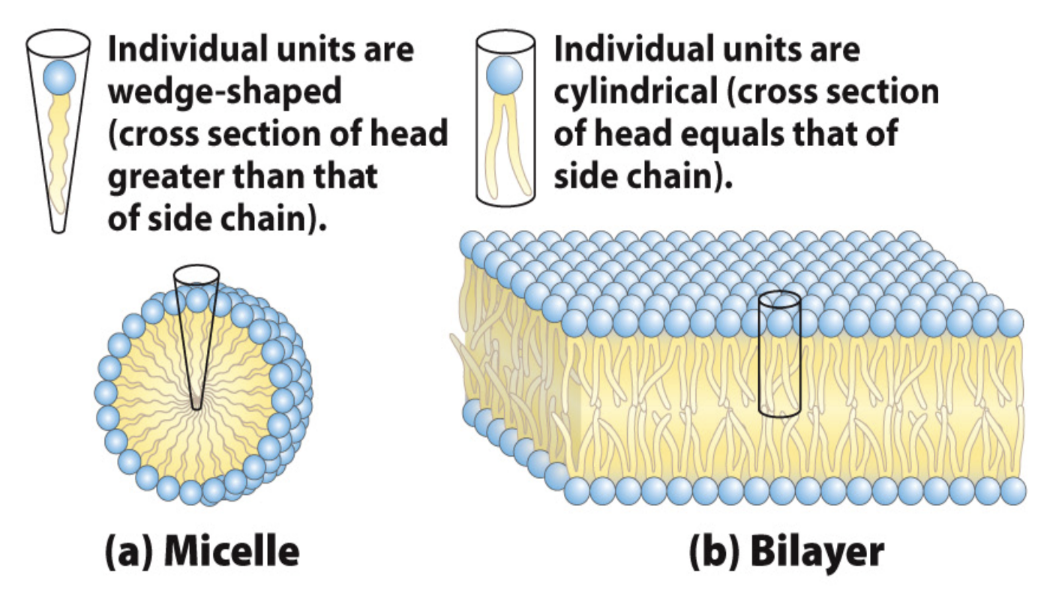
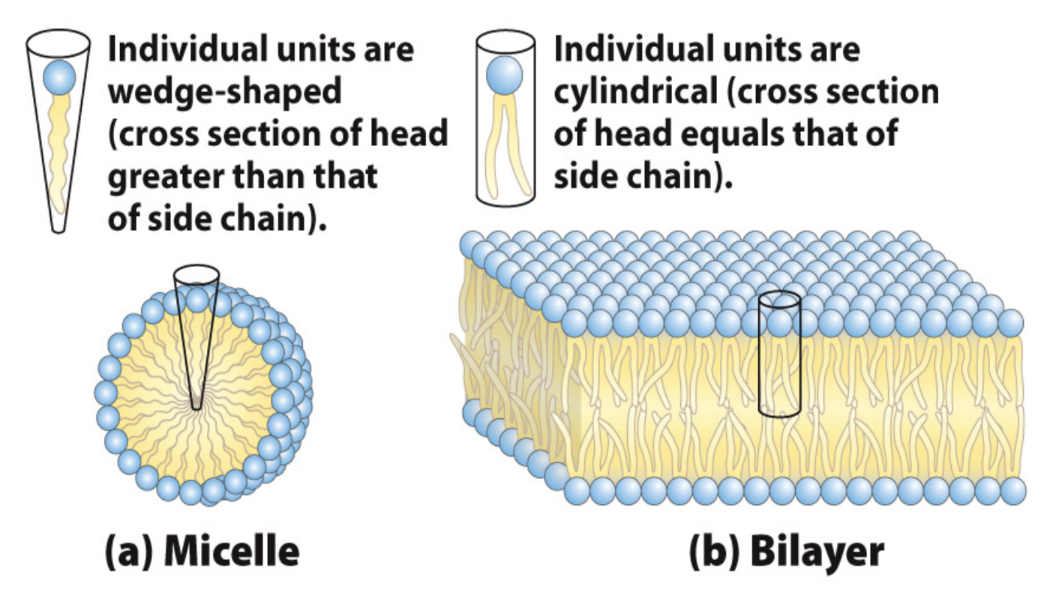
What determines micelle vs bilayer formation?
Wedge-shaped → micelles
Cylindrical → bilayers
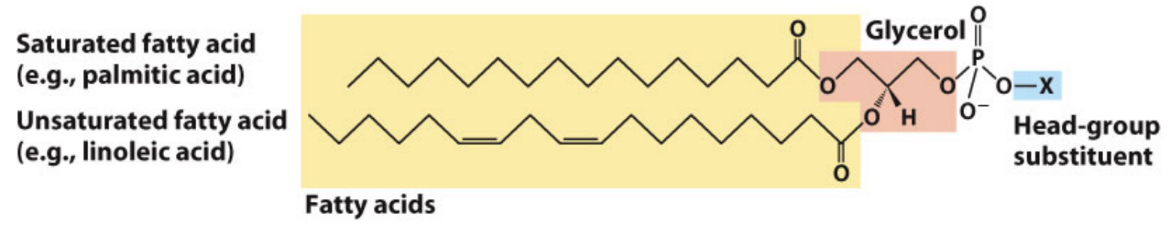
What are glycerophospholipids?
Diacylglycerol + phosphate + head group (phosphodiester bond)
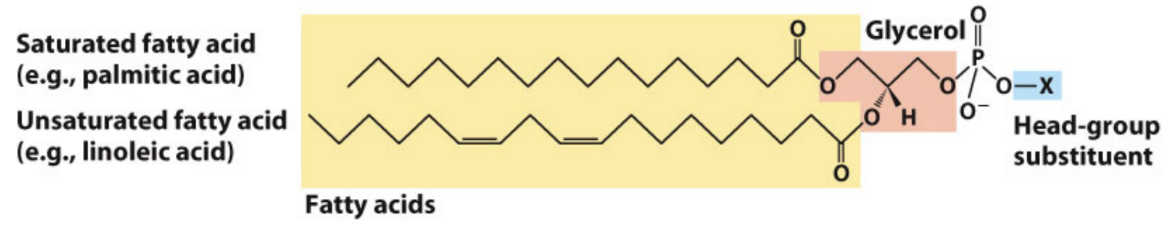
What is phosphatidic acid?
Parent compound of glycerophospholipids
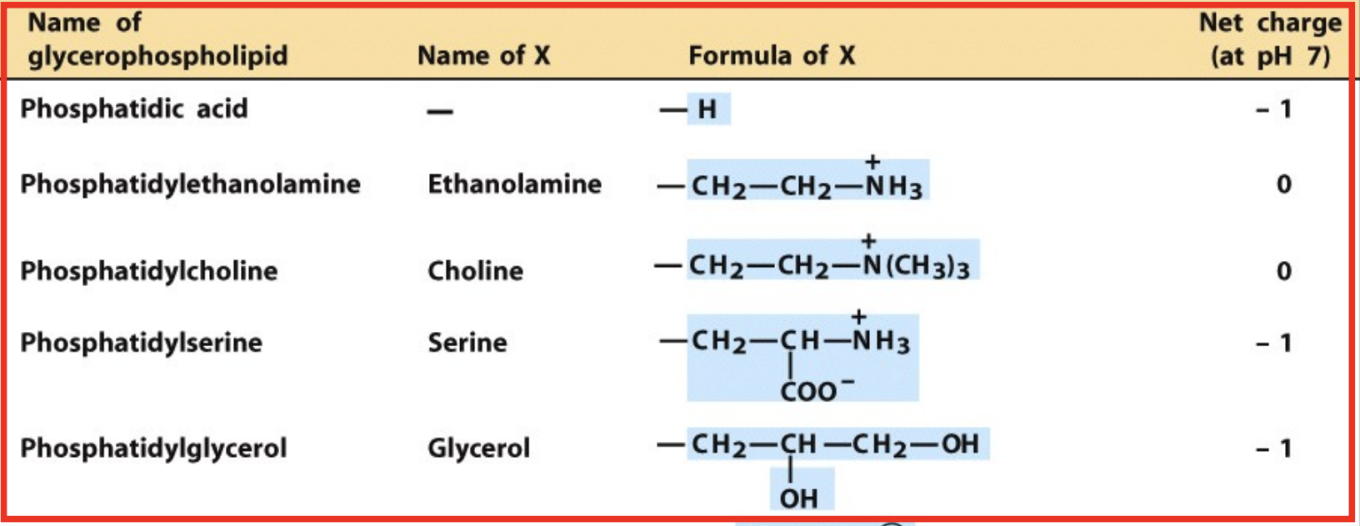
Which phospholipids are neutral at pH 7?
Phosphatidylethanolamine (PE)
Phosphatidylcholine (PC)
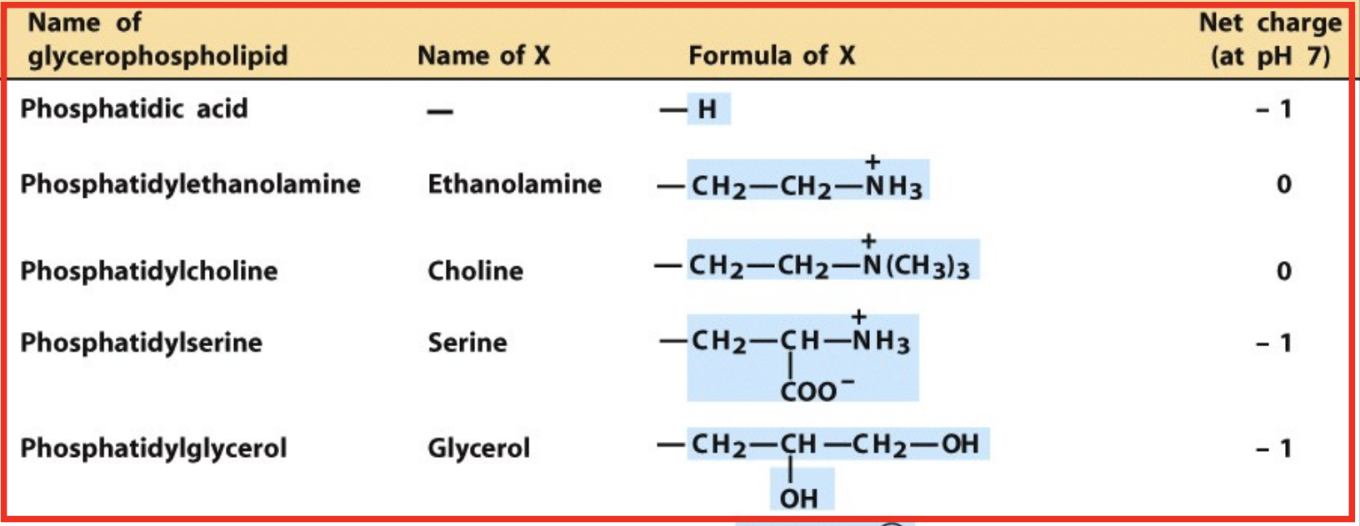
Which phospholipids are negatively charged at pH 7?
Phosphatidic acid (-1)
Phosphatidylserine (PS) (-1)
Phosphatidylglycerol (PG) (-1)
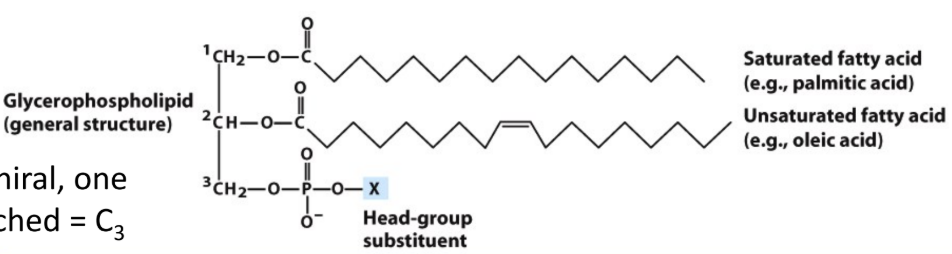
Why is glycerol prochiral?
Adding phosphate creates asymmetry
Two major lipid signaling types?
Slow acting, steroid hormones (activate nuclear receptors)
Fast acting eicosanoids (bind membrane receptors)
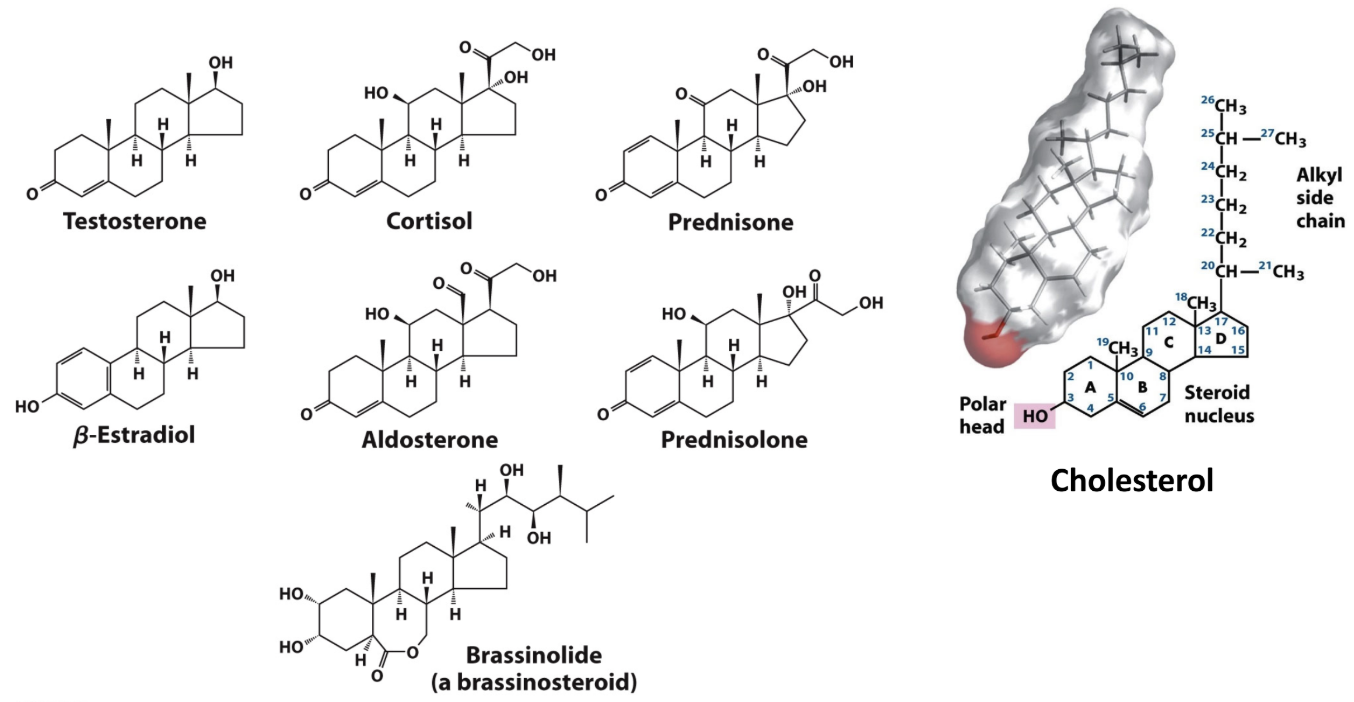
Role of cholesterol?
Membrane component + precursor of steroid hormones and bile acids
What is endocrine signaling?
Signal acts on other distant cells
What is autocrine signaling?
Signal acts on same cell
What is paracrine signaling?
Signal acts locally around secreting gland
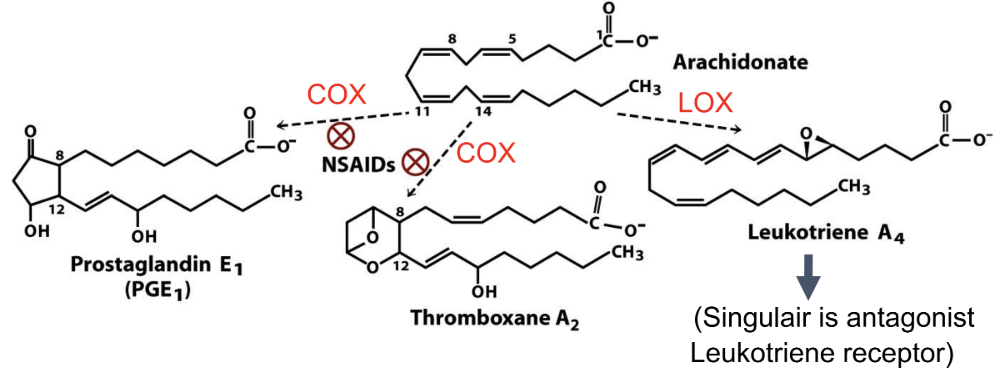
What comes from arachidonic acid?
Prostaglandins, thromboxanes, leukotrienes
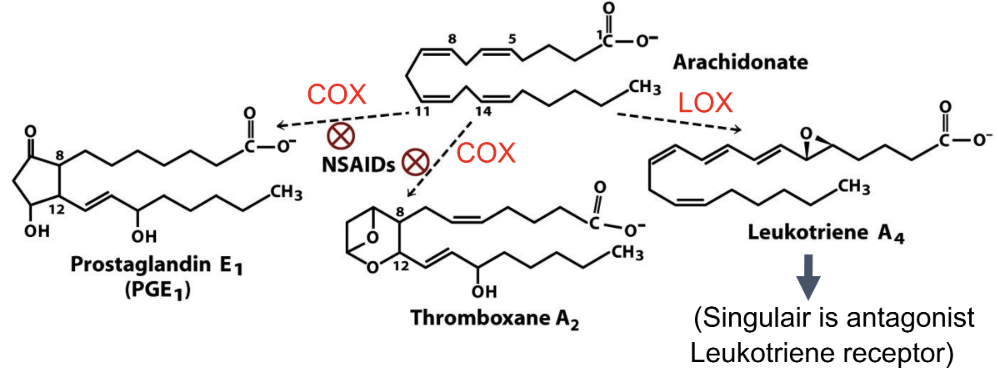
What converts arachidonic acid to prostaglandins?
COX (cyclooxygenase)
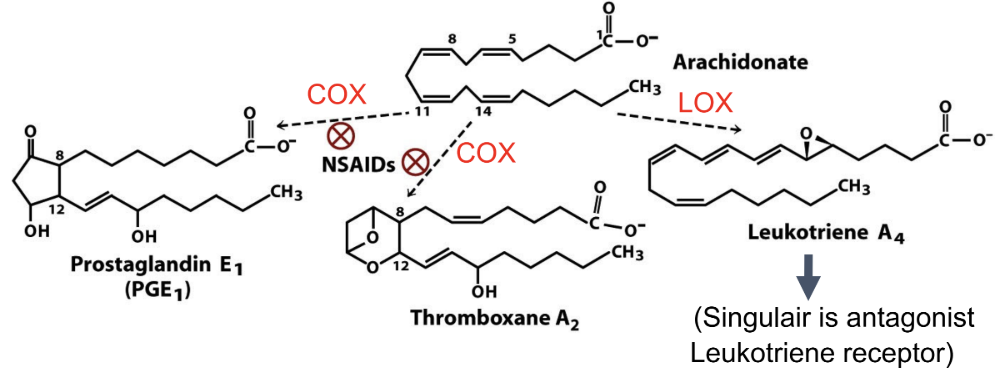
What pathway makes leukotrienes?
LOX (lipoxygenase)
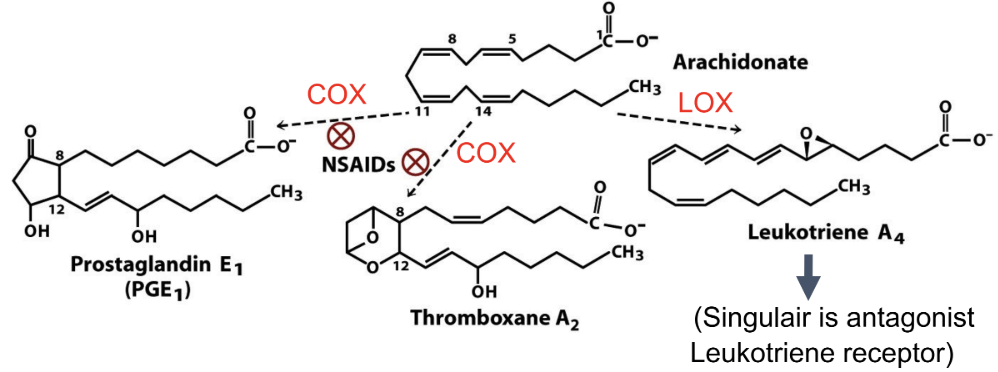
How do NSAIDs work?
Inhibit COX → block formation of prostaglandins/thromboxanes from arichidonate
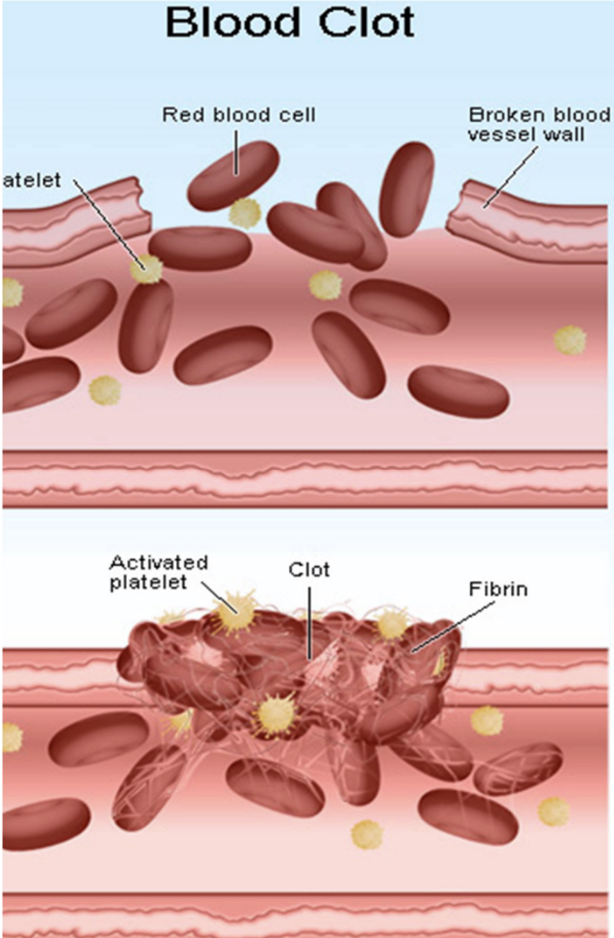
Why does blocking thromboxane reduce clotting?
Thromboxane promotes platelet aggregation
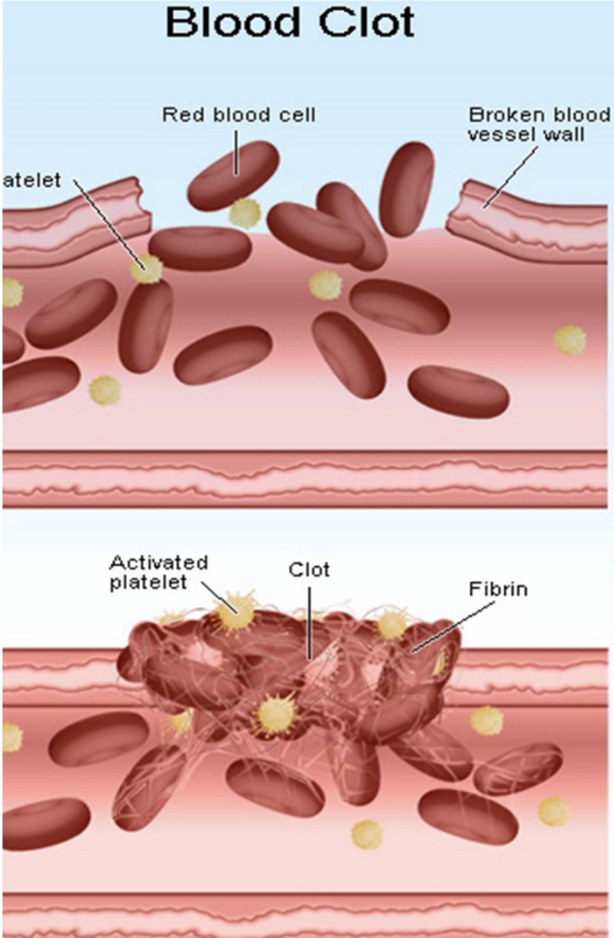
How does aspirin and ibuprofen affect specific inhibitors (COX1 and COX2)?
Aspirin and ibuprofen inhibit COX1 (causes bleeding) and COX2 (mediates pain and inflammation)
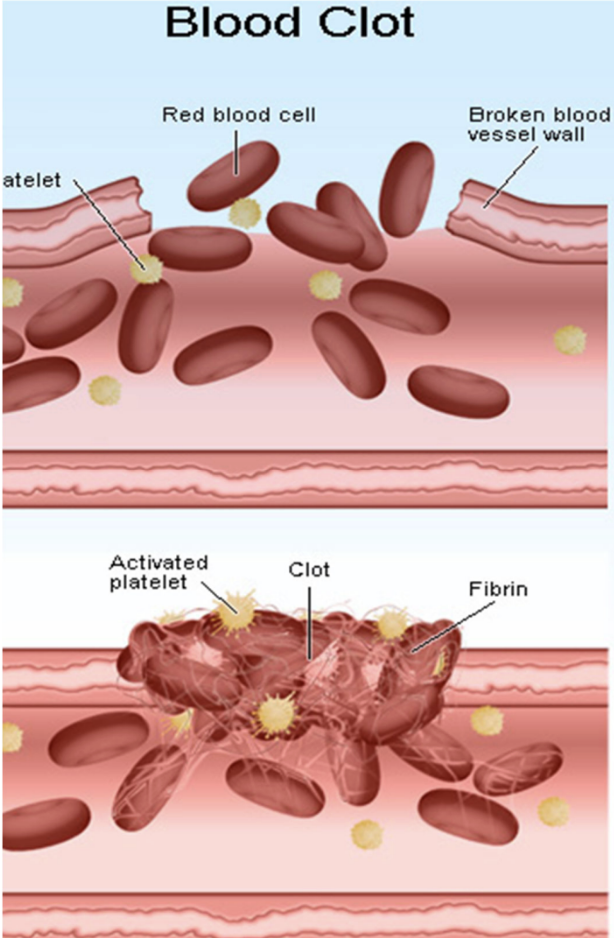
Why did COX-2 inhibitors increase CV risk?
Reduces body’s prostacyclin → relatively more clotting
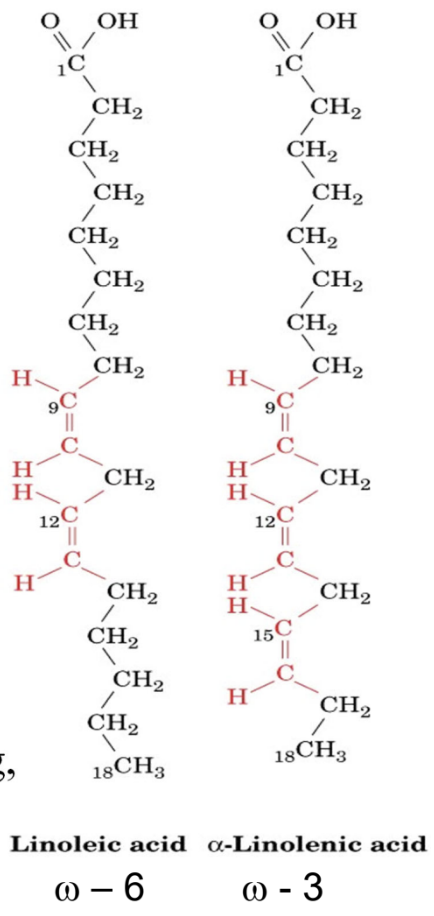
Essential fatty acids?
Alpha-linolenic (ω-3) and linoleic (ω-6)
Why are omega-3s anti-inflammatory?
Converted at a slower rate to arachidonic acid → less pro-inflammatory signaling
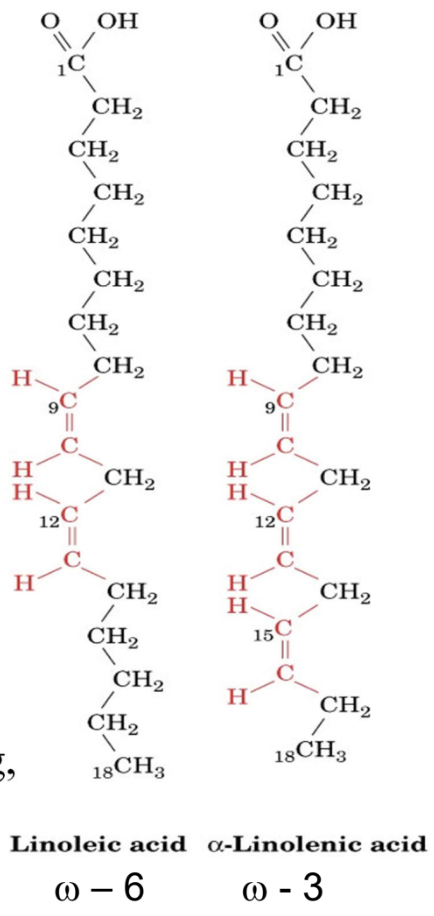
Key omega-6 inflammatory precursor?
Arachidonic acid
Cis vs trans effect on membrane fluidity?
Cis ↑ fluidity; trans ↓ fluidity (like saturated)
Why do trans fats behave like saturated fats?
Linear → tight packing
Effect of chain length on membrane fluidity?
Longer ↓ fluidity
Shorter ↑ fluidity
Effect of unsaturation on fluidity?
More double bonds ↑ fluidity
Role of cholesterol in membranes?
Buffers fluidity: ↓ at high temp, ↑ at low temp
Key C16–C20 fatty acids to know?
Palmitic (16:0)
Stearic (18:0)
Oleic (18:1)
Linoleic (18:2)
α-linolenic (18:3)
Arachidonic (20:4)
Function of phospholipase A2?
Releases arachidonic acid from phospholipids
Functions of eicosanoids?
Prostaglandins: inflammation
Thromboxanes: clotting
Leukotrienes: inflammation/asthma
What is the fluid mosaic model?
Fluid lipid bilayer with laterally mobile proteins; flip-flop restricted