OCHEM Fundamentals
1/43
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
wedge bonds
represent atoms coming out of the page
dashed bonds
represent atoms going into the page
constitutional/structural isomes
compounds with the same molecular formula but different connectivity of atoms
steric number
number of electron domains an atom has
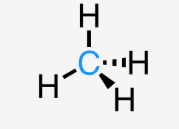
steric number 4
sp3
1 s orbital = 25% s character
3 p orbitals = 75% p character
steric number 3
sp2
1 s orbital = 33% s character
2 p orbitals = 66% p character
steric number 2
sp
1 s orbital = 50% s character
1 p orbital = 50% p character
tetrahedral
sp3
4 bonds, 0 lone pairs
109.5°
ex: methane
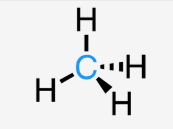
trigonal pyramidal
sp3
3 bonds, 1 lone pair
<109.5°
ex: ammonia
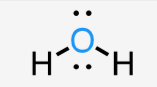
bent
sp3
2 bonds, 2 lone pairs
<109.5
ex: water
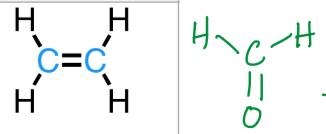
trigonal planar
sp2
3 bonds, 0 lone pairs
120°
ex: ethylene, formaldehyde
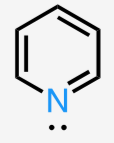
bent
sp2
2 bonds, 1 lone pair
<120°
ex: pyridine
linear
sp
2 bonds, 0 lone pairs
180°
ex: acetylene
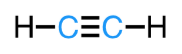
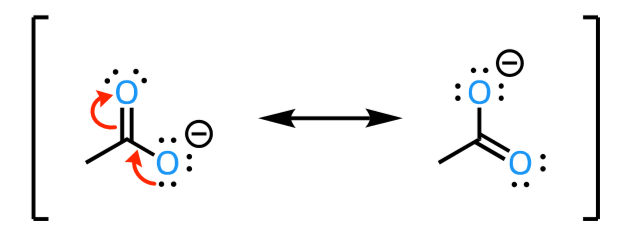
resonance rules
must maintain same skeleton (no movement of atoms)
be a valid Lewis structure
have same net charge
resonance hybrid
average of all of a molecule’s resonance structures and indicates areas of high and low electron density
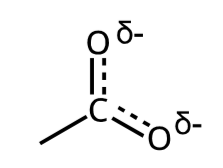
most significant resonce contributor (more stable)
maximize the # of complete octets
minimize the # of formal charges
negative charge on more electronegative atom
positive charge on less electronegratie atom
if an atom’s lone pair electrons are delocalized through resonance,
it does not contribute to the atom’s total # of electron domains
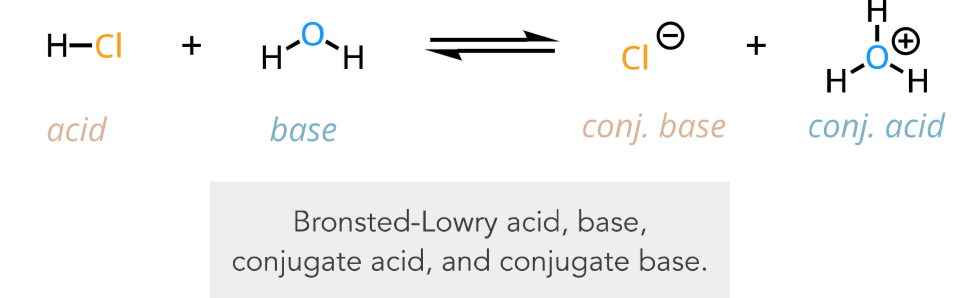
bronsted-lowry acid
proton donor
bronsted-lowry base
proton acceptor
bronsted-lowry acid-base reaction
a proton transfer occurs
products: conj base of acid and conj acid of base
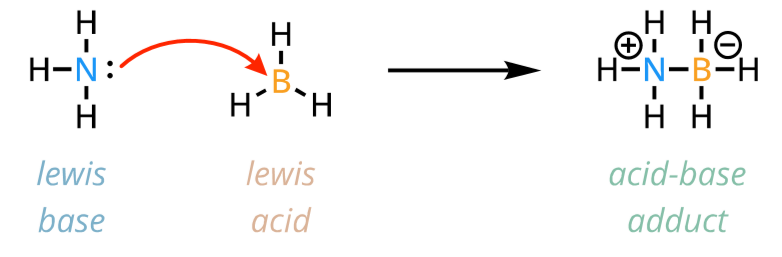
lewis acid
electron pair acceptor
lewis base
electron pair donor
lewis acid-base reaction
lewis acid-base adduct is formed
nomenclature prefixes
meth-
eth-
prop-
but-
pent-
hex-
hept-
oct-
non-
dec-
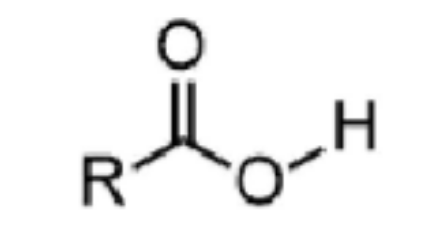
carboxylic acid - prefix & suffix
n/a
-oic acid
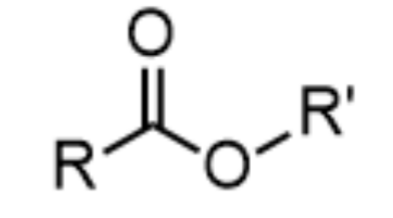
ester - prefix & suffix
alkoxycarbonyl-
-oate
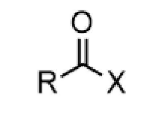
acid halide - prefix & suffix
halocarbonyl-
-oyl halide
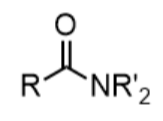
amide - prefix & suffix
amido-
-amide
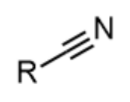
nitrile - prefix & suffix
cyano-
-nitrile
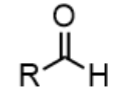
aldehyde - prefix & suffix
oxo-
-al
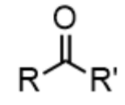
ketone - prefix & suffix
oxo-
-one
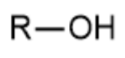
alcohol - prefix & suffix
hydroxy-
-ol
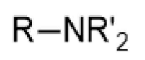
amine - prefix & suffix
amino-
-amine
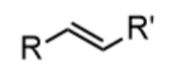
alkene - prefix & suffix
alkenyl-
-ene
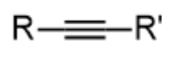
alkyne - prefix & suffix
alkynyl-
-yne
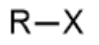
alkyl halide - prefix & suffix
halo-
n/a
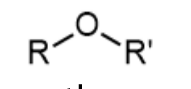
ether - prefix & suffix
alkoxy-
n/a
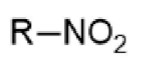
nitro - prefix & suffix
nitro-
n/a
highest priority → lowest priority
carboxylic acid
acid anhydride
ester
acid halide
amide
nitrile
aldehyde
ketone
alcohol
amine
alkene
alkyne
akyl, halogen, alkoxy, nitro
strong acids
low pKa, high Ka
7 strong acids include: Chloric acid (HClO3), Hydrobromic acid (HBr), Hydrochloric acid (HCl), Hydroiodic acid (HI), Nitric acid (HNO3), Perchloric acid (HClO4), and Sulfuric acid (H2SO4).
acidity increases as we go down a column in ptable because atomic size increases
larger atoms are more acidic since their conjugate base is more stable (weaker)
strong bases
high pKa, low Ka
ex: NaOH, KOH, Ca(OH)2, Ba(OH)2
acid base reactions favor formation of the weaker acid (higher pKa)
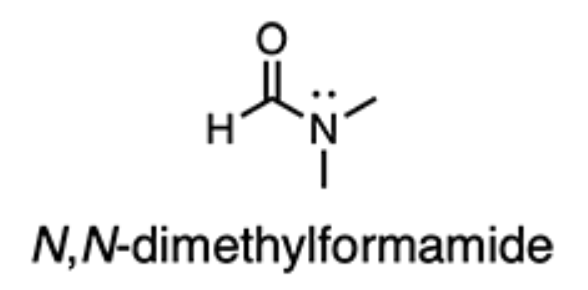
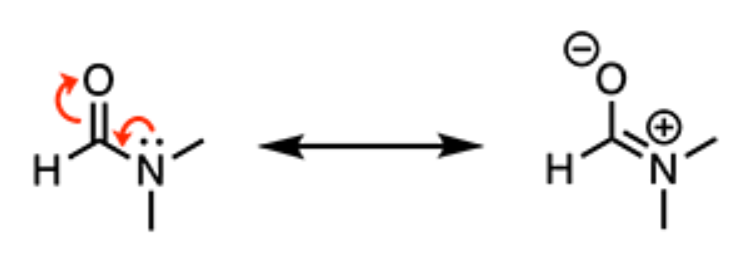
What is the hybridization state and approximate bond angle of the nitrogen atom of N,N-dimethylformamide?
sp2, 120°
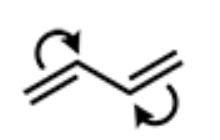
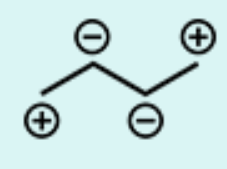
What is the product of the following arrow-pushing mechanism?
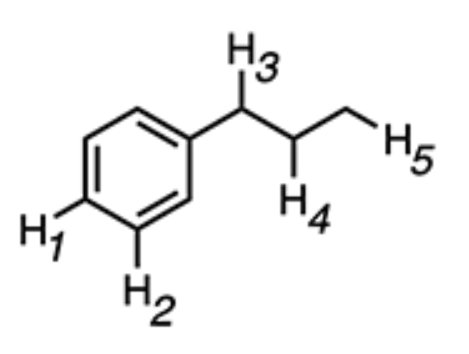
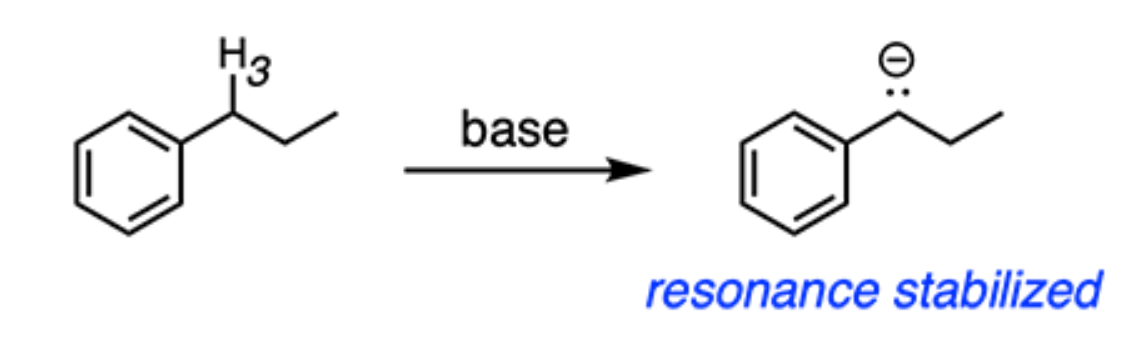
Identify the most acidic hydrogen in the compound below.
H3 → If H3 is removed, the conjugate base is stabilized by resonance delocalization from the benzene ring. This resonance stabilization makes the conjugate base more stable, and hence, H3 is the most acidic hydrogen.