topic 6a: introduction to organic chemistry
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
empirical formula
simplest ration of the number of atoms in a molecule
molecular formulae
the numbers of each sort of atom present in the molecule
general formula
the general rule for a particular homologous series
structural formula
shows how various atoms are bonded
displayed formula
shows all the bonds in the molecule as individual lines
homologous series rules
same general formula
similar chemical properties
successive members that differs by CH2
gradation of physical properties
made by similar methods
prefix 6-10
6: hex
7: hept
8: oct
9: non
10: dec
functional groups
anything that differs from a simple alkane
C=C
alkene
-X
halogenalkane
-OH
alcohol
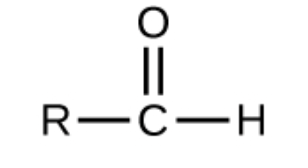
aldehyde
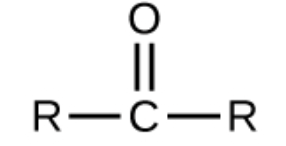
ketone