Enzymes
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
What is an enzyme?
Biological catalyst
Usually made of proteins
Why are enzymes important
Speed up reactions
By lowering activation energy (easier for reactions to start)
Breaks down food —→ usable nutrients
Turns food —→ energy for cells to use
Build and repair molecules
What do enzymes affect?
Rate of reaction —→ makes reactions happen faster
By lowering activation energy needed
What molecules do enzymes act on?
Substrates
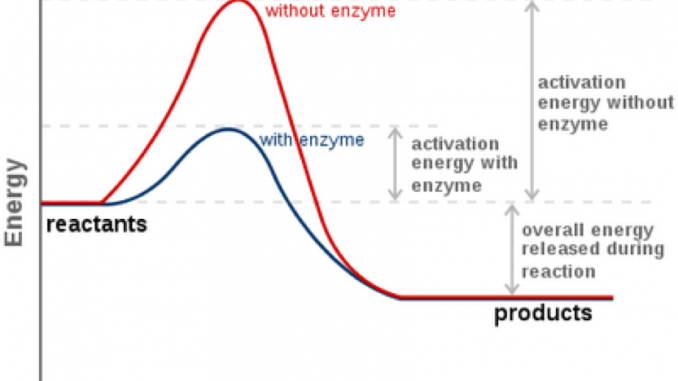
What is activation energy (AE)?
The minimum energy needed to start a chemical reaction
Do enzymes change ΔG?
No, they only lower activation energy (AE)
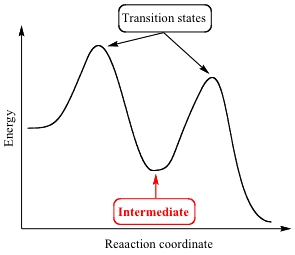
What is the transition state?
A high-energy unstable stage where:
Old bonds break
New bonds form
Enzymes help substrates reach this state more easily
What is enzyme specificity?
Means an enzyme usually works on…
one specific substrate
OR
One specific type of reaction
How are enzymes named/classified?
“-ase”
Ends in -ase suffix
What is an active site?
Region on the enzyme where the substrate binds —→ reaction happens
What is the “Lock and Key Model?”
Enzyme active site has a FIXED shape that matches the substrate
What is the “Induced Fit Model?”
Enzyme CHANGES shape to better fit the substrate
Which matters more—shape or chemistry at the active site?
Chemistry
The active site’s chemical interactions DRIVE the reaction
What happens in the active site?
Substrate binds active site (enzyme + substrate bond) —→ Substrate becomes unstable —→ activation energy decreases —→ product forms —→ product released
How do enzymes lower activation energy?
By…
Holding substrates in the right position
Strain the substrate bonds
Form temporary covalent bonds
Stabilizing transition state = less energy needed to begin for reaction
How can the reaction rate be measured
By measuring…
Decrease in substrate concentration
OR
Increase in product concentration (over time)
What is enzyme kinematics
Study of reaction rates and factors affecting them
What happens when substrate concentration increases?
Reaction rate INCREASES —→ enzymes get full —→ rate stops increasing
When all enzyme active sites become saturated/full
Saturated = All active sites are occupied by substrate
Effect of temperature on enzyme activity
LOW temp (cold) —→ Slow activity
Ideal temperature (depends on the organism) —→ Highest activity
HIGH temp (hot) —→ enzyme denatures —→ Activity drops
Effect of pH on enzyme activity
Each enzyme has an optimum pH depending on the organism
Too high/low pH
Changes charges on amino acids
Changes enzyme shape
REDUCES ACTIVITY
What are cofactors?
Non-protein helper molecules (NOT PROTEINS!!!)—- they’re extra molecules or ions
Enzymes need in order to work
Attaches to the enzyme to activate it —→ lets the reaction happen
Types of cofactors (3)
Activators
Coenzymes
Prosthetic groups
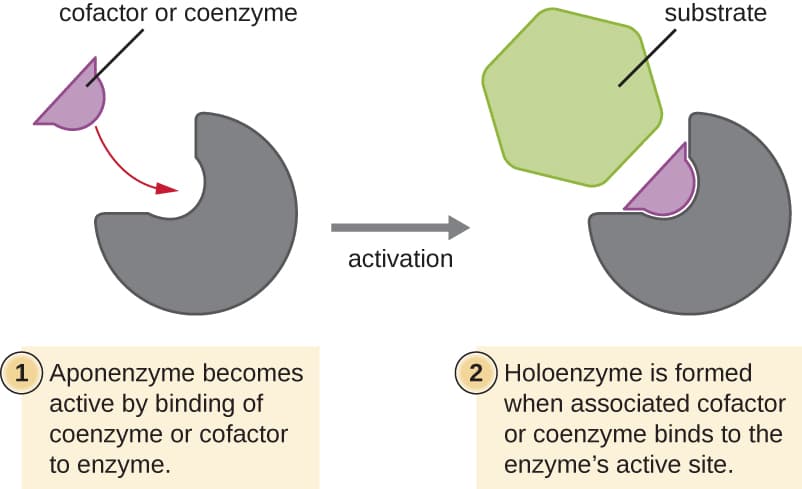
Coenzymes
Organic molecules that help carry electrons OR chemical groups during reactions
Prosthetic Groups
Organic molecules permanently attached to the enzyme
What are enzyme inhibitors?
A molecule that slows or stops an enzyme from working
By blocking or reducing enzyme activity
Why do enzymes control metabolism instead of just speeding reactions?
Enzymes control metabolism because…
They decide which reactions HAPPEN and WHEN
Regulating them lets the cell control its chemical reactions
Why do enzymes lower activation energy but not ΔG?
Enzymes lower the energy needed to start a reaction, but they don’t change the total energy released or required
ΔG is the energy difference between reactants and products
Enzymes don’t change the energy of reactants or products