Exothermic and endothermic reaction
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
What do exothermic reactions involve
Exothermic reactions involve a transfer of energy from the reactants to the surroundings
What is transferred and what will it show in an exothermic reaction
energy is transferred to the surroundings this will show an increase in temperature
What is an example of an exothermic reaction
Examples of exothermic reactions include combustion, freezing and condensing
What do endothermic reactions involve
Endothermic reactions involve a transfer of energy from the surroundings to the reactants
What is taken in in an endothermic reaction and what will it show
energy is taken into the reactants a decrease in temperature will be shown
What is an example of an endothermic reaction
Examples of an endothermic reactions include thermal decomposition, melting and boiling
What do energy level diagrams show
Energy level diagrams show the values of energy between the reactants and the products in a reaction
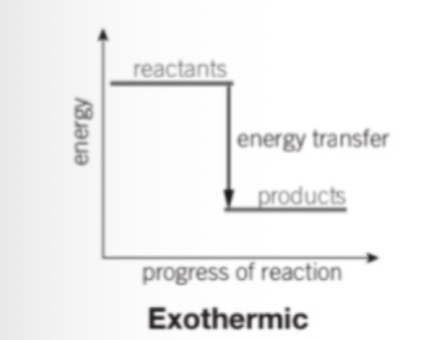
What does it mean if there is more energy in the reactants than the products
If the energy is greater in the reactants than the products then the reaction is exothermic and energy has been given out to the surroundings
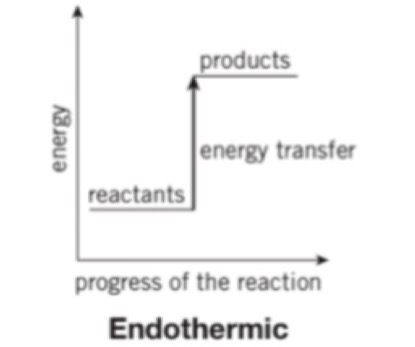
What happens if the energy is lower in the reactants than the products
If the energy is lower in the reactants then the reaction is endothermic as energy has been taken in from the surroundings,
An ice cube melts as energy is transferred from the drink it is in to an ice cube. Why is melting endothermic?
In ice, forces of attraction hold water molecules in a pattern. When energy is transferred to the ice, these forces are disrupted. The water molecules move out of their arrangement, and the ice melts
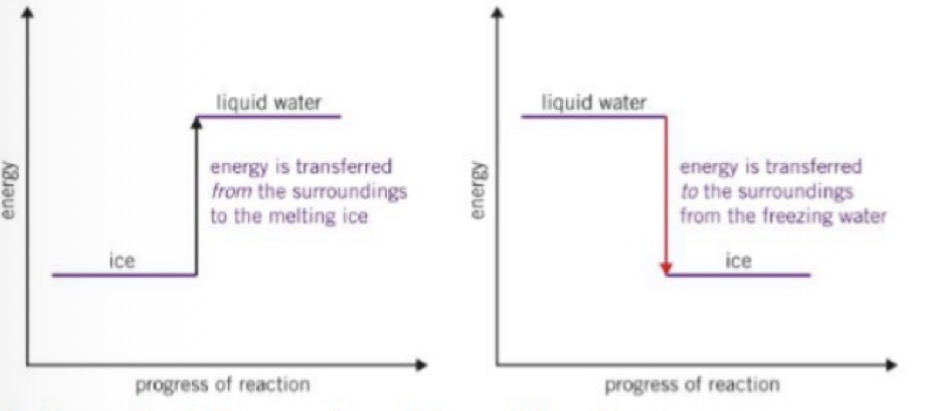
What happens on energy level diagrams for changes of state and what does each diagram show about change of state
Both diagrams show that liquid water stores more energy than the same amount of ice. The first diagram shows that ice takes energy from the surroundings as it melts. The second shows that water gives out energy to the surroundings as it freezes.
Is freezing exothermic
Freezing is exothermic