AICE Environmental Management Unit 7: Air Pollution
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Carbon Dioxide (CO2)
greenhouse gas; waste product of combustion & decompostion
acid deposition
as a mix of air pollutants
that deposit from the atmosphere as acidic
wet deposition (with a pH <5.6) or acidic dry
deposition
wet deposition
snow, rain, hail, fog
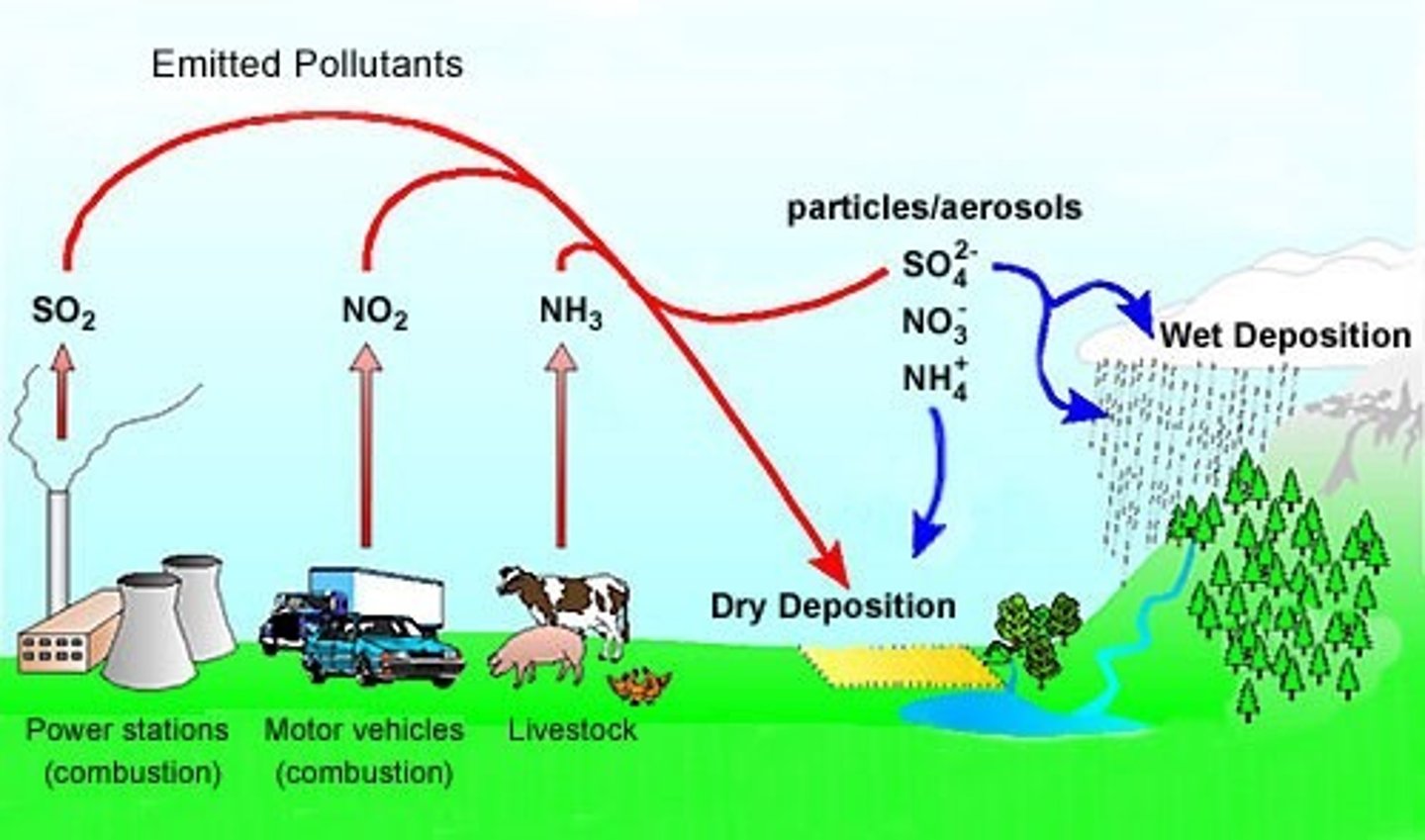
dry deposition
- dust and gases

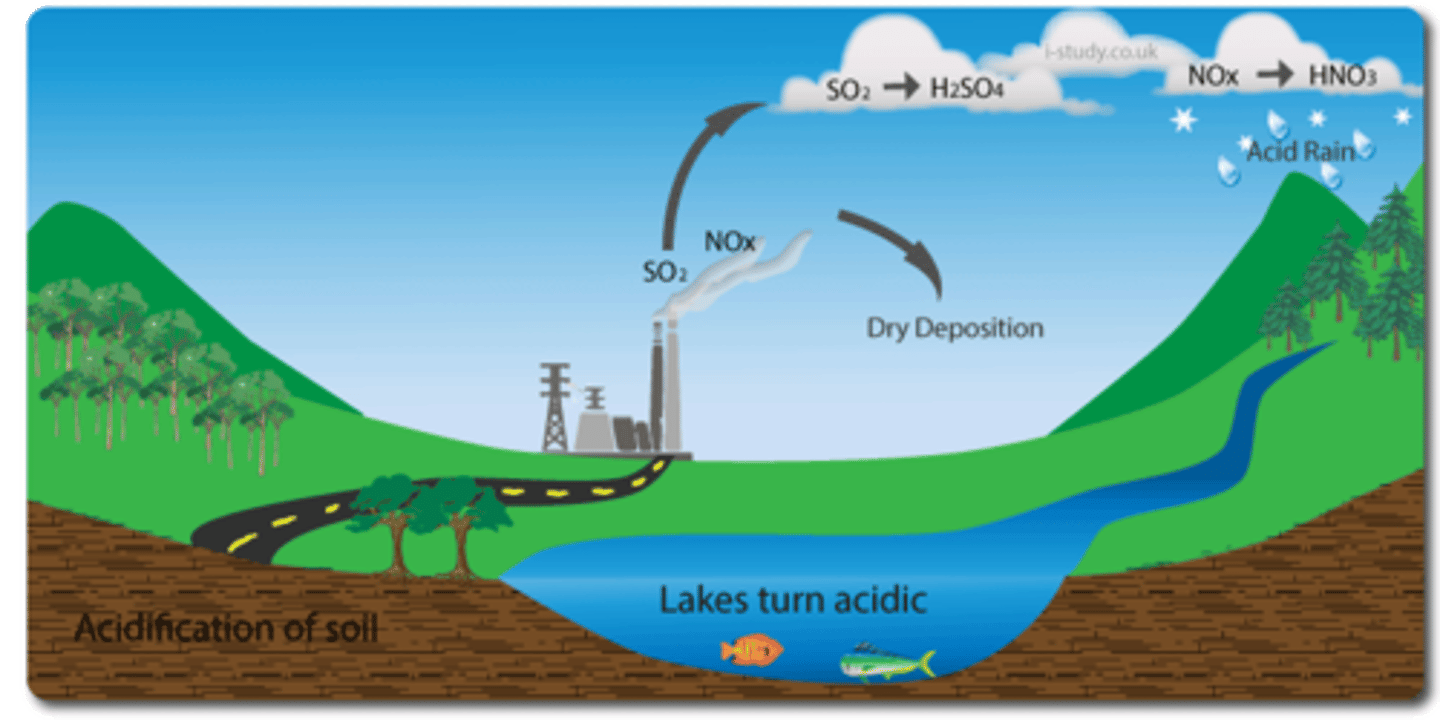
outline the formation of acid deposition
1. fossil fuels contain sulfur compounds
2. combustion of fossil fuels releases sulfur dioxide
gas
3. sulfur dioxide gas reacts with water and oxygen
in the atmosphere to form sulfuric acid
4. nitrogen from the atmosphere reacts with
oxygen in the high temperatures of vehicle
engines to form nitrogen monoxide gas
5. nitrogen monoxide gas is released into the
the atmosphere in vehicle emissions
6. nitrogen monoxide gas reacts with oxygen and
water in the atmosphere to form nitric acid
impacts of acid deposition on aquatic environments
affects fish gills and fish populations

Impacts of acid deposition on vegetation and crops
defoliation and reduced crop yield (think plant experiment)
impacts of acid rain on stone and brick buildings
enhanced chemical weathering

Photochemical smog
a mixture of air pollutants and particulates, including ground level ozone, which is formed when oxides of
nitrogen and volatile organic compounds (VOCs)
react in the presence of sunlight

ground level ozone
a secondary pollutant that forms when some of the pollutants released during fossil fuel combustion react with atmospheric oxygen in the presence of sunlight

impacts of photochemical smog
eye and respiratory irritation
• decreased crop yields
• deterioration of plastics and rubber

strategies for managing air pollution
• reduced use of fossil fuels
• reducing emissions of:
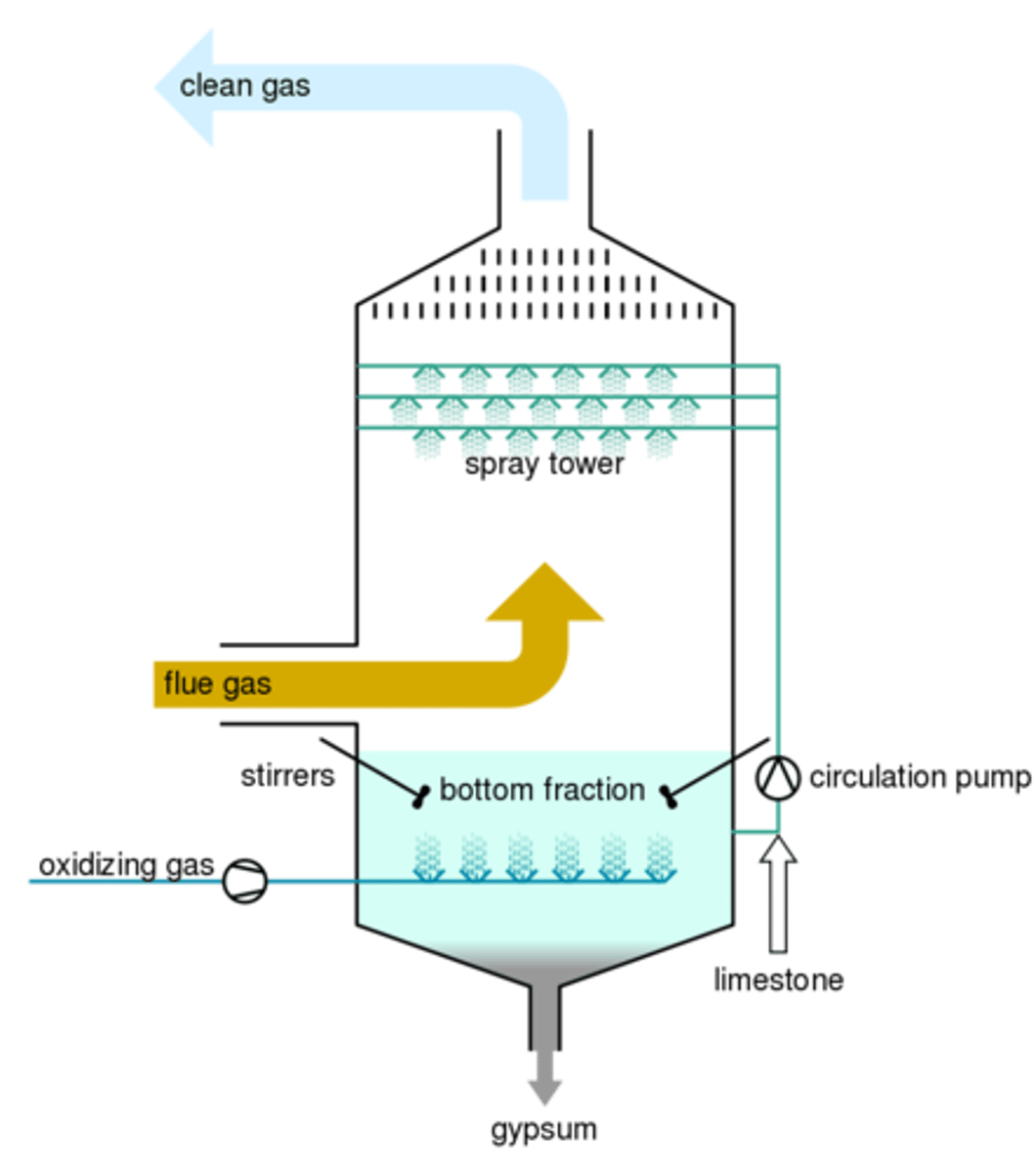
- sulfur dioxide by flue gas desulfurisation and
fuel desulfurisation
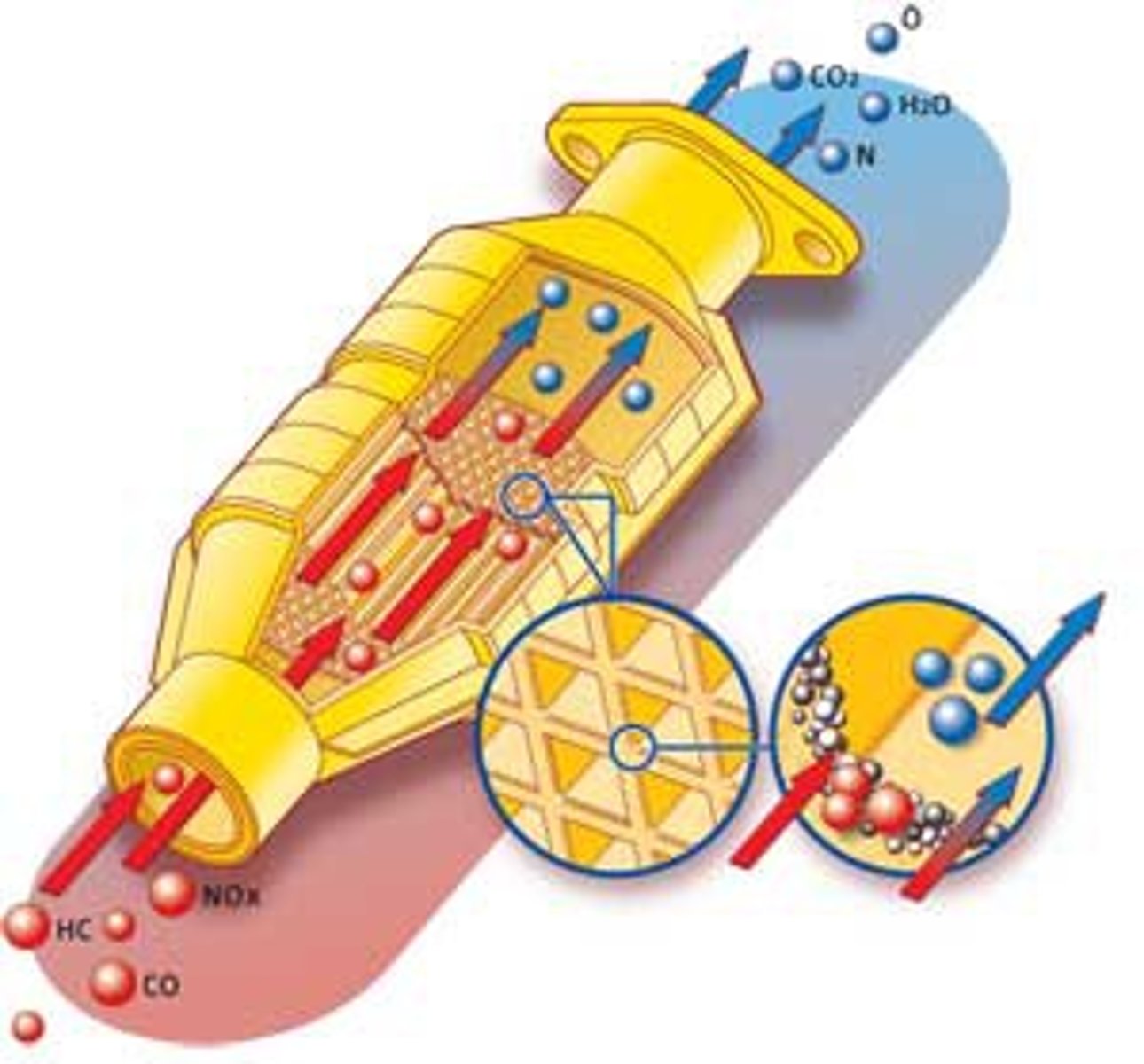
- oxides of nitrogen by catalytic converters
- particulates using electrostatic precipitators
- volatile organic compounds (VOCs)
○ safe usage, storage and disposal of
household products
• restricting vehicle use in urban areas
• legislation
- local, national and international legislation
(detailed knowledge of specific legislation
and agreements is not required)
- polluter pays principle
Gas desulfurization
set of technologies used to remove sulfur dioxide (SO2) from exhaust flue gases of fossil-fuel power plants, and from the emissions of other sulfur oxide emitting processes such as waste incineration.
catalytic converters
a device incorporated in the exhaust system of a motor vehicle, containing a catalyst for converting pollutant gases into less harmful ones.
electrostatic precipitators
A device used for removing particulates from smokestack emissions. The charged particles are attracted to an oppositely charged metal plate, where they are precipitated out of the air.
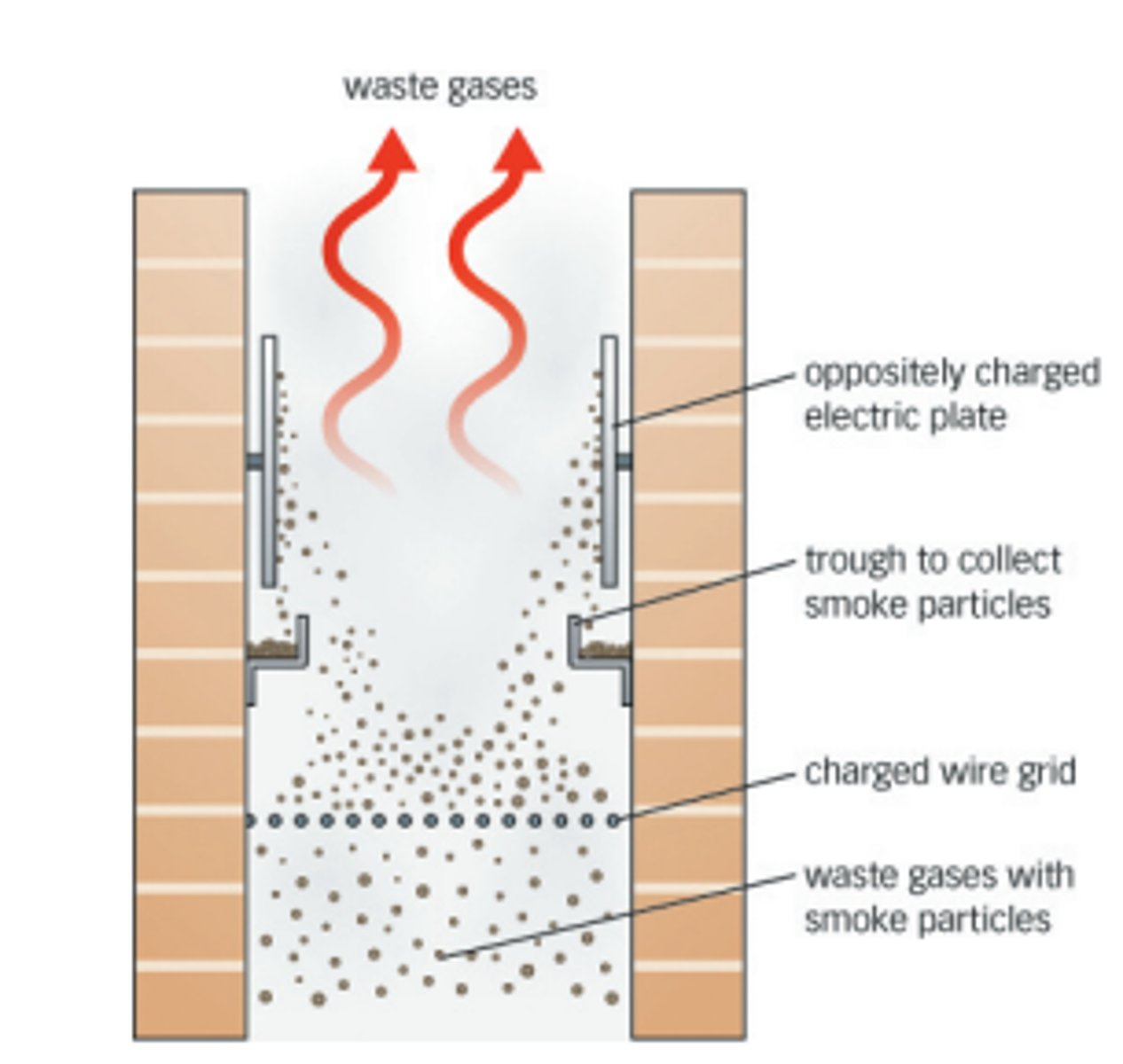
volatile organic compounds (VOCs)
Organic compounds that exist as gases in the atmosphere and act as pollutants, some of which are hazardous.

reducing VOCs
safe usage, storage and disposal of
household products
Montreal Protocol (1987)
A treaty put together to cut emissions of CFCs by 35% between 19889 and 2000.

outline how ozone depletion occurs
• chlorofluorocarbons (CFCs) from aerosols and
refrigerants are unreactive compounds and are
not broken down in the troposphere
• CFCs move into the stratosphere and break down
in the presence of ultraviolet light to release a
chlorine atom
• rapid reactions between chlorine atoms and
ozone breaks down ozone (O3) to oxygen (O2),
causing ozone depletion
• chlorine atoms remain in the stratosphere and
can continue to destroy ozone
detailed chemical mechanisms are not required
the Dobson Unit
state that ozone concentration is measured
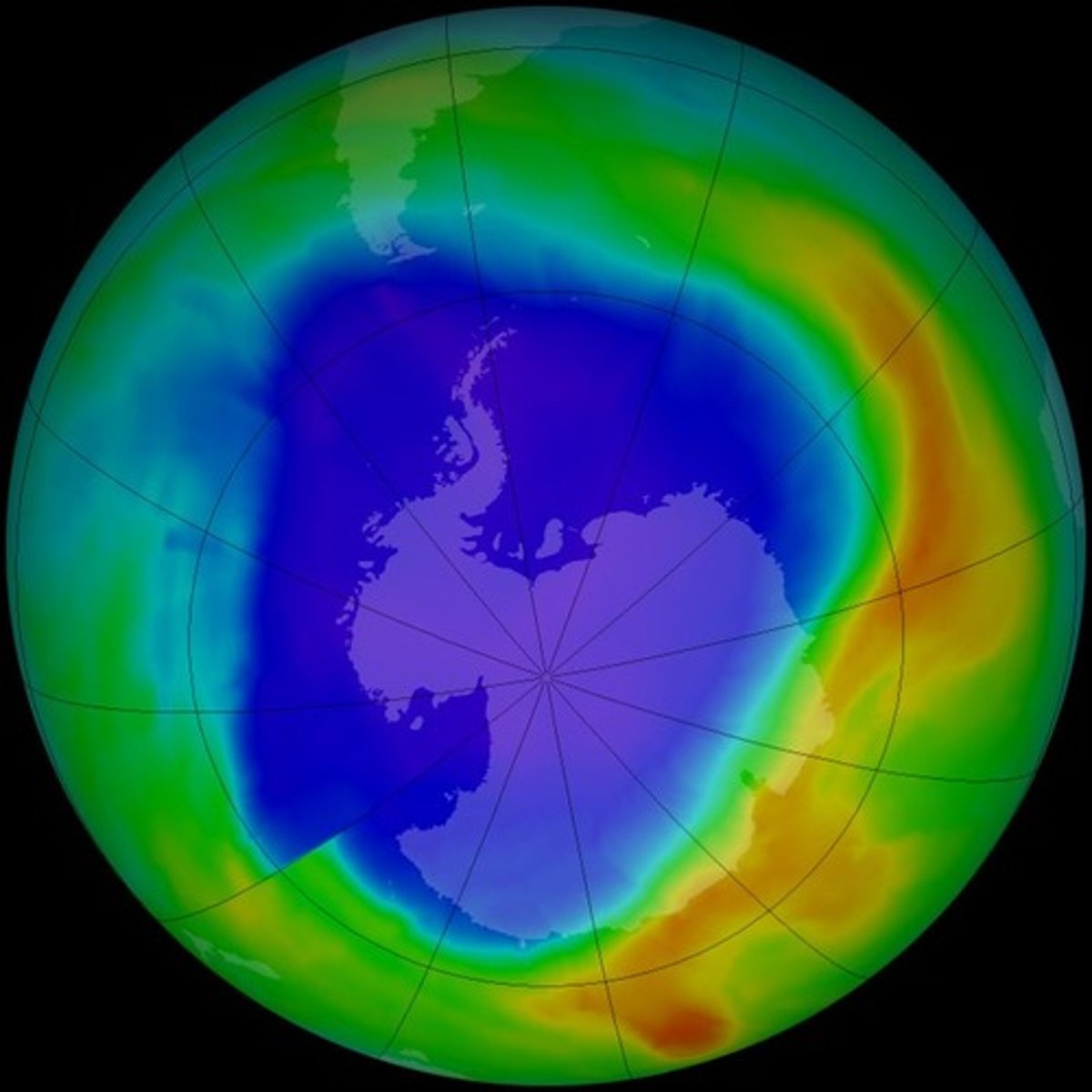
ozone hole
the term as an area where the
average concentration of ozone is below 100
Dobson Units ---NOT CLIMATE CHANGE
explain why ozone depletion has been greatest
over Antarctica
temperature, polar vortex, polar
stratospheric clouds (PSCs)
polar vortex
A huge swirling mass of very cold air that is isolated from the rest of the atmosphere until the sun returns a few months later.
stratospheric clouds (PSCs)
clouds in the winter polar stratosphere at altitudes of 15,000-25,000 m (49,000-82,000 ft).
Why is the ozone hole over Antarctica?
damage occurs due to the presence of chlorine and bromine from ozone-depleting substances in the stratosphere and the specific meteorological conditions over the Antarctic.
impacts of ozone depletion due to
the increased amounts of ultraviolet radiation
• human health (cataracts, skin cancer)
• decreased crop yields
• biodiversity of terrestrial and aquatic ecosystems
• degradation of materials used in clothing and
construction
Was the Montreal Protocol successful?
The Montreal Protocol has been successful in slowing and reversing the increase of ozone-depleting gases (halogen source gases) in the atmosphere. An important measure of its success is the change in the value of effective stratospheric chlorine.
alternatives to ozone depleting substances
• hydrochlorofluorocarbons (HCFCs)
• fluorinated gases (F-gases)
Hydrochlorofluorocarbons (HCFCs)
Group of chemicals containing hydrogen, chlorine, fluorine, and carbon, produced as a potential substitute for CFCs
Fluorinated gases (F-gases)
greenhouse gas emitted during industrial process including refrigeration
Fluorinated gases (F-gases)
man-made gases that can stay in the atmosphere for centuries and contribute to a global greenhouse effect.
Rowland-Molina hypothesis
this hypothesis proposes that long living halogen compounds can cause ozone depletion
• initially the main hypothesis was not accepted
• some of the auxiliary hypotheses were not
backed up by experimental evidence
• the hypothesis led to further research and data
collection by other scientists, which confirmed
that CFCs are ozone depleting
this shows the importance of experimental evidence
to support a hypothesis
Great Smog London (1952)
A smog that was so dense for 4 days, the people in London could not see what was in front of them; 4000 people died.
Donora Smog of 1948
In late October of 1948, the Donora Smog rocked the small Pennsylvanian community. Instead of another ‘average’, hazy day that came and passed, large clouds of polluted air from the nearby zinc plant were trapped by a weather inversion from readily moving along. Of Donora’s 14,000 total residents, 6,000 were afflicted by toxic air – 26 lost their lives. To this day, the Donora Smog crisis is considered the worst air pollution incident to occur in the United States and one of the worst in the world.
Management of the Great Smog of London
Slow to act at first, the British government ultimately passed the Clean Air Act four years later, in 1956, as a direct response to the lethal fog. The act established smoke-free areas throughout the city and restricted the burning of coal in domestic fires as well as in industrial furnaces.
Management of Donora Smog Event
Following the deadly smog, President Truman convened the first national air pollution conference in 1950. Congress didn't pass its first Clean Air Act until 1963, but progress continued steadily after that, with President Nixon creating the Environmental Protection Agency in 1970, the same year that Congress passed a more comprehensive Clean Air Act. But the work of protecting the environment is never entirely finished, as new industries and technologies take the place of previous ones.
Sulfur Dioxide (SO2)
Air pollution Formed when fossil fuels, which contain sulfur, are burned
Has the ability to dissolve in water vapor to form sulfuric acid and interact with the particulate matter and gases in the air to form other products
Primary Pollution
carbon monoxide
a colorless, odorless toxic flammable gas formed by incomplete combustion of carbon.
Paris Agreement (2015)
Agreement made between the United Nations countries to cut down on the emissions of greenhouse gases in an attempt to slow down global warming. More wealthy and producing countries have to cut down more
Particulate Matter (PM)
Solid or liquid particles suspended in air; also known as particles and particulates.
Lead pollution
At the national level, major sources of lead in the air are ore and metals processing and piston-engine aircraft operating on leaded aviation fuel. Other sources are waste incinerators, utilities, and lead-acid battery manufacturers. The highest air concentrations of lead are usually found near lead smelters
Clean Air Act of 1970
Required EPA to develop and enforce regulations to protect the public from airborne contaminants; forced states to follow and make sure the laws for followed in relation to the EPA. States allowed to decide officials for enforcement.