Homogenous catalysts
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
What are homogenous catalysts?
a catalyst in the same phase as the reactants.
Usually aqueous/gaseous reactions.
These are much less common in industry, due to the difficulty in separatign products and catalysts
They form intermediates durign a reaction
reaction between iodide ions, I- (aq) with peroxodisulfate ions, S2O8 2-
The iodide ions are oxidised to form I2
S2O82- + 2I- → I2 + 2SO42-
the reaction is very slow. Both reactants are negatively charged so repel each other (high Ea)
Fe 2+ is added as a catalsyt ot speed up rate of reaction
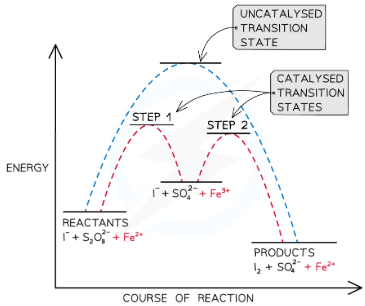
mechanism for reaction of iodide ions with S2O82-
1) S2O8 2- (aq) + 2Fe2+ (aq)—>2SO4 2- (aaq) + 2Fe3+ (aq)
The reactants are oppositely charged, so attract, rather than repel.
Fe2+ is oxidised to form Fe3+
2) 2I- (aq) + 2Fe3+ —>I2(aq) + 2Fe2+ (aq)
Fe 3+ oxidises the I- to form iodine
Fe2+ has been reduced back to Fe3+ - the catalyst is regenerated
As all of the reactants and products and the catalyst are in the aqueous phase this reaction is an example of homogeneous catalysis
how can Fe3+ be used as a catalyst
1) 2I- + 2Fe3+ → I2 + 2Fe2+
2) S2O82- + 2Fe2+ → 2SO42- + 2Fe3+
reverse order of equations
manganate (MnO4-) reaction with ethanedioate ions, C2O42-
MnO4- ions oxidise the ethanedioate ions. This forms carbon dioxide and water, in acidic conditions
2MnO4-(aq) + 16H+ (aq) + 5C2O4 2- 9aq)—> 2Mn2+ (aq) + 8H2O (l) + 10 CO2 (g)
The reaction is used in a titration to confirm the cocnentraiton of KMnO4 - potassium manganate Vll
Activation energy of reaction btw MnO4- and C2O4 2-
The reactants both have negative charges, so they repel eachother. Ea is high and rate of reaction is slow.
The catalyst for this reaction is Mn2+, one of the products
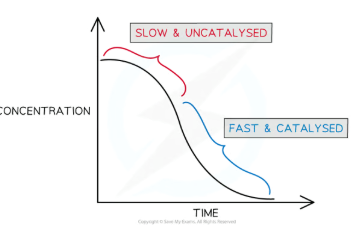
As the titration proceeds, teh rate of reaction increases
reaction mechanism for reaction between manganate(VII) ions and oxalate (ethandioate) ions
As more manganese(II) is formed the reaction speeds up
Like to the role of iron(II) in the previous section, manganese(II) ions take part in a redox cycle between two different oxidation states (+2 → +3 → +2)
4Mn2+ (aq) + MnO4– (aq) + 8H+ (aq) → 5Mn3+ (aq) + 4H2O (aq)
2Mn3+ (aq) + C2O42- (aq) → 2CO2 (g) + 2Mn2+ (aq)
The manganese(II) is not present in the beginning of the reaction, but as it is formed is speeds up the reaction and is re-generated during the redox cycle
This reaction is easily followed on a colorimeter as the rate at which the purple manganate(VII) ion is consumed accelerates with time
autocatalysis meaning
the product of the reaction acts as a catalyst for the reaction