Lab final CHEM 2211
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
what are the 5 steps of recrystallization
Choose an appropriate solvent 2. dissolve the impure mixture in the hot solvent 3. remove the insoluble impurities from the hot solvent 4. cool filtered solution, crystallize the solid 5. isolate the pure compound
is you aren’t seeing crystals while crystalizing your solid, what can you do?
add a seed crystal 2. boil off some solvent and re-cool 3. scratch the inner wall of the flask with stirring rod
what is the technique isolation? (recrystillization)
the crystals were purified and isolated via the five steps of recrystallization
what is the eutectic point?
the melting point of the mixture of compounds A and B; the melting point will be broadened and depressed
what is distillation?
liquid vaporizations followed by condensations. the purity of the distilled liquid depends on the conditions of the experiment
what is simple distillation?
used to separate miscible liquids that have a boiling point difference of 100 degrees or greater
what is fractional distillation?
used to separate liquids that have a boiling point difference less than 100 degrees, also utlilizes a series of concurrent simple distillations within the fractional column
describe the set up of the simple and fractional distilation.
dont forget the boiling chips put in the solution to prevent bumping
what is an azeotrope
when two miscible liquids behave as a pure liquid (the mixture has one boiling point).
what is Raoult’s law?
Ptotal= (Xa●Pa)+(Xb●Pb)…
X stands for the mole fraction (you add the ratio together then divide by each individual to get the mole fraction)
what does increasing altitude do to pressure?
decrease pressure (lower boiling point)
solid-liquid extraction
component separation based on a compound’s solubility in a particular solvent system
liquid-liquid extraction
component separation based on a compound’s varying solubility between two immiscible liquid phases
how is TLC utilized in the lab?
compound purity determination, compound/multi-component mixture identification, monitoring the process of a chemical reaction
what is the stationary phase made of for TLC?
silica gel (polar)
what is the mobile phase made of?
ethyl acetate containing 0.5% acetic acid (nonpolar)
what is the retention factor equation?
Rf= Distance from origin to sample/ Distance from origin to solvent front
what does it mean if a compound has a low Rf (polar or non polar)?
Low Rf = High polarity (wants to stay near bottom of silica plate where more polar)
how does solubility change in the Separation of 3 component mixture lab?
solubility changes as compounds become protonated/deprotonated
what is the basic component in the Separation of 3 Component mixture? Molecule?
benzocaine
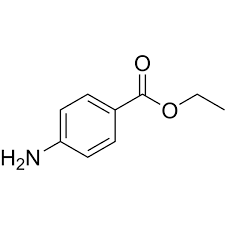
what is the neutral component in the Separation of 3 Component mixture? Molecule?
Diphenylmethanol

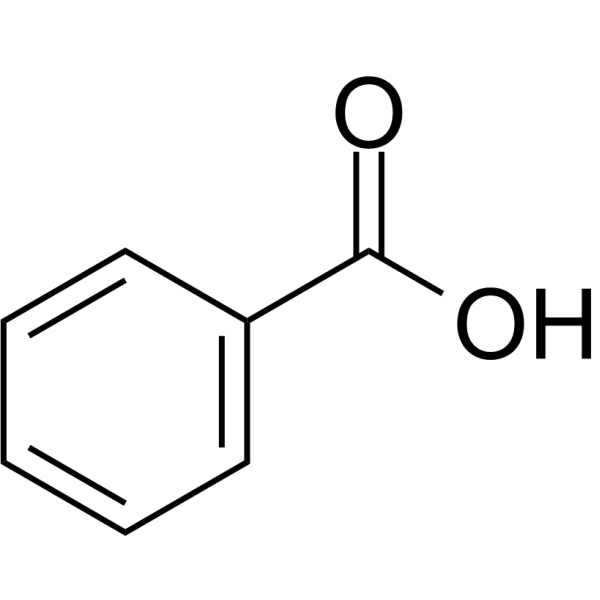
what is the acidic component in the Separation of 3 Component mixture? Molecule?
Benzoic acid
what is IR and H-NMR used for in the separation lab?
to confirm identity of basic and neutral components, used to identify acidic component
what is added first to the solution to separate the solution into organic and aqueous? which components are aqueous? which are organic?
2M HCl
Aqueous- benzocaine (gain H)
Organic- Benzoic acid, diphenylmethanol
what is added to the aqueous layer after the first step?
6M NaOH
turns the benzocaine insoluble and neutral
what is added to the organic layer after the first step?
1 NaOH
Aqueous- benzoic acid (removed H)
what is added in the third step to the organic layer to further isolate the solution?
6M HCl
Aqueous- benzoic acid, insoluble and neutral
organic- diphenylmethanol
what is the reagent used in the Transfer Hydrogenation of Olive Oil lab?
cyclohexene (H donor)
what is the formula (drawn out) of the triolein into tristearin reaction.
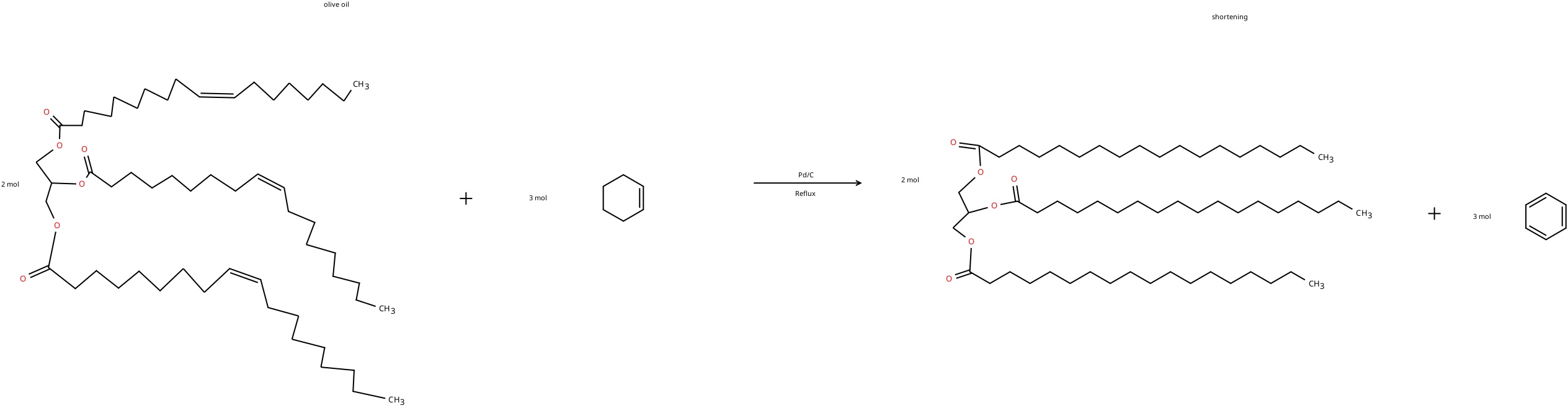
know the balanced chemical reaction from the post lab questions
yurt
what is the reagent used in the Preparation of Diphenyl Acetylene lab?
Pyridinium Hydrobromide Perbromide (PHPB)
what is the reaction (drawn out) of Trans-stilbene into Diphenylacetylene?
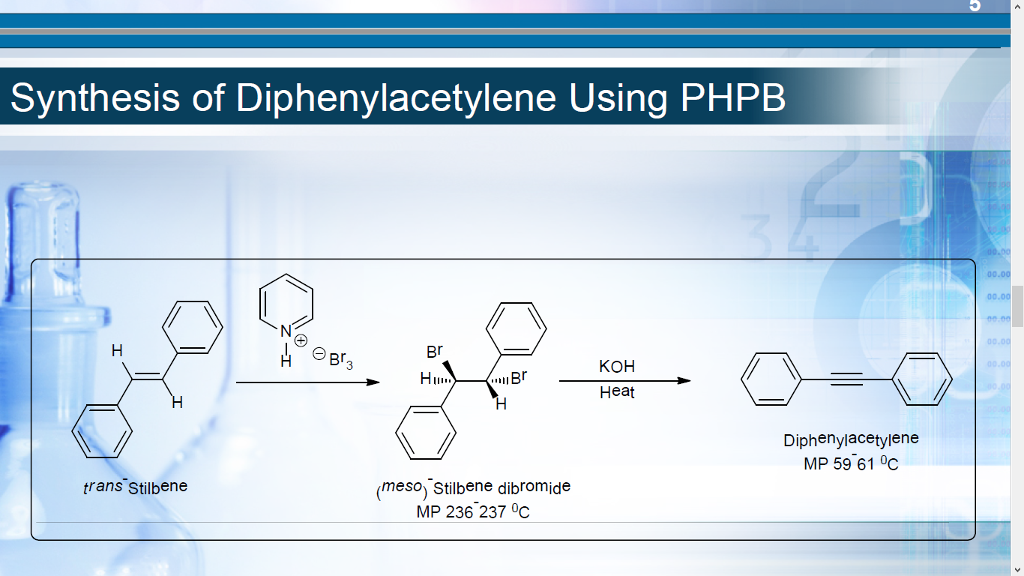
what is the mechanism for the Tran stilbene into diphenylacetylene reaction?
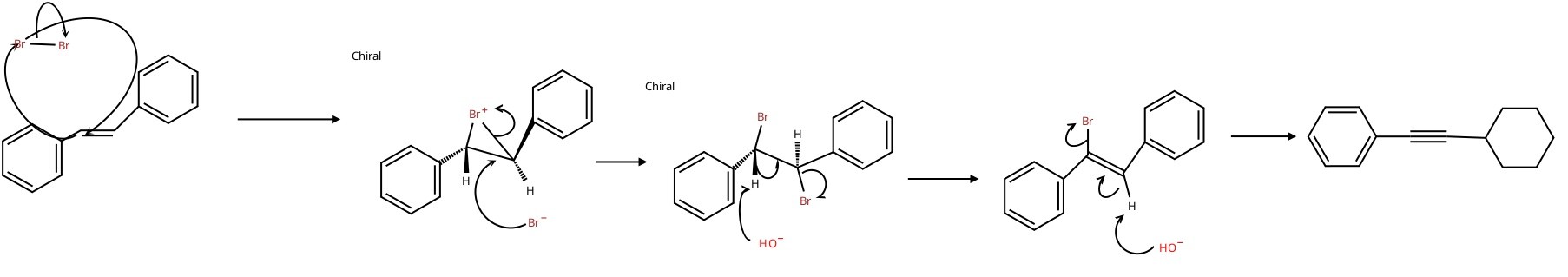
what are the ways to adjust the way a compound separates in a GC?
change the temperature of oven (turn up heat)
change column stationary phase (stationary phase)
change column length (stationary phase)
change flow rate of gas (mobile phase)
is bromide or chloride a better nucleophile?
bromide because it is larger (takes more water molecules to surround it then Cl)
in this experiment however, Cl was a better nucleophile
what is the catalyst in the William Ether Synthesis?
potassium Hydroxide, Tetra-N-Butylammonium Bromide (TBAB)
what does TBAB do?
Tetra-N-Butylammonium Bromide is a phase-transfer catalyst that works by helping ions cross between the organic and aqueous phase
what is the reaction mechanism in the William Ether Synthesis lab?
know this
what is microwave reflux?
a technique to heat reactions without losing solvent. microwaves speed up reaction times by holding the reaction at a constant temperature and pressure
what is basification?
the organic layer was washed with potassium hydroxide to remove impurities
what is the drying agent in the Williamson lab?
Organic layer was dried with sodium sulfate and removed via gravity filtration
what is used for the mobile phase?
ethyl acetate (less polar)
what is used for the stationary phase?
silica gel (more polar)