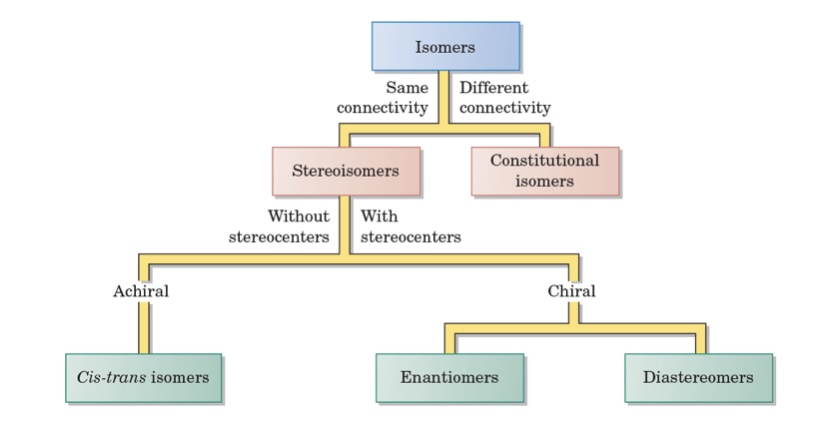
CHIRALITY
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
stereoisomers
same molecular formula and connectivity
differ in orientation/3d
constitutional isomers
same molecular formula, differ in connectivity
type of stereoisomer without stereocenters
(achiral) cis trans isomers
type of stereoisomer with stereocenters
(chiral) enantiomers, diastereomers
types of isomers
stereo, consitutional
types of stereoisomers
achiral, chiral
enantiomer
nonsuperposable mirror images, chiral, show handedness
same physical properties
cause of enantiomers
carbon w 4 different groups (stereocenter)
nonsuperposable
when you flip the image/make a mirror image and it doesnt align/look the same
enantiomers always come in
pairs
a "pair" of \(R,R\) and \(S,S\) or \(R,S\) and \(S,R\)
what makes something achiral
objects that are superposable on mirror images (look the same even when flipped and it aligns)
rs config: what to do if least priority is in front
reverse the current answer
counting from 1 to 2 to 3 during rs configuration
ignore 4 if put in between
clockwise direction is R or S?
R
R or S counterclockwise
S = Counterclockwise (left turn)
R = Clockwise (right turn) [1, 2, 3]
stereocenter formula
2^n
diastereoisomers
superposable NON-mirror images, no pairs
different physical properties e.g. melting, boiling point
Ordinary light
Light waves oscillating in all planes perpendicular to its direction of propagation.
Plane polarized light
Light waves oscillating only in parallel planes.
Polarimeter
instrument for measuring ability of compound to rotate plane of plane polarized light.
Optically active
compound is capable rotating the plane of plane polarized light.
for chiral molecules (twists light waves)
dextrorotatory
enantiomer rotating light to right (clockwise)
levorotatory
enantiomer rotating light to left (counterclockwise)
compound with exactly 1 stereocenter is always
chiral and exists as a pair of enantionmers
no plane of symmetry possible
be careful when answering # of chirality centers or stereoisomers
stereoisomers uses formula 2^n. chirality centers just asks for # without using formula yet.
chirality of molecules in plant/animal living systems
chiral (except for inorganic salts, and a few low-molecular-weight organic substances)
how many stereoisomers is found in nature
1 specific
example of enzymes with many steroecenters
chymotrypsin (digestion of proteins in intestines of animals)
251 stereocenters so max possible 2251
only one of these stereocenters is produced and used
most produce/react if they match requirements
S ibuprofen vs. R
S is active, R is inactive
S naproxen vs. R
S is active
R is liver toxin
isomer diagram
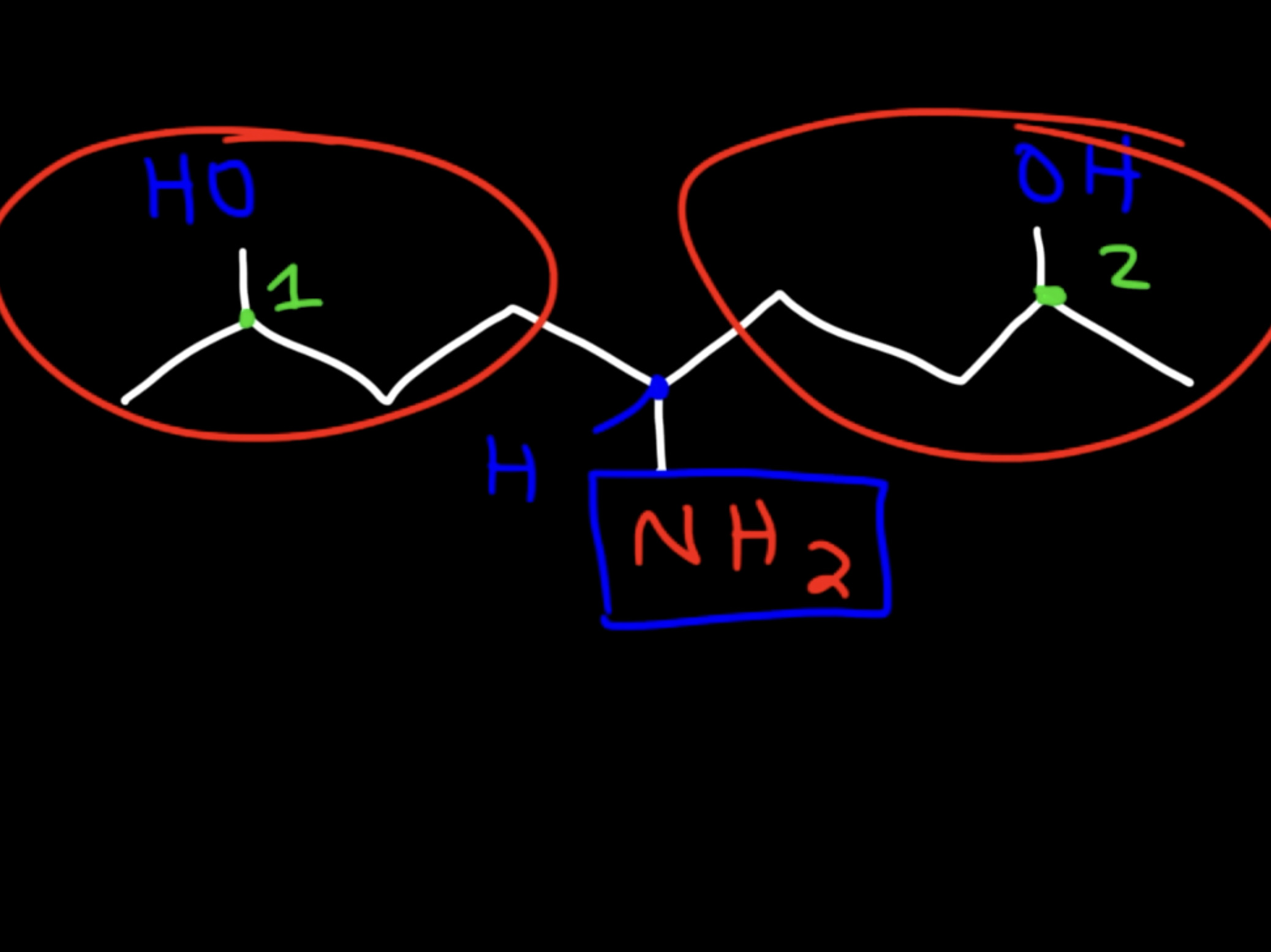
how to find stereocenter
there might be a hidden h counted as 1 different group
IGNORE
double bond attached things
secondary carbons without any substituent attached to it
CH3 at end/primary carbons at the end
WATCH OUT
watch out for identical groups bc that is equivalent to 1
finding stereocenter but it's a ring
follow path of carbon all the way back to starting point counterclockwise and clockwise
if paths in both directions same = NOT stereocenter (chiral center)
it's not the same if you encounter a substituent e.g. OH group sooner compared to the opposite path
enantiomers vs. diastereomers
enantiomers
nonsuperposable
mirror images
come in pairs
minimum chiral center: 1
same physical properties (except optical stuff)
no plane of symmetry
diastereomers
nonsuperposable
NOT mirror images
has plane of symmetry
minimum chiral center: 2
different physical properties