Fluid, Electrolyte, and Acid-Base Balance Overview
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What percentage of total body mass do body fluids make up in adults?
Between 55% and 65%.
What are the two main compartments of body fluids?
Intracellular fluid (⅔) and extracellular fluid (⅓).
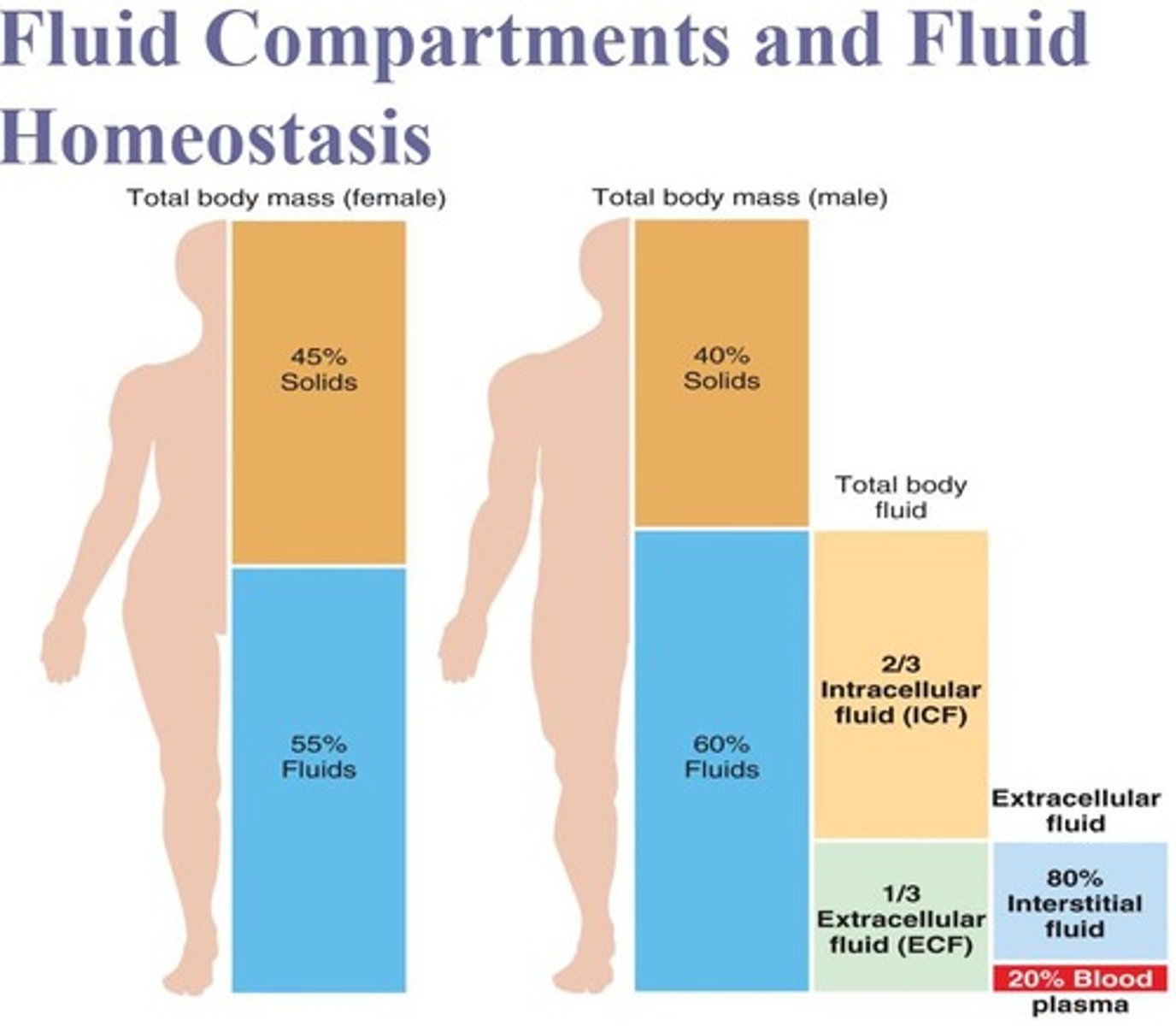
What is the primary component of intracellular fluid?
Cytosol.
What are the two subdivisions of extracellular fluid?
Interstitial fluid (80%) and blood plasma (20%).
How do plasma membranes and blood vessel walls contribute to fluid compartment separation?
Plasma membranes separate intracellular fluid from interstitial fluid, and blood vessel walls separate interstitial fluid from blood plasma.
What happens to cells when extracellular fluid is isotonic?
Cells do not shrink or swell.
What are the main pathways through which water enters the body?
Ingestion and metabolic synthesis.
How does the body primarily lose water?
Through urine, sweat, exhaling, and feces.
What is the role of the thirst center in the body?
It regulates water intake.
What are the main solutes in urine?
Sodium ions (Na+) and chloride ions (Cl-).
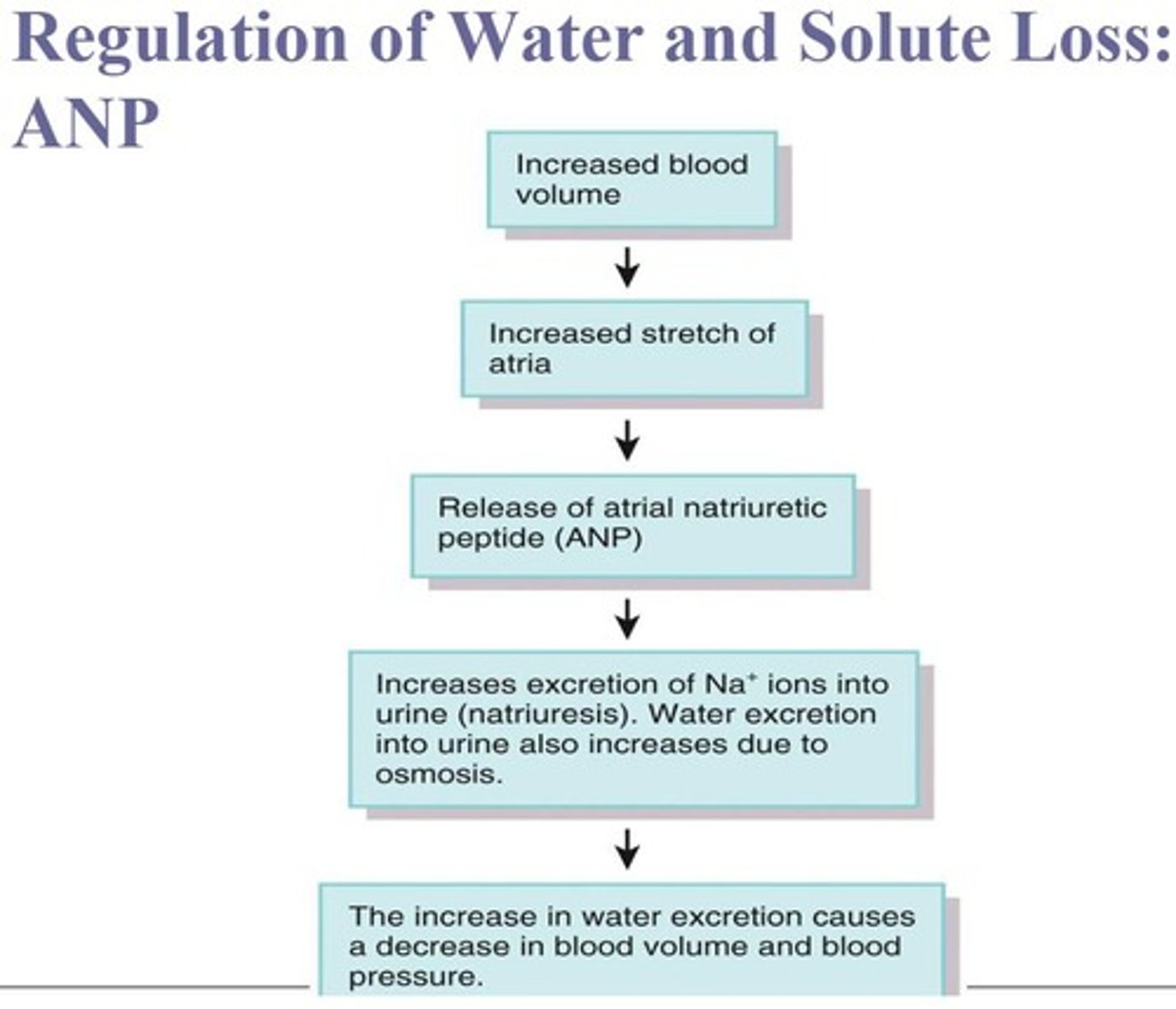
What are the three major hormones that control Na+ and Cl- homeostasis?
Angiotensin II, Aldosterone, and Atrial natriuretic peptide (ANP).
What hormone regulates water loss in the body?
Antidiuretic hormone (ADH).
How do electrolyte concentrations differ among plasma, interstitial fluid, and intracellular fluid?
Plasma contains many protein ions, interstitial fluid has few protein ions, and intracellular fluid has high concentrations of K, Mg, HPO4, and SO4.
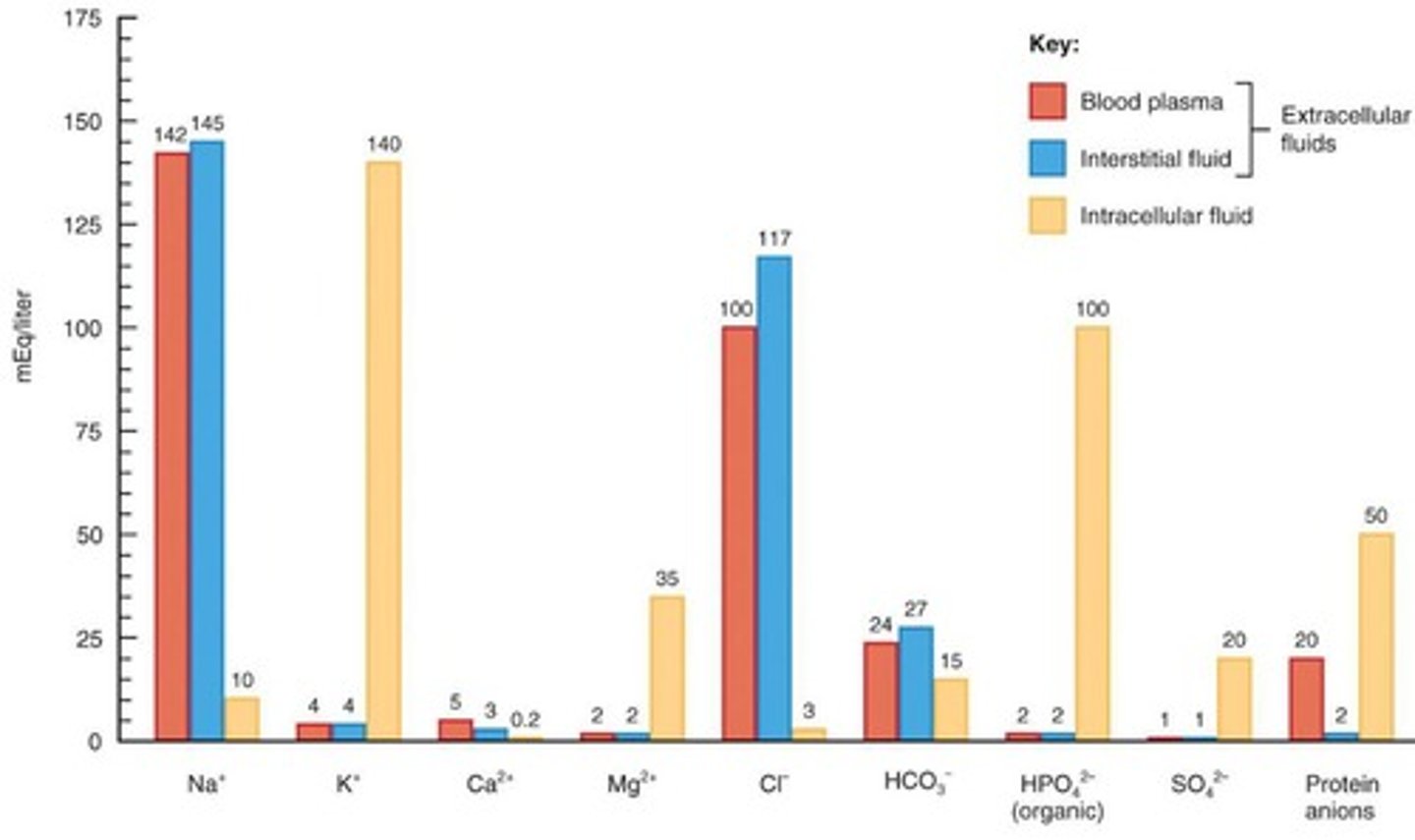
What is the primary cation in extracellular fluid and its functions?
Sodium; it is involved in impulse transmission, muscle contraction, and fluid balance.
What is the primary anion in extracellular fluid and its functions?
Chloride; it helps regulate osmotic pressure and forms HCl in the stomach.
What is the role of potassium in intracellular fluid?
It is involved in fluid volume, impulse conduction, and muscle contraction.
What is the function of bicarbonate in the body?
It acts as a major component of the plasma acid-base buffer system.
What is the most abundant mineral in the body and its functions?
Calcium; it is essential for bone structure, blood coagulation, and muscle excitability.
What is the role of magnesium in the body?
It activates enzymes involved in metabolism and is important for myocardial function.
How do ADH, aldosterone, and ANP regulate water and solute homeostasis?
ADH promotes water reabsorption, aldosterone increases sodium reabsorption, and ANP promotes sodium excretion.
What is the normal pH range of arterial blood?
7.35 to 7.45.
What mechanisms maintain the normal pH range of blood?
Buffer systems, exhalation of carbon dioxide, and kidney excretion of H+.

What is acidosis and its blood pH level?
Acidosis occurs when blood pH is below 7.35.
What is alkalosis and its blood pH level?
Alkalosis occurs when blood pH is above 7.45.
What causes respiratory acidosis?
Excessive CO2 leading to increased H2CO3.
What causes respiratory alkalosis?
Excessive loss of CO2, often due to hyperventilation.
What is metabolic acidosis?
It occurs when arterial blood levels of H+ increase and HCO3- falls.
What is metabolic alkalosis?
It occurs when blood pH rises, H+ levels fall, and HCO3- levels rise.