AP Bio Unit 1 - Chemistry of Life
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
What kind of molecule is water and how do water result in polar covalent bond?
Water is a polar molecule
The O2 is more electronegative, resulting in an unequal sharing of e- between the O2 and H
Therefore, results in polarity or polar covalent bond.
What kind of bonds do H2O molecules form and what does it result in?
Water molecules can form hydrogen bonds with each other from the + and - regions of the 2 seperate molecules
Results in surface tension by pulling the water molelues tighter together from that strong cohesive force.
Surface tensions also explain how other animals, like insects or lizards, can walk on water.
What is cohesion?
water molecules stick and form hydrogen bonds together
Cohesion results in surface tension
Also allows water to absorb a lot of thermal energy
What is adhesion and what property does it give to water?
Ability for water moleclues to stick to other surfaces
For example, water being able to move against gravity by moving from the roots of the tree to the top of the trees or to the leaves is the result in adhesion property where the water molecule sticks to the xylem walls of the tree. Its cohesion property also allows the water molecules to stick together, therefore moving the water moleclues along when water evaporates from the leaves due to sunlighlt or transpiration.
Allows water to have a high solvency (dissolve, mix with other substances like salt) ability
What is capillary action?
The adhesive force to the walls of vessel is stronger than cohesive force between liquid moleclues.
H2O resist changes in termperaure (high-heat capacity) due to
hydrogen bonds
This property help living organisms by
Maintaining homeosatsis and temperature
what is high heat vaporization and how does it help living orgranisms?
Allows water to absorb high heat energy before changing into vapor
Crucial for temperature regulation in organisms like the processes of sweating
what do living systems constantly need and where do they used this energy?
Living systems constantly need energy inputs (aexchange of matter) to sustain, grow, and reproduce
They used energy stored in chemical bonds
What element built biological molecules?
Carbon build biological moleclues (carbonhydrates, proteins, nucleic acids, lipids)
What is 96% of living things are made of?
CHON: Carbon, hydrogen, oxygen and nitrogen (96%)
including S (sulfur) and P (phosphorus) (4%)
____ is used to build proteins and nucleic acids
Nitrogen
_____ is used to build nucleic acids and certain lipds
Phosphorous
How does carbon build these bioogical macromolclues (ex: lipids)?
Carbon bonds with other carbon molecule to create a carbon skeleton that allows other moelcules to attached themselves there —- creating large, complex molecules
Carbon is also used to store energy and form cells
What is the chemical subunit to build polymers?
Monomers
What is used to build polymers and describe its steps
Dehydration Synthesis form covalent bonds between molecules to build polymers:
The subcomponenets of water are removed from interacting monomers
ONe monomer provides OH
The other monomer providdes H
the H and OH then form a water molecule, (a byproduct reaction)
What is used to break down polymers and describe its steps?
Hydrolysis reactions are used to break down polymers by breaking down covalent bonds:
Uses water to hydrolyzed (break down) polymers and the covalent bonds that form the polymer
The water is break down into H and OH and are attached to where the covalent bond used to be
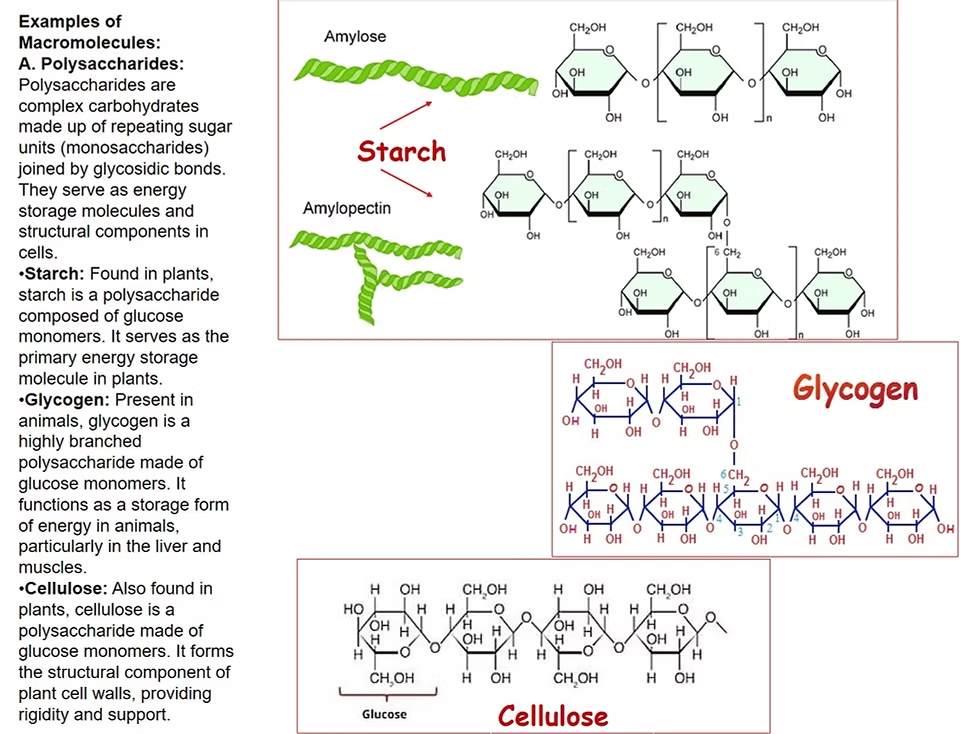
What are carbonhydrates compromise of and what are they connected by?
Carbohydrates are made of linear chains of sugar monomers
They are connected by covalent bonds known as glycosidic linkages
What can carbonhydrates polymers be and what group do these macromoleclues commonly consist?
They can be branched or linear
Carbohydrates consist of carbon, hydrogen, and oxygen (CHO)
Acronyms to remember: CHO, CHONS, CHOP, CHONP
Carbohydrates: CHO (carbon, hydrogen, oxygen)
Proteins: CHONS (carbon, hydrogen, oxygen, nitrogen, sulfur)
Lipids: CHOP (carbon, hydrogen, oxygen, phosphorus)
Nucleic acids: CHONP (carbon, hydrogen, oxygen, nitrogen, phosphorus)
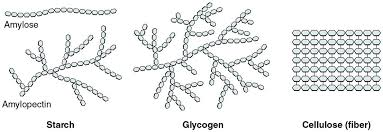
What type of carbonhydrate functions in plants and organisms energy storage?
Starch - function in plants for energy storage
Glycogen - a function in organisms for energy storage
Glycogen is highly branched than starch.
Cellulose
Type of carbohydrate that provides strength in plants´ wall
How are complex carbonhydrate polymers form?
Covalent bonds link monomers (monossacrchides)
Monossarchides are linked through glycosidic linkages
This happens through dehydration synthesis where a H atom from one monossachride from the OH combined with the OH from another monossarchride, releasing water as a byproduct and forming glycosidic linkages.
Results in the formation of polysaccharides (complex carbohydrates)
Describe the characteristics of lipids
Nonpolar
hydrophobic (has a hydrophobic tail)
their strcuture and function dervied from they way they are assembled
What do lipids contain and what are the two types?
Lipids contain fatty acids and there are two types:
Saturated fatty acid: Contain only single bonds between carbon atoms
Unsaturated fatty acid: Contain at least one single double bond between carbon atoms, which causes the carbon chain to kink
The more double bonds in fatty acid, the more
unsaturated and liqiduity it becomes
How does fat play a role in living functions?
Provide energy storage and supports cel function
Provides insulation
What happens when there is a small direction change in the components of the molecules?
Results in functional differences in moleclues.
For example, changes in carbonhydrate structure (like the branching or bond type) can alter their function by determining whether they are used for energy storage (quick or slow relsease) and structual support.
Ex: Since glycogen has a longer branch structure, it can quickly add and remove glucose, leading to faster energy release.
Example 1 of lipids: Steroids
Hormones that support psychological functions
Ex 2 of lipids: Phospholipid
Group together to form lipid bilayers in plasma and cell membranes
Phospholipid contain glycerol backbone, two fatty acid tails, and a phosphate group.
Has a hydrophilic head that interacts with the aqueous enviroment
The hydrophobic tail is on the inside to avoid the aqeuous enviroment.
Therefore, this structure creates a bilayer that acts as a selective entrance for substances.
What are lipids made out of and are they different from other large macromoleclues?
Lipids are made from fatty acid and glycerols (monomers) through dehydration synthesis
The COOH from fatty acids reacts with the OH from glycerol; water is removed during the dehydration synthesis
Forms a covalent bond known as Ester linkage between glycerol and each fatty acid
Triglycerides: Forms after 3 dehydration synthesis reactions.
Although lipids are polymers, they are not true polymers since they do not consist of long chains of repeating monomers like proteins and carbohydrates do.
Explain/Exemplify how lipid structure supports energy storage, membrane fluidity, and signaling.
Examples include:
Phospholipid with its amphiatic nature makes them have a selective permeable membrane.
Unsaturated with its double bonds prevent them from tight packing making them more liquidity.
What are the components of a nucleotide?
Each nucelotide has this following structual component:
A phosphate group
A nitrogenous base
A five carbon sugar (deoxyribose or ribose)
What is the difference between DNA and RNA based on structure and function?
DNA:
A deoxyribose sugar
Contains thymine
Double stranded
Anti-parallel
stores and pass down genetic information (acts like a blueprint)
RNA:
A ribose
contains uracil
single stranded
Interpret and carry out genetic information to synthesizing proteins.
Explain the base pairing rules for nucleic acids and their importance for information storage.
In DNA:
Adenine pairs with thymine (via two hydrogen bonds)
Guanine pairs with Cystonine (via three hydrogen bonds)
In RNA:
Adenine pairs with uracil
These specific base pairing rules create the stable structure of a double helix, providing a strong framework for genetic information and the replication of DNA during cell division
Describe the directionality of the nucleic acids
Each linear strand of nucleotides has a 5’ phosphate and a 3’ hydroxl of the sugar
The two sugar phosphate backbone run in opposite directions of 5’-3’ is known as antiparallel
During nucleic acid synthesis, nucleotides are added to the 3’ end of the growing strand resulting in the formation of covalent bonds between nucleotides.
Explain the process of dehydration synthesis in nucleotides.
Nucleotides form nucleic acids through dehydration synthesis where:
A hydrogen atom from the phosphate group in on the 5’ form a covalent bond (phosphodiester bond) with the hydroxyl group on the 3’
H2O then becomes an additional byproduct
How does the structure of DNA play a role in genetic information and storing?
The specific sequences of the bases from the polymers of nucleoties such as A, C, T, G, U in RNA and DNA forms the genetic code.
The DNA’s double helix provides a stable mechanism for copying genetic code
Proteins are...
macromoleclues made out of linear chains of amino acids.
Has a carboxyl group (COOH)
Has an amine group (NH2)
Has a central carbon
A unique R group
How does the amino acids form covalent bonds
Amino acids form peptide bonds through condensation reaction (dehydration synthesis)
The Carboxyl reactions with the amine group to form a peptide bond between the carbon in COOH and the nitrogen in NH2.
What is important about R-groups in amino acids?
The R groups gives amino acids properties and whether they are polar/nonpolar/acidic/basic
R-group interactions determine the shape
The shape then determines the function of the protein
Primary structure
Sequences of amino acids in a protein
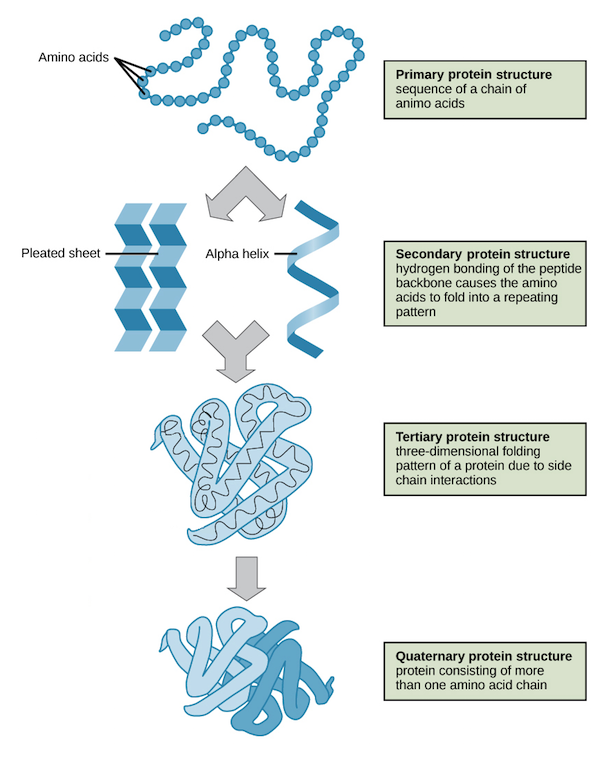
Secondary structure
Involves folding of a protein into alpha helices or beta pleated sheets due to hydrogen bonds that form between COOH and NH2 groups of the polypeptide backbone.

Tertiar ystructure
Protein’s 3D shape is determined by R-group interactions:
Hydrophobic interactions: Between nonpolar r-groups and has hydrocarbon rings
Hydrogen bonds: Forms between polar R-groups and has H-O-H-O
Disulfide bond: Form between the sulfur containing R-groups
Ionic bonds: Form between two oppositely charged R-groups (NH3+ ———- O-)

Quaternary structure
Determine by interactions between polypeptides
All four levels of structure determine the protein’s functions.
