Chemistry Final
1/120
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
121 Terms
reduction
+ to -
oxidizing agent
“gain”
oxidation
- to +
reducing agent
“loss”
Breaking bonds is: endo or exothermic?
endothermic
Hmixture
less than 0 —> exothermic
Hsolution
Hsolute + Hsolvation
Hlattice + Hhydration
Hhydration
less than 0 —> exothermic
Hsolute
greater than 0 —> endothermic
Hsolvent
greater than 0 —> endothermic
osmosis
lower concentration —> higher concentration
deals specifically with movement of solvents —> NOT ions
attempting to reach equilibrium
Highest freezing point means…
the smallest change (in m x i)
“concentration”
M- molarity
higher charge density means…
releasing more energy, smaller size, and larger Hhydration
more Hhydration means…
stronger ion-dipole forces
Solution has higher/lower entropy compared to pure solvent/solute?
HIGHER
g —> s
s < 0
exothermic
s —> g
s > 0
endothermic
Main factors that affect rxn rate
concentration: molecules must collide to react
temperature: molecules must collide with enough energy
higher T = more energy = more collisions
physical state: molecules must mix to collide
greater surface area = more contact to react
catalyst: substance that accelerates chemical rxns —> not consumed itself
rate formula
change in concentration/change in time
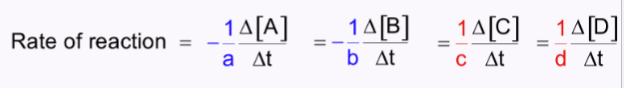
Reaction rates
average
instantaneous
initial
A catalyst is seen in the ____ side first, and the ____ side second
A catalyst is seen in the reactant side first (consumed first), and then in the product side second (produced second)
average kinetic energy depends on…
ONLY on the temperature —> the the same temperature, two molecules will have the same kinetic energy
average speed depends on…
the SIZE —> a bigger molecule will have a SLOWER average speed
what does a catalyst do to a rxn?
it lowers the activation energy; you know its a catalyst because it is above the arrow
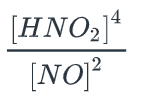
order of HNO2, order of NO; what if HNO2 is tripled; what if NO is doubled; overall order?
order HNO2: 4
order NO: -2
HNO2 tripled: 34 = 81
NO doubled: 22 = 1/4
overall order: 2
Density equation
mass/volume
how is rxn order found?
found experimentally
collision theory
must collide
collide with enough energy
collide at right orientation
# collisions = product # particles
asking for how much of a product in a rate law question?
solve for amount of reactant, subtract from initial amount, and multiply by the balanced equation
overall rate is always positive/negative?
positive
the more likely for a collision to occur, the higher/lower the rate?
higher
what kind of substances dissolve in water?
ionic and polar substances —> like dissolves like
small alcohols will more easily dissolve in…
ionic compounds
large alcohols will more easily dissolve in…
hexane
1 cm3 =
1 mL
colligative properties depend on…
the number of particles present
“vapor pressure lowering”
delta P
for a BP question with water as the solvent, always add…
100 degrees C
for a FP question with water as the solvent, always subtract from…
0 degrees C
molality
mol solute / kg solvent
solute vs. solvent
solute = what is dissolved
solvent = what it is being dissolved in
are gases very soluble?
NO
solvation
surrounding a solute particle with solvent particles
solvation in water
hydration
charge density
ratio of ion’s charge to its volume
entropy is also known as
disorder
the solubility of gases in water increases/decreases as temperature increases?
decreases —> solubility of gases in water and temperature are inversely related
units Sg is usually in
gas solubility is usually in mol/L
molarity equation
amount of solute / volume of solution
examples of colligative properties
VP
BP
FP
osmotic pressure
simple cubic
1 atom/unit cell
a = 2r
r = a/2
coordination number = 6
worst packing efficiency
body cubic
2atoms/unit cell
√3a = 4r
r = a√3/4
coordination number = 8
middle packing efficiency
face centered
4 atoms/unit cell
√2a = 4r
r = a√2/4
coordination number = 12
highest packing efficiency
Angstrom to cm
1 A = 1 × 10-8 cm
volatile liquids
easily vaporized
high VP
weak IMK
polar shapes
bent
trig pyramidal
T-shaped
see-saw
square pyramidal
As IMF increase…
VP decreases
BP increases
size increases
polarizability
increases as size increases
Less cats, more ants!!
cation < neutral
anion > neutral
convex
strong cohesion but weak adhesion
concave
strong adhesion but weak cohesion
bond length
more bonds = shorter
bigger bond order = longer
finding enthalpy: given bond order
reactants - products
bond order
larger bond order = stronger bond
a colling curve in endo/exothermic?
endothermic
a heating curve in endo/exothermic?
exothermic
“normal BP”
use 1 atm
the more dispersion forces, and larger/smaller?
larger
viscosity increases/decreases as IMF increases
increases
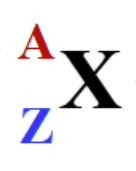
what do A and Z represent
A = mass number —> protons plus neutrons
Z = atomic number —> protons
group 1
alkali metals
group 2
alkaline earth metals
group 7
halogens
group 8
noble gases
covalent bonding
between two non-metals
share electrons
ionic bonding
between a metal and a non-metal
transfer of electrons
charge of Hg2
2+
Alkanes are ____ compounds
organic
My Enormous Penguin Bounces Pretty High, Helping Our Neighbor David
CxH2x+2
heterogeneous mixture
visible boundary, not uniform
oil and vinegar
homogeneous mixture
no visible boundaries
solution
table salt dissolved in water
ways to separate a mixture
filtration
distillation
crystallization
chromatography
pure substances include…
elements and compounds
mass fraction
mass X / mass entire compound
theoretical yield definition
max product yield that can be expected based on the masses of the reactants —> can never be 100%
overall yield
multiply the yields of each step together (as decimals)
water’s solvent power
uneven charge distribution
bent molecular shape
molecular polarity —> dissolves polar and ionic substances
strong vs. weak vs. non electrolytes
strong electrolytes: completely dissolve —> ionic compounds
weak electrolytes only partially dissolve
nonelectrolytes: do not dissolve —> covalent compounds
strong acids
hydro___ic acid
nitric acid
perchloric acid
sulfuric acid
Strong bases
b on the periodic table
acid + base —>
salt + water
T/F: acids and bases must be ionic if they can dissociate?
FALSE
Bronsted-Lowry acid and base
acid donates an H+
base accepts an H+
How to find value of x for a hydrate?
find mols of hydrate
find mols of salt
divide mols hydrate by mols of salt
molecular vs. empirical formula
empirical is the formula you solve for initially, molecular formula is multiplied by an integer
effusion
x effuses said times amount as y
rate x / rate y = √y / √x
how to rank density of compounds
least dense = smallest molar mass
“neutralizes” means find…
which equation given an even number mols of H+ and OH-
“collected over water”
find mols of thing you are looking for → subtract pressure of water from total pressure —> solve for volume
relationship between P and V
P and V are inversely related
relationship between T and V
proportional
Pfinal
Pinitial x Vinitial / Vfinal
partial pressure x
mole x / mole x + mole y