5 - chemical energetics
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
enthalpy
(heat content) The total chemical energy inside a substance
When chemical reactions take place, changes in chemical energy take place and therefore the enthalpy changes
(energy can only be given out or taken in in the form of heat, so effectively, all molecules and substances have heat energy stored inside them)
enthalpy change is represented by the symbol
ΔH (Δ= change; H = enthalpy)
an enthalpy change can be positive or negative
a reaction is exothermic when (give examples)
the products have less energy than the reactants
Heat energy is given off by the reacting particles to the surrounding particles (making them vibrate more and increasing their temp)
The temperature of the environment increases - this can be measured on a thermometer
The temperature of the system decreases
eg - neutralisation, combustion, condensing, solidifying
ΔH during exothermic reactions
there is an enthalpy decrease during the reaction so enthalpy change is negative
Exothermic reactions are thermodynamically possible (because the enthalpy of the products is lower than that of the reactants)
However, if it is possible, why is the rate sometimes too slow to observe any appreciable reaction?
This could be because the reaction is kinetically controlled
This means the reaction could have a high activation energy which is preventing the reaction from taking place.
an endothermic reaction is when (give examples)
the products have more energy than the reactants
Heat energy is absorbed by the reaction from the surroundings
The temperature of the environment decreases because the surrounding particles have less energy to move and vibrate - this can be measured with a thermometer
The temperature of the system increases
eg - photosynthesis, melting, evaporating
enthalpy change during endothermic reactions
There is an enthalpy increase during the reaction so ΔH is positive
system
the molecules that are reacting in the reaction itself
surroundings
everything surrounding the reacting molecules/ outside the system that aren’t taking place in the reaction (eg - the flask the reaction is taking place in or the water molecules in an aqueous reaction)
examiner tip : It is important to specify the physical states of each species in an equation when dealing with enthalpy changes as any changes in state can cause very large changes of enthalpy.
For example:
Na+Cl- (s) → Na+ (aq) + Cl- (aq) ΔH = +4 kJ mol-1
Na+Cl- (g) → Na+ (g) + Cl- (g) ΔH = + 500 kJ mol-1
-
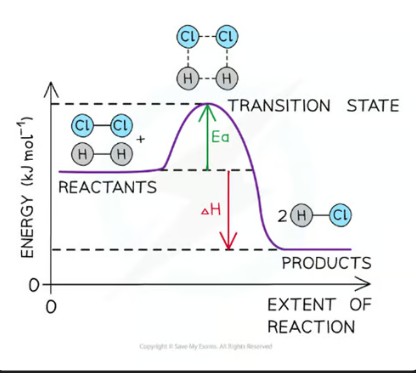
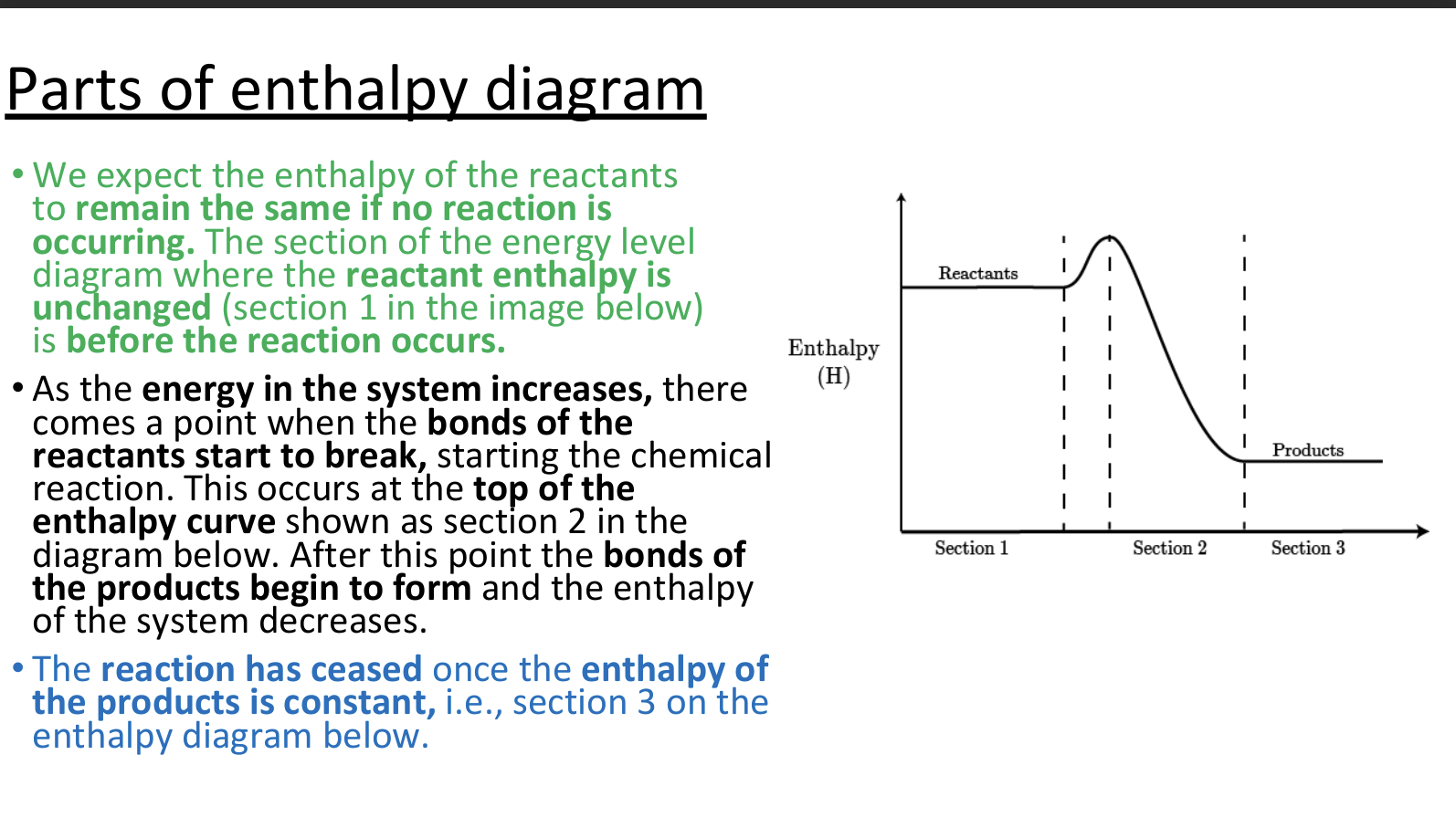
a reaction pathway diagram/ energy level diagram shows
the energies of the reactants, the transition state(s) and the energies of products of the reaction with time
always plot the enthalpy of the system on the y-axis and the progress of the reaction on the x-axis.
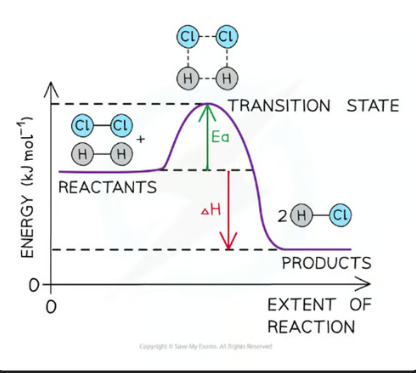
transition state
a moment during a chemical reaction when the original bonds are not completely broken and the new bonds are not fully formed. It’s a highly unstable, short-lived state that cannot be isolated. On an energy profile, the transition state is represented by the highest point — it has more energy than both the reactants and the products.
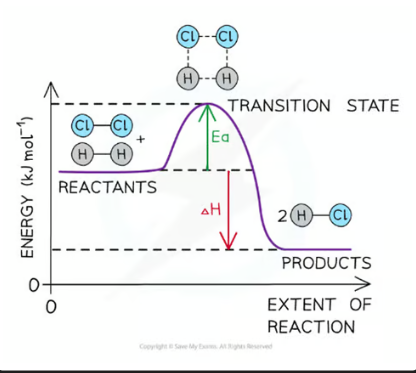
activation energy (Ea)
the minimum amount of energy needed for reactant molecules to have a successful collision and start the reaction (the energy needed to reach the transition state) - it will always be a positive value
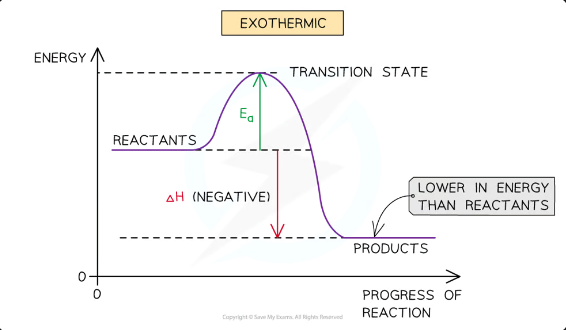
reaction pathway diagrams for exothermic reactions
In an exothermic reaction, the reactants are higher in energy than the products This means the products of exothermic reactions are more thermodynamically stable than the reactants (lower energy = more stable)
The reactants are therefore closer in energy to the transition state
This means that exothermic reactions have a lower activation energy compared to endothermic reactions
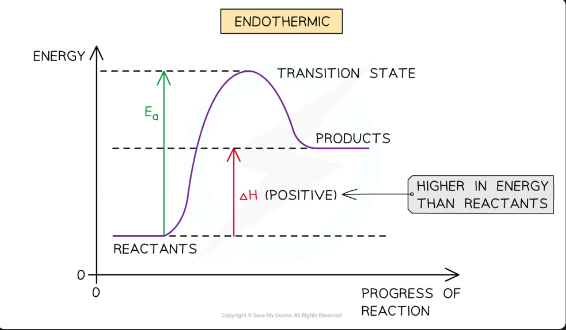
reaction pathway diagrams for endothermic reactions
In an endothermic reaction, the reactants are lower in energy than the products. This means the reactants of endothermic reactions are more thermodynamically stable than the products.
The reactants are therefore further away in energy to the transition state
This means that endothermic reactions have a higher activation energy compared to exothermic reactions
The activation energy is the energy difference from
reactants to the transition state (top of the curve)
The enthalpy change of the reaction is the energy difference from
reactants to products
To enable fair comparisons to be made between changes in enthalpy between different types of reactions, all reactions should be carried out under standard conditions
These standard conditions are:
a pressure of 101 kPa
a temperature of 298 K (25 oC)
Each substance involved in the reaction is in its normal physical state (solid, gas or liquid)
all amounts are per mole/molar quantities
which symbol is used to show that a reaction has been carried out under standard conditions
⦵
what is the definition of standard enthalpy change of reaction, its symbol and is it exothermic or endothermic
The enthalpy change when the reactants in the stoichiometric equation react to form the products under standard conditions
ΔHθr
both
what is the definition of standard enthalpy change of formation, its symbol and is it exothermic or endothermic
The enthalpy change when one mole of a compound is formed from its elements under standard conditions
ΔHθf
both
what is the definition of standard enthalpy change of combustion, its symbol and is it exothermic or endothermic - give a reason
The enthalpy change when one mole of a substance is burnt in excess oxygen under standard conditions
ΔHθc
exothermic (because even though breaking bonds on reaction side is endothermic, the products release much more energy when forming bonds than what was absorbed when breaking bonds)
what is the definition of standard enthalpy change of neutralisation, its symbol and is it exothermic or endothermic
The enthalpy change when one mole of water is formed by reacting an acid and an alkali under standard conditions
ΔHθneut
exothermic
what is the definition of standard enthalpy change of solution, its symbol and is it exothermic or endothermic
the enthalpy change when one mole of solute is dissolved in a solvent to form an infinitely dilute solution (a solution where the solute concentration is negligible)
ΔHθ sol
both
what is the definition of standard enthalpy change of atomisation, its symbol and is it exothermic or endothermic
the enthalpy change when one mole of gaseous atoms is formed from its element under standard conditions
ΔHθ at
endothermic (because energy needed to break the bonds)
what is the definition of standard enthalpy change of hydration of an anhydrous salt, its symbol and is it exothermic or endothermic
the enthalpy change when one mole of hydrated salt is formed from one mole of the anhydrous salt under standard conditions
ΔHθ hyd
exothermic
why is enthalpy change used and not just enthalpy (also explain how it should be measured)
It is extremely difficult to measure the enthalpy of a system.
Thus, we always refer to a change in enthalpy (ΔH). This is the difference in enthalpy between the reactants and products in a chemical reaction.
it should be measured at constant pressure because pressure changes can cause changes in temp (eg high pressure = high temp)
The ΔHθf of an element in its standard state is
zero
why do enthalpy changes take place during a reaction
because bonds are being broken and formed
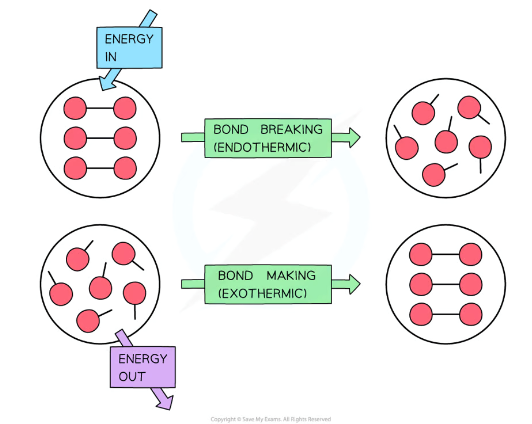
is bond breaking endothermic or exothermic
endothermic because energy (in the form of heat) is needed to overcome attractive forces between atoms and they gain kinetic energy as they move apart (temp decreases)
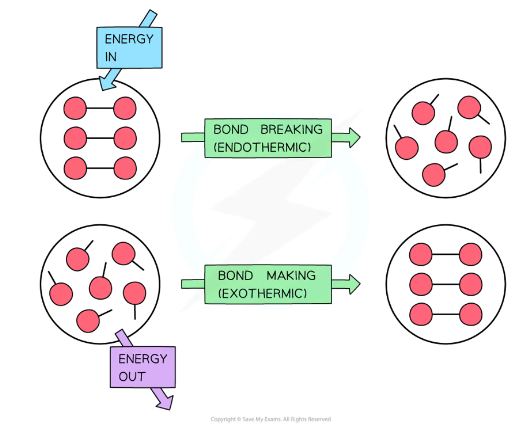
is bond forming endothermic or exothermic
exothermic because energy is released from the reaction to the surroundings (as heat) when new bonds are formed. When a chemical bond forms, the atoms slow down slightly as some of their kinetic energy is turned into stored chemical potential energy in the bond. At the same time, the rest of the energy is released as heat into the surroundings.
when is the reaction endothermic in terms of bond forming/breaking
If more energy is required to break bonds than energy is released when new bonds are formed, the reaction is endothermic
when is the reaction exothermic in bond forming/breaking
If more energy is released when new bonds are formed than energy is required to break bonds, the reaction is exothermic
bond dissociation energy
The amount of energy required to break one mole of a specific covalent bond in the gaseous state
Bond dissociation energy (E) is also known as exact bond energy or bond enthalpy
how is the type of bond broken written in bond enthalpy
The type of bond broken is put in brackets after E
Eg. EE(H-H) is the bond energy of a mole of single bonds between two hydrogen atoms
average bond energy/ mean bond enthalpy
The energy needed to break a bond can change depending on what other atoms are around it in the molecule.
Because of this, scientists don't just use the bond energy from one molecule — they average the bond energies from several different molecules where the same bond appears.
This average is called the average bond energy (or average bond enthalpy)
since bond energies cannot be determined directly, what is used to calculate average bond energy
enthalpy/hess cycles or using experimental data
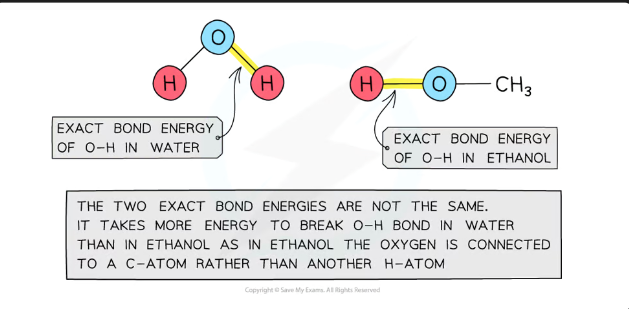
Bond energies are affected by other atoms in the molecule
-
what are bond energies used for
to find the ΔHrꝋ / overall enthalpy change of a reaction when this cannot be done experimentally
E.g. the Haber Process
The equation to calculate the standard enthalpy change of reaction using bond energies is:
ΔHθr = enthalpy change for bonds broken + enthalpy change for bonds formed
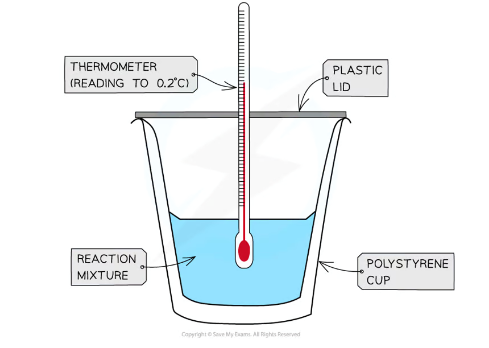
what technique is used to measure changes in enthalpy of chemical reactions
calorimetry - a calorimeter can be made up of a polystyrene drinking cup, a vacuum flask or metal can
explanation and calculation of a reaction using calorimeters
The energy needed to increase the temperature of 1 g of a substance by 1 oC is called the specific heat capacity (c) of the liquid
The specific heat capacity of water is 4.18 J g-1 oC-1
The energy transferred as heat can be calculated using the following equation:
q = m x c x ΔT
examiner tip : When new bonds are formed the amount of energy released is equal to the amount of energy absorbed when the same bonds are broken
For example:
O2 (g) → 2O (g) E (O=O) = +498 kJ mol-1
2O (g) → O2 (g) E (O=O) –498 kJ mol-1
Aqueous solutions of acid, alkalis and salts are assumed to be largely water so you can just use the m and c values of water when calculating the energy transferred
To then calculate any changes in enthalpy per mole of a reactant or product the following relationship can be used:
ΔH = –m x c x ΔT
When there is a rise in temperature, the value for ΔH becomes negative suggesting that the reaction is exothermic
When there is a fall in temperature, the value for ΔH becomes positive suggesting that the reaction is endothermic
Also, remember that ΔT is the same in oC and K!
-
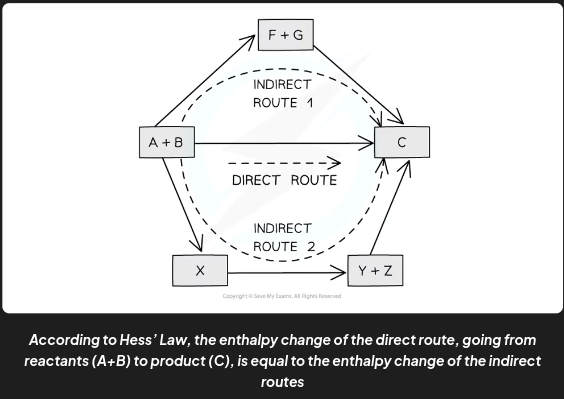
Hess’s law states that
The total enthalpy change in a chemical reaction is independent of the route by which the chemical reaction takes place as long as the initial and final conditions are the same.
This means that whether the reaction takes place in one or two steps, the overall enthalpy change of the reaction will still be the same
These two processes (breaking and forming chemical bonds) are opposites of each other and demonstrate that
any process which is endothermic in one direction will be exothermic in the opposite direction. (forward/backward reaction)
Hess’ Law is used to
calculate enthalpy changes which can’t be found experimentally using calorimetry, e.g.:
3C (s) + 4H2 (g) → C3H8(g)
ΔHf (propane) can’t be found experimentally as hydrogen and carbon don’t react under standard conditions
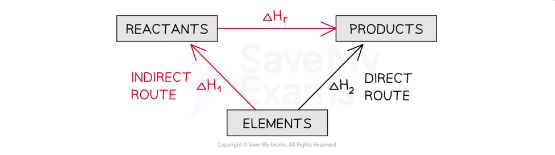
Calculating ΔHr from ΔHf using Hess’s Law energy cycles
The products can be directly formed from the elements = ΔH2
OR
The products can be indirectly formed from the elements = ΔH1 + ΔHr
equation for calculating ΔHr from ΔHf using Hess’s Law energy cycles
ΔH2 = ΔH1 + ΔHr
Therefore,
ΔHr = ΔH2 – ΔH1
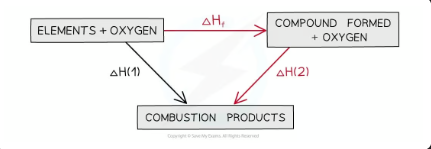
Calculating ΔHf from ΔHc using Hess’s Law energy cycles
The combustion products can be formed directly from elements to combustion products = ΔH1
OR
The combustion products can be formed indirectly from elements to compounds to combustion products = ΔHf + ΔH2
equation for calculating ΔHf from ΔHc using Hess’s Law energy cycles
ΔH1 = ΔHf + ΔH2
Therefore,
ΔHf = ΔH1 – ΔH2
Calculating average bond energies using Hess's cycles
Bond energies cannot be found directly so enthalpy cycles are used to find the average bond energy
This can be done using enthalpy changes of atomisation and combustion or formation
The enthalpy change of atomisation (ΔHθat ) is the enthalpy change when one mole of gaseous atoms is formed from its elements under standard conditions.
E.g. ΔHθat [H2] relates to the equation:
½H2 (g) → H (g)
examiner tip
Remember to take into account the number of moles of each reactant and product.
For example, there are two moles of NaHCO3 (s) so the ΔHf value is multiplied by 2.
law of conservation of energy
energy is neither created nor destroyed, but is converted from one form to another. While the total energy remains the same, the form of the energy can change.
chemical energy (also explain potential energy)
Chemical energy is the energy stored in the bonds of chemical substances. It's a type of potential energy, and it can be released or absorbed during a chemical reaction.
(Potential energy is stored energy—it’s the energy something has because of its position or structure, and it has the potential to do work or be released.)
Strong bonds = lower potential energy (more stable)
Weak or strained bonds = higher potential energy (less stable)
thermal/heat energy and how is heat different from temperature
the energy a substance has because of the total average movement (kinetic energy) of its particles.
Temperature is a measure of the average kinetic energy of the particles in a substance.
Heat is the transfer of thermal energy from one substance to another due to a temperature difference. Heat energy always flows from objects at a higher temp to lower temp
Even if you give the same amount of heat energy to two different substances, they won't always heat up the same way—one might get hotter than the other. Which 2 factors are the reason for this?
the amount of substance (its mass)
the nature of the substance (its specific heat capacity)
explain how the mass/amount of a substance causes two substances given the same amount of heat energy to heat up differently
The same amount of heat flowing into two different objects may result in different temperature changes. An object with a lower number of particles will experience a greater change in temperature than an object with a greater number of particles.
The object with a smaller number of particles has to share the heat energy between fewer particles, therefore each individual particle will experience a greater change in kinetic energy. This will cause a greater change in the average kinetic energy and temperature of the object.
eg : using the same amount of batter to make a cake vs 12 cupcakes - the cupcakes will bake faster at the same oven temp
explain how the nature/specific heat capacity of a substance causes two substances given the same amount of heat energy to heat up differently
Substances that are made up of different types of atoms or molecules will also have different types of bonds between their atoms or molecules. These different types of bonds have different capacities to absorb heat.
Some bonds will be capable of absorbing large amounts of heat and therefore experience large changes in temperature when exposed to heat energy. Other bonds are not very capable at absorbing heat and objects with these bonds will not experience significant changes in temperature when exposed to heat energy.
-
-
The two processes (breaking and forming chemical bonds) are opposites of each other and demonstrate that
any process which is endothermic in one direction will be exothermic in the opposite direction. (forward/backward reaction)