CHEM-2021 Lab FINAL (Shilabin/Bryant)
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
Esterification
A reaction between a carboxylic acid (or acid halide or anhydride) with an alcohol to form an ester.
Acid Ion-Exchange Resin
Cross-linked polystyrene beads that bear sulfonic acid groups; it generates acid (H+) in a solution although the resin remains insoluble.
Azeotropic Distillation
Distillation of certain compounds together with water at a temperature different than the normal boiling point of the pure compound.
Hydrolysis
A reaction that involves addition of water to the molecule which results in splitting it up into smaller molecules.
Ester Hydrolysis
Splitting the ester, upon addition of water, into carboxylic acid and alcohol.
Saponification
Basic (alkaline) hydrolysis of ester to form alcohol and the metal salt of carboxylic acid (carboxylate).
Fatty Acids
Long chain carboxylic acids (>10 C). Natural fatty acids contain an even number of carbons.
Hard Water
Contains relatively high concentrations of calcium and/or magnesium salts which reduce solubility of soap.
Condensation
Addition reaction between two molecules that is associated with loss of a small molecule such as water and alcohol.
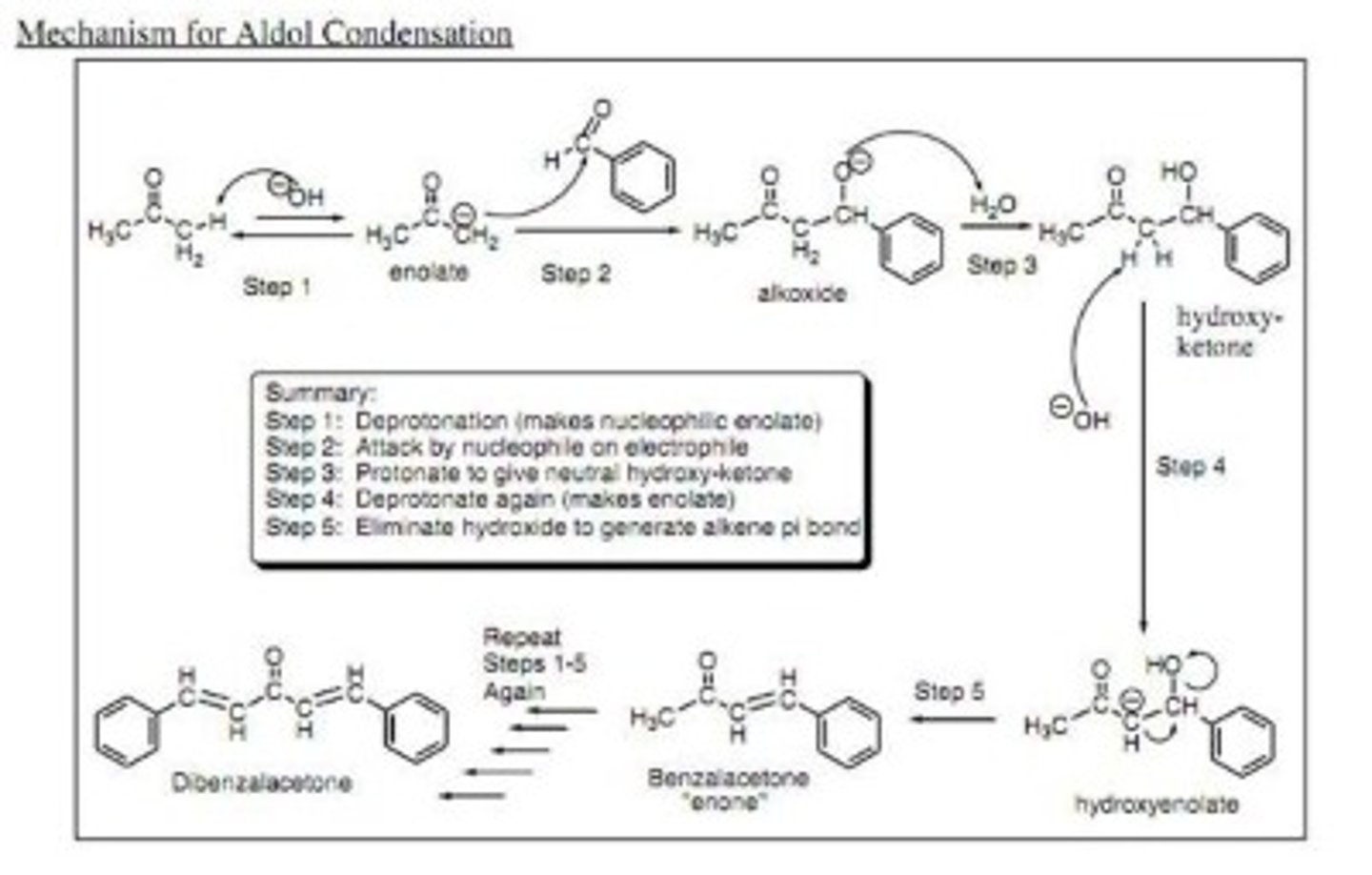
Aldol
A compound containing an aldehyde (ald) group and alcohol (ol) group.
Aldol Addition
Nucleophilic addition reaction between two aldehyde molecules; the product is normally beta-hydroxy aldehyde (aldol).
Aldol Condensation
Condensation reaction between two aldehyde molecules with a loss of water; the product is normally an alpha,beta-unsaturated aldehyde.
Enolate Ion
Resonance structure of the carbanion in which the negative charge is on an oxygen atom.
Electrophile
A positively charged atom, or a positive end of a dipolar compound, that seeks electrons to complete its octet shell.
Electrophilic Aromatic Substitution (EAS)
A reaction by which hydrogen on aromatic rings are replaced by electrophiles.
Regio-Selectivity
A selective direction of a chemical reaction toward one part of a compound, rather than other parts.
Reaction Mechanism
A step-by-step description of a chemical reaction at the molecular level.
Activating/Deactivating Group
A group on an aromatic ring that increases/decreases its reactivity toward electrophiles.
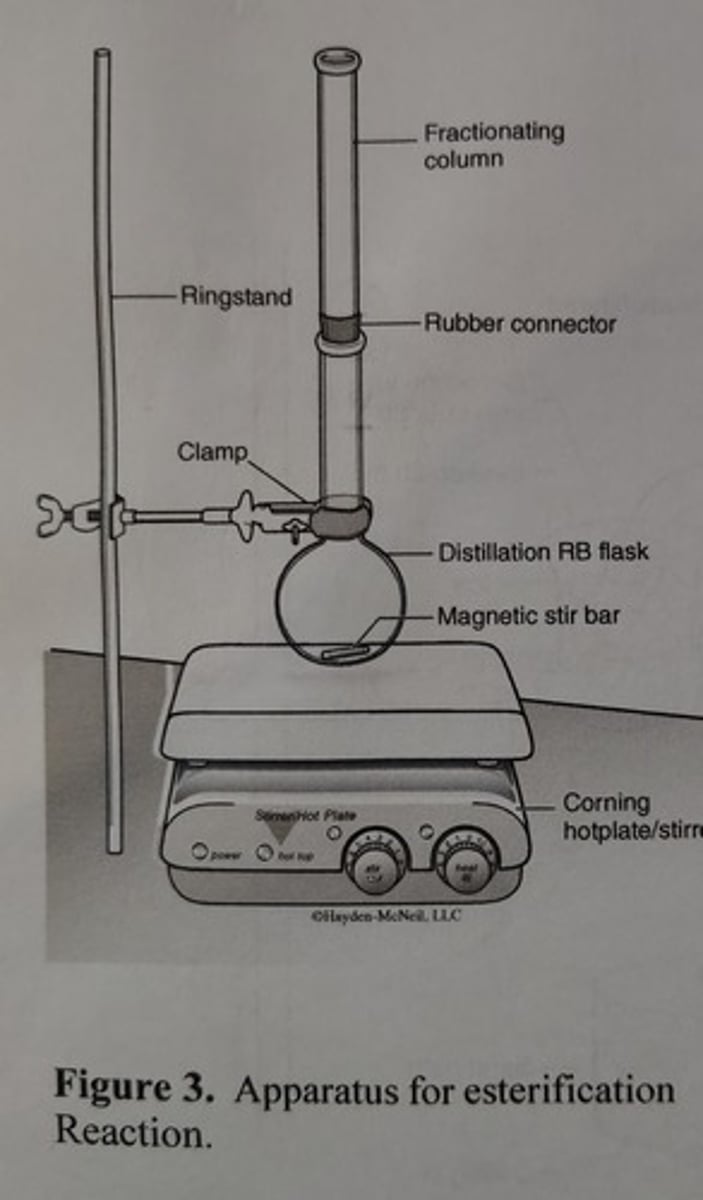
Apparatus for Esterification Reaction
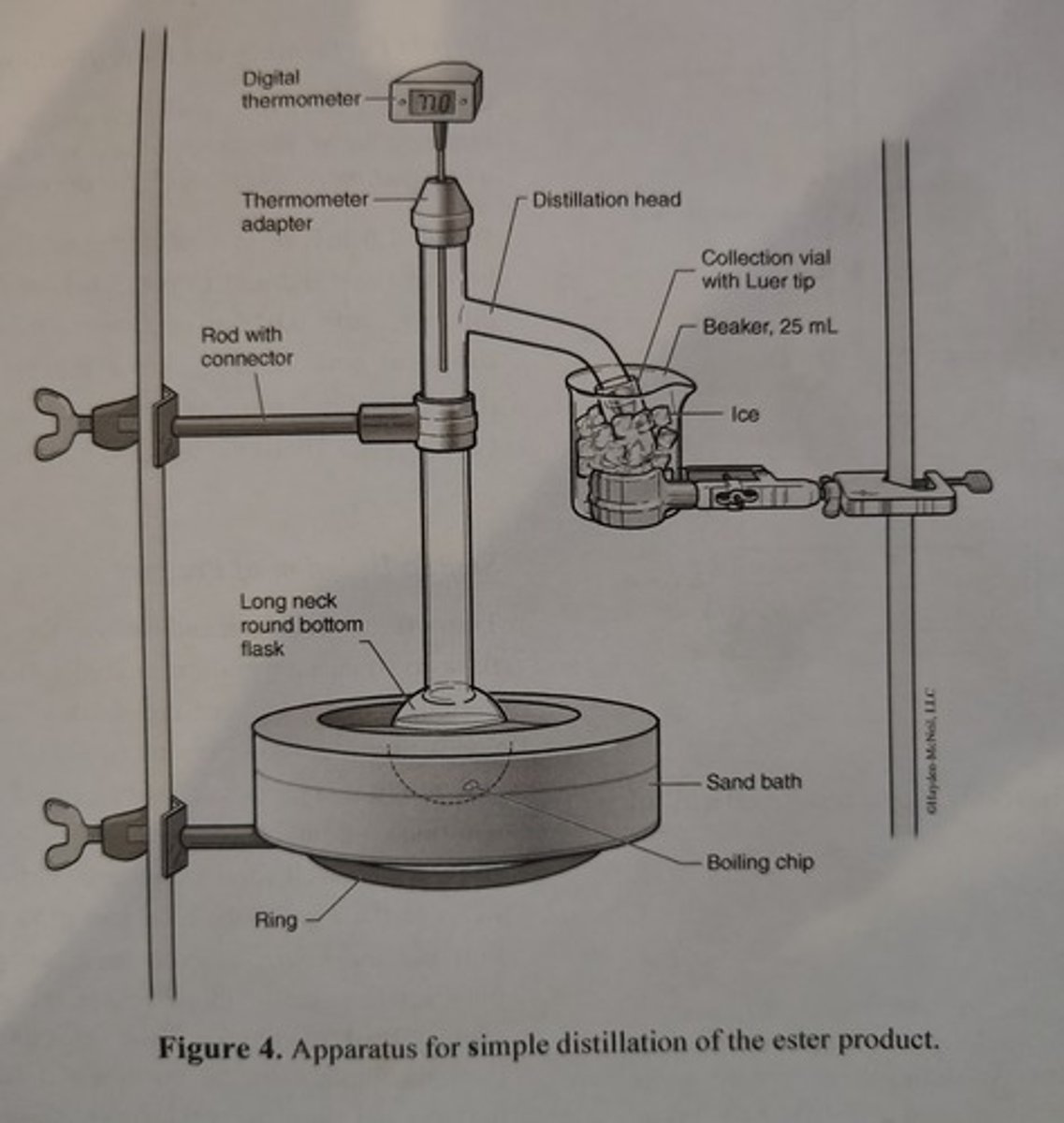
Apparatus for Simple Distillation of the Ester Product
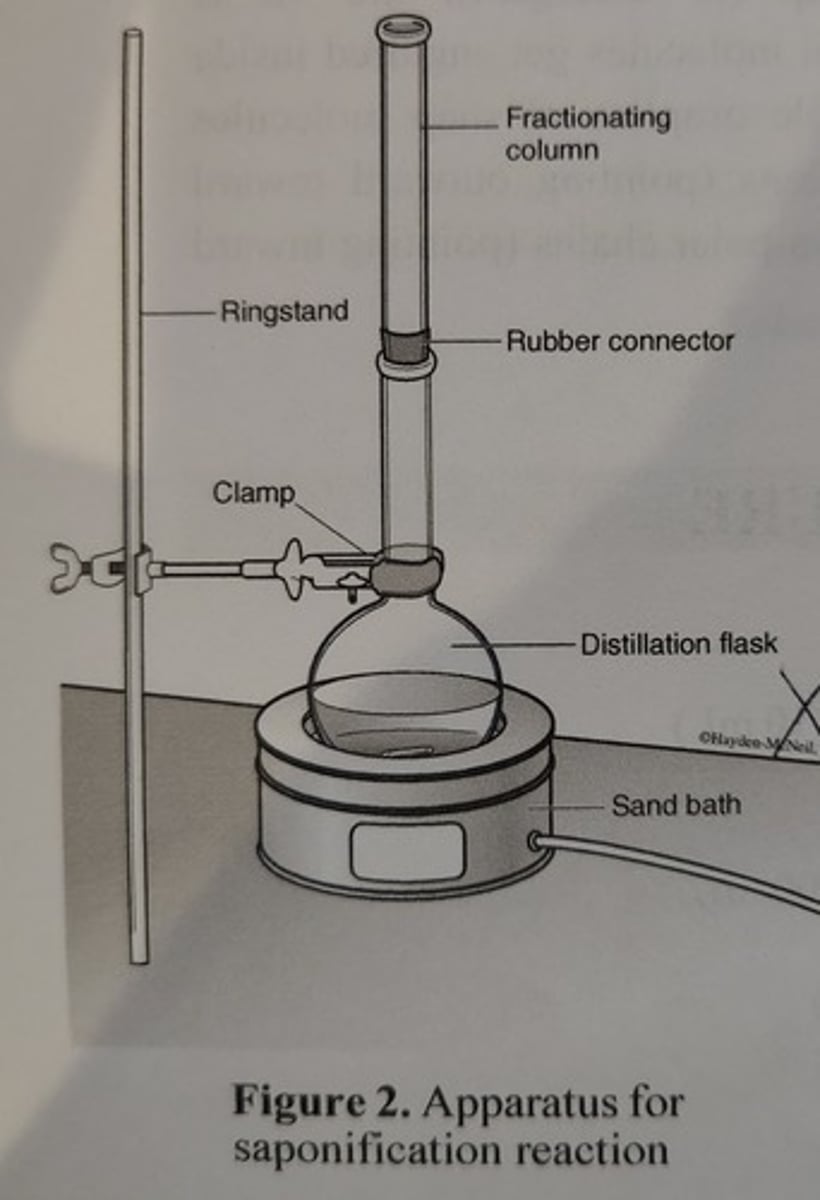
Apparatus for Saponification Reaction
Write a balanced chemical equation for the Fischer esterification reaction between i-pentyl alcohol and acetic acid.

Calculate the volume (mL) of water formed from full esterification of 1 g of acetic acid (in the above reaction) assuming the reaction is 100% complete.
MM of CH3COOH: 60.052 g/mol
MM of H2O: 18 g/mol
1 g acetic acid / 60 g acetic acid = 0.017 mol acetic acid
0.017 mol H2O x 18 g H2O = 0.3 g H2O
Ans: 0.3 g H20 = 0.3 mL H2O
Explain how the IR spectra can distinguish between i-pentyl alcohol and acetic acid.
The distinct functional groups (OH and CH respectively) in the two compounds can be distinguished by stretches and peaks in the 3200-3600 cm^-1 and 1050-1100 cm^-1 regions respectively.
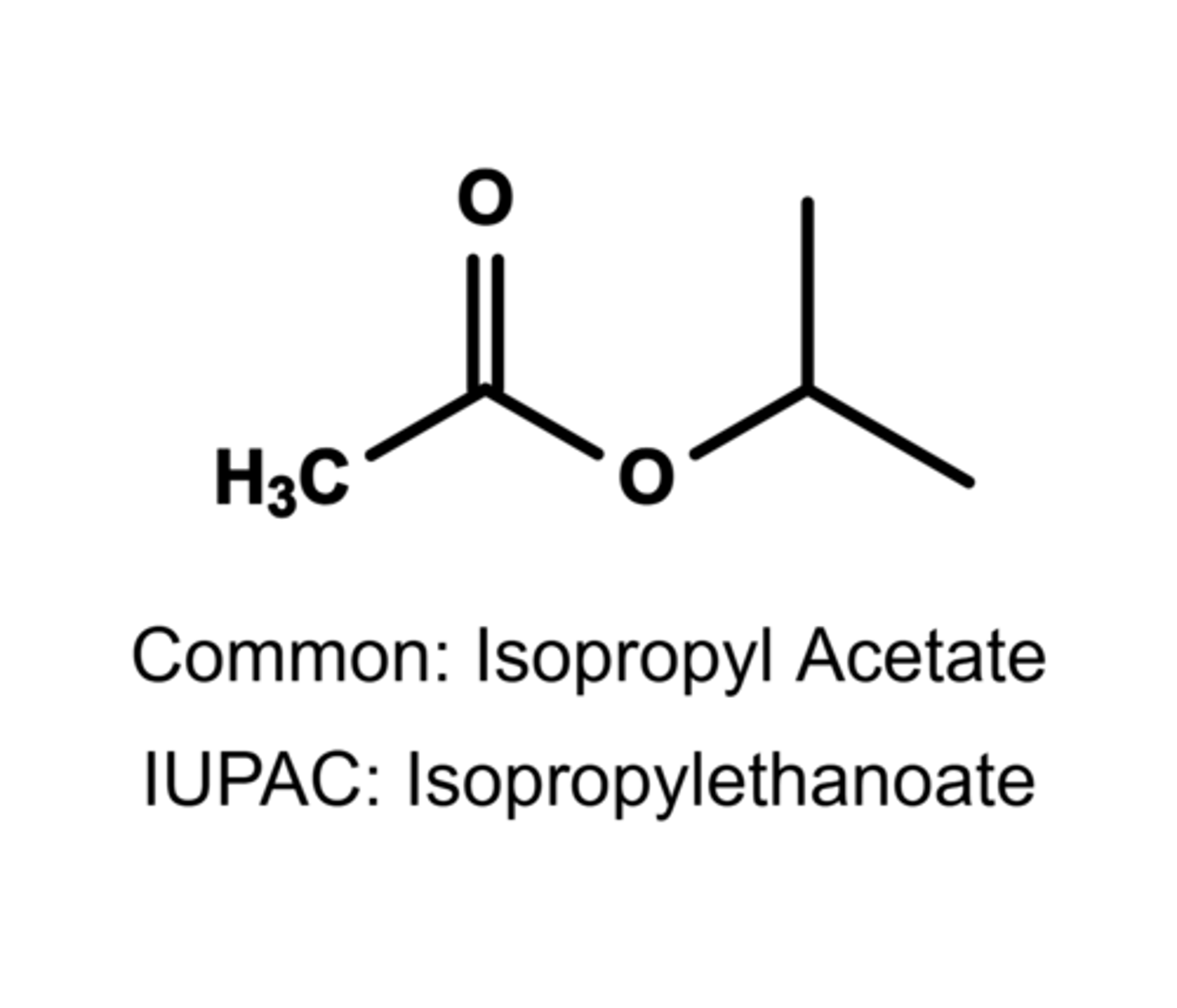
(a) Draw the structure of the ester that can be prepared from 2-propanol and acetic acid.
(b) Give both the common and IUPAC names of the ester product.
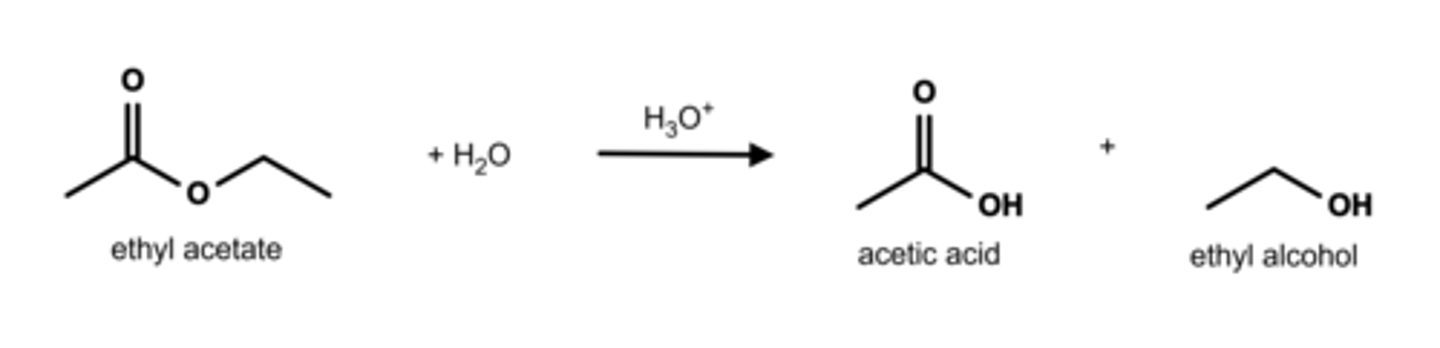
Esters can undergo hydrolysis under acidic conditions to form carboxylic acids and alcohols. Write a chemical equation for the hydrolysis of ethyl acetate.
Explain the purpose of washing the organic layer containing the ester with 10% aqueous NaHCO3.
It removes acidic impurities by converting them into water soluble salts.
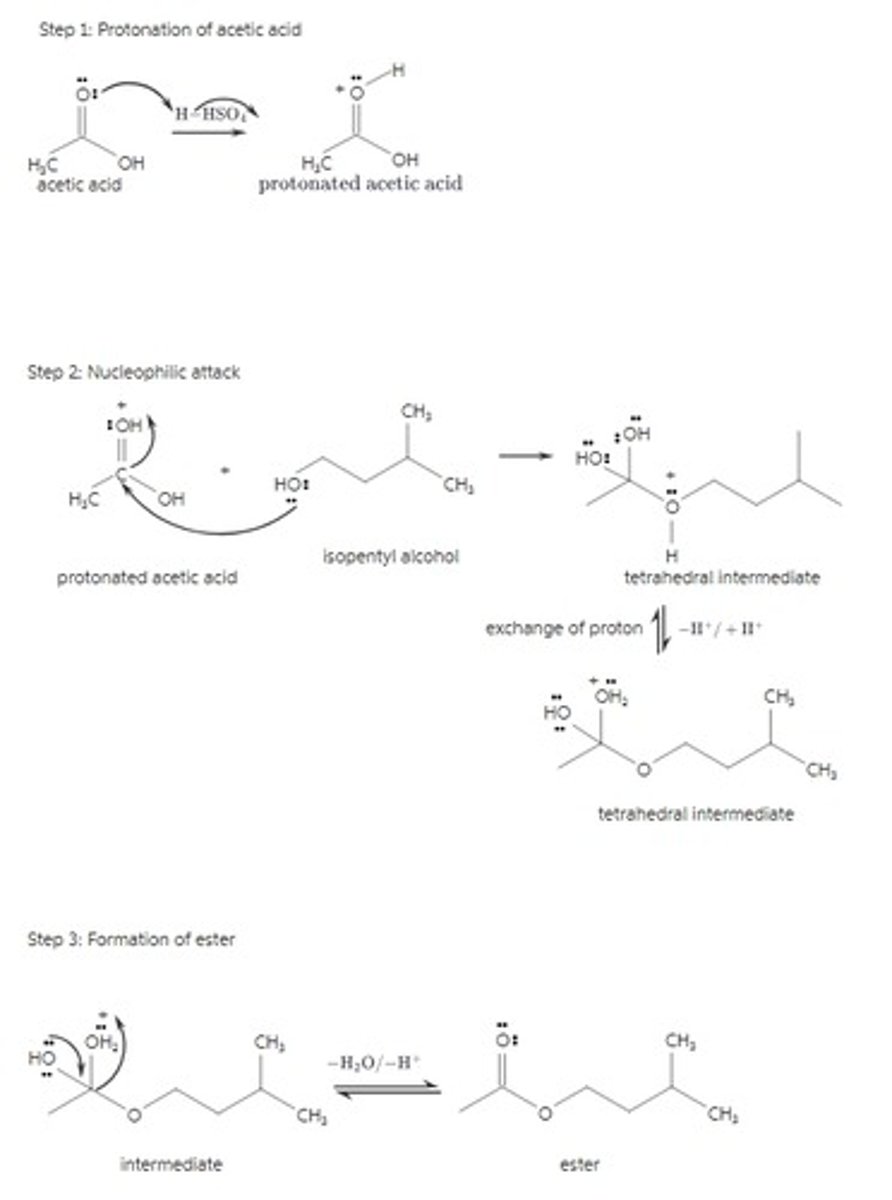
Write a detailed mechanism of the acid-catalyzed esterification reaction between i-pentyl alcohol & acetic acid.
(Sourced from Chegg)
Give the IUPAC name i-pentyl acetate.
3-methylbutyl ethanoate
Explain the health hazards of ingesting industrial ethanol.
Nausea, dizziness, or death due to severe depression of the CNS.
Draw the structure of the triglyceride that can be formed from stearic acid CH3(CH2)16COOH.

Give two examples of synthetic detergents. Give the name of the active surfactant in each.
1. Gain Laundry Detergent: Linear alkylbenzene sulfonate.
2. Tide Laundry Detergent: Sodium laureth sulfate.
Define each of the following:
(a) Soap
(b) Micelle
(a) Sodium or potassium salt of fatty acids formed by the reaction of fats and oils with an alkali.
(b) Aggregation of 8 to 10 molecules which contain a hydrophobic core and hydrophilic outer layer that traps dirt and grime.
Explain why sodium salts of fatty acids, although they are salts, are not very soluble in water.
The long hydrocarbon chains are hydrophobic and resist interaction with polar water molecules.
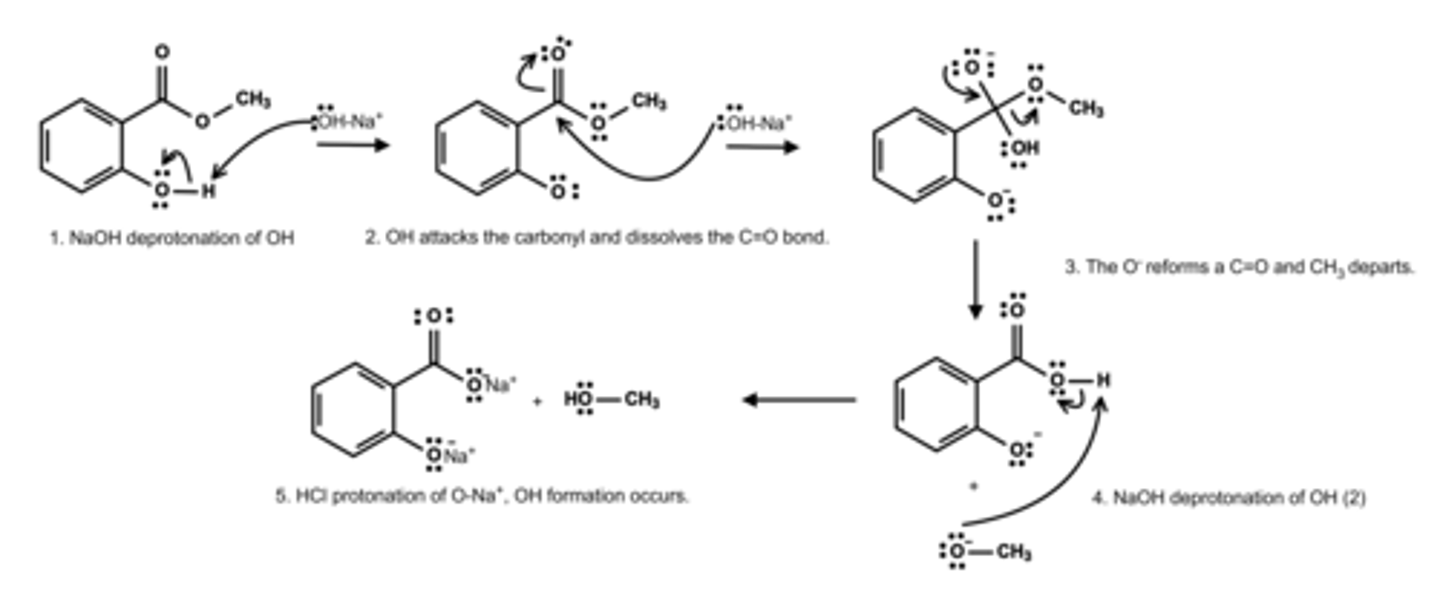
Write a mechanism for alkaline hydrolysis of methyl salicylate.
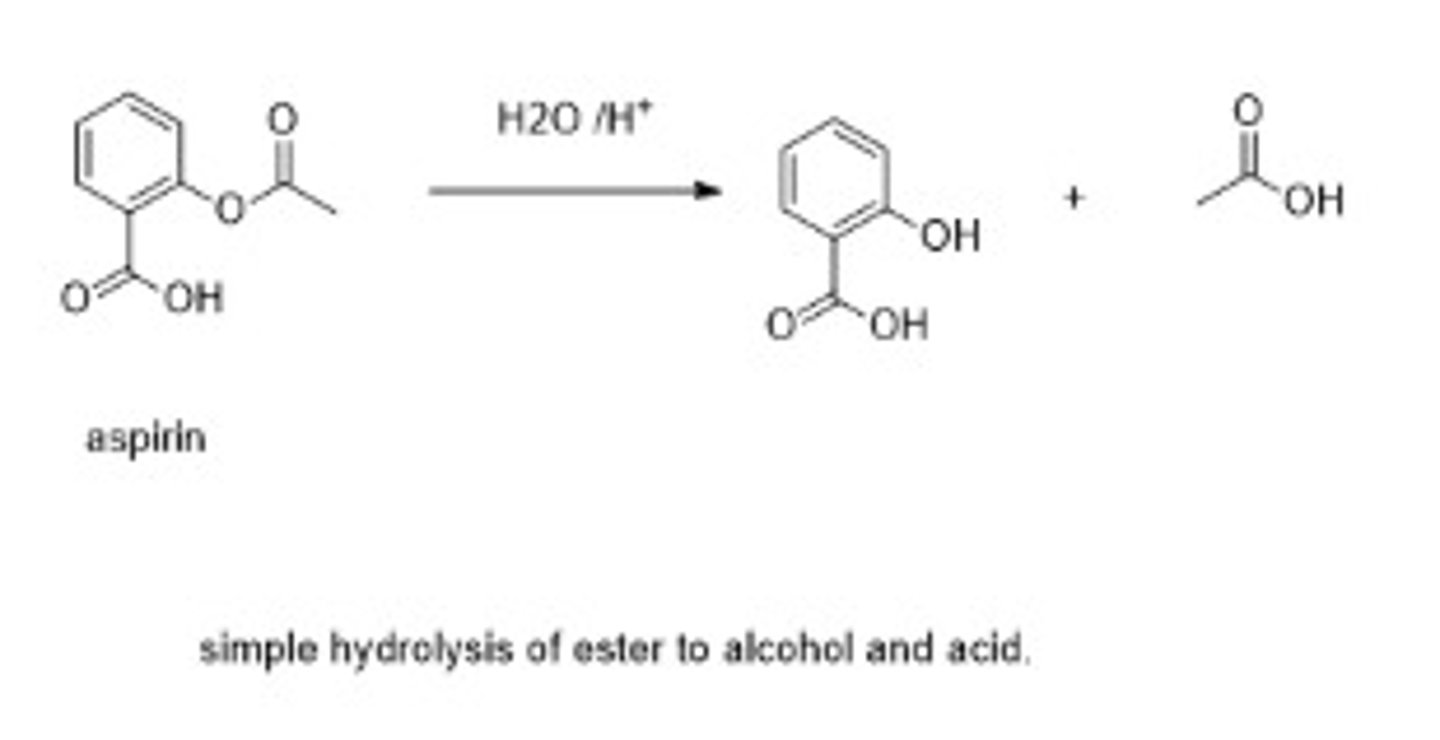
Write the chemical equation for acidic hydrolysis of aspirin.
(Sourced from Chegg)
What is the purpose of washing the soap product with cold water?
Removes NaOH + Salts and prevents soap from dissolving while doing so.
Why do magnesium and calcium salts diminish the cleansing effectiveness of soap?
They react to form INSOLUBLE salts or "scum" which is incapable of forming a soap lather.
Explain why hard water does not affect the cleansing effectiveness of synthetic detergents.
Detergents form SOLUBLE salts and do not precipitate out (less reactive with hard water).
Draw the structure of the aldol condensation reaction between cyclopentanone and two equivalents of benzaldehyde.
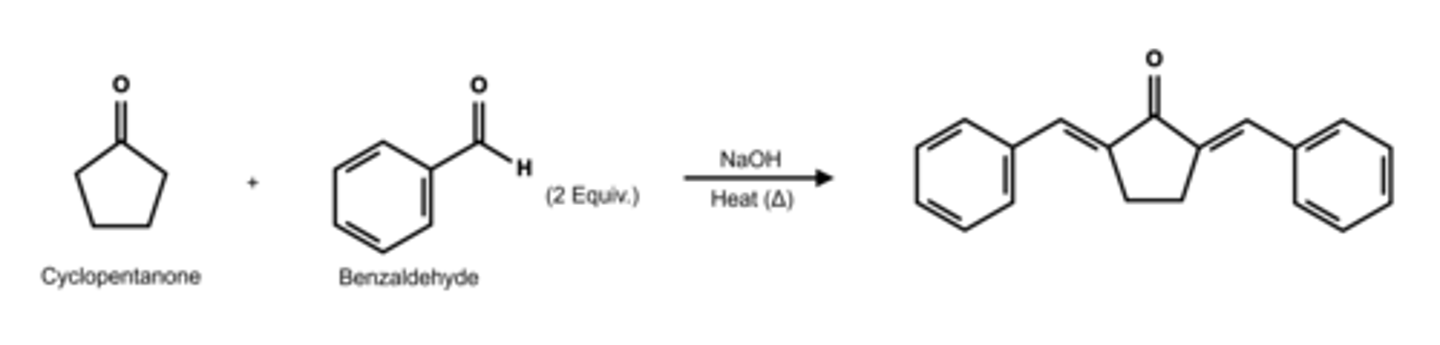
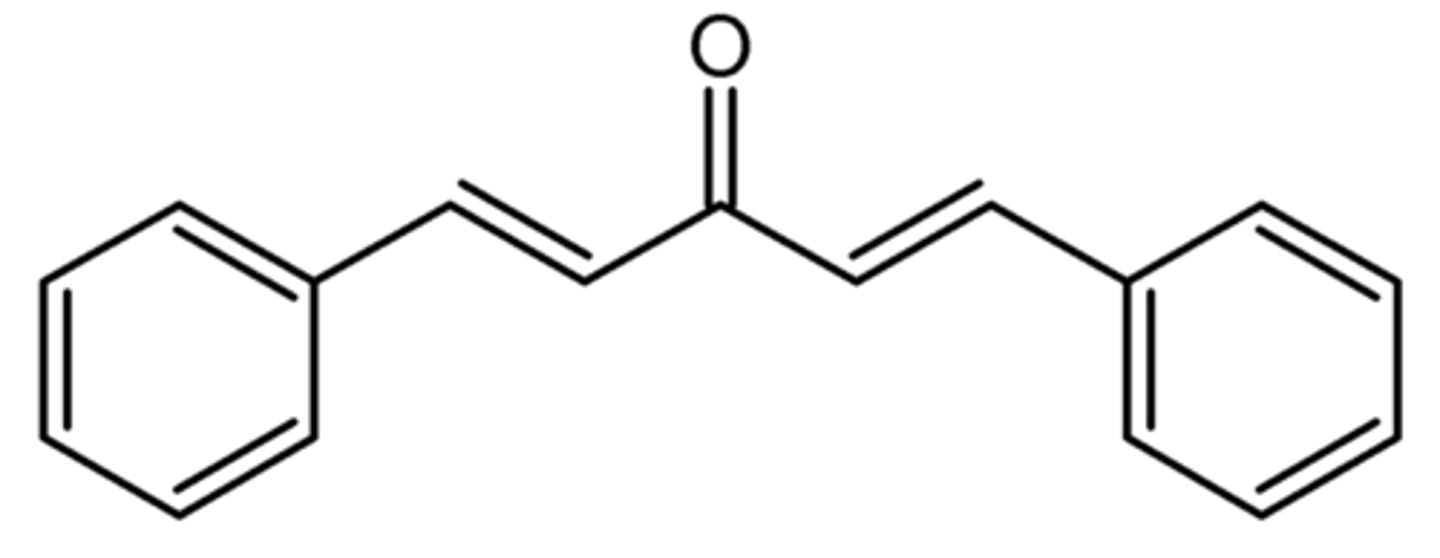
Draw the structure of dibenzalacetone.
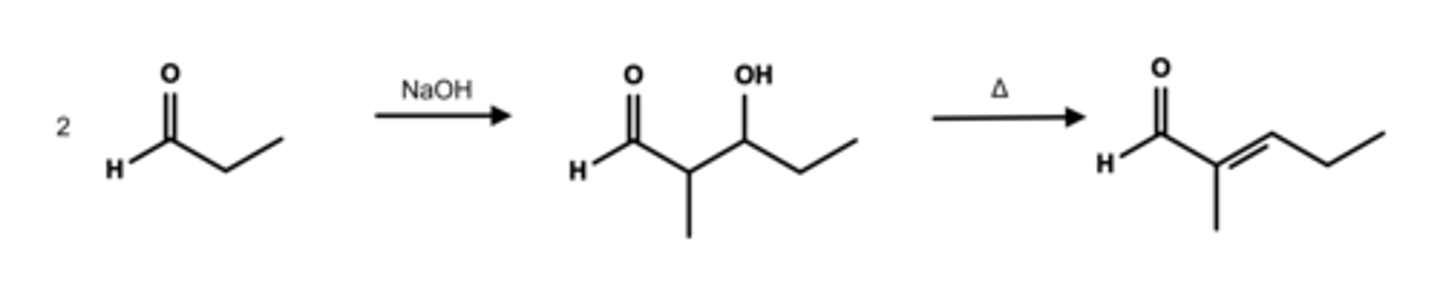
Write a complete chemical equation of base-catalyzed aldol condensation involving two molecules of propanol.
Define:
(a) Enolate Ion
(b) Aldol Molecule
(a) A carbanion with a negative charged placed on the oxygen.
(b) A compound containing both an aldehyde (or ketone) and an -OH group through aldol condensation.
Explain why dibenzalacetone is used as a sunscreen.
The highly conjugated structure and delocalized pi orbitals allow for absorption of UV rays.
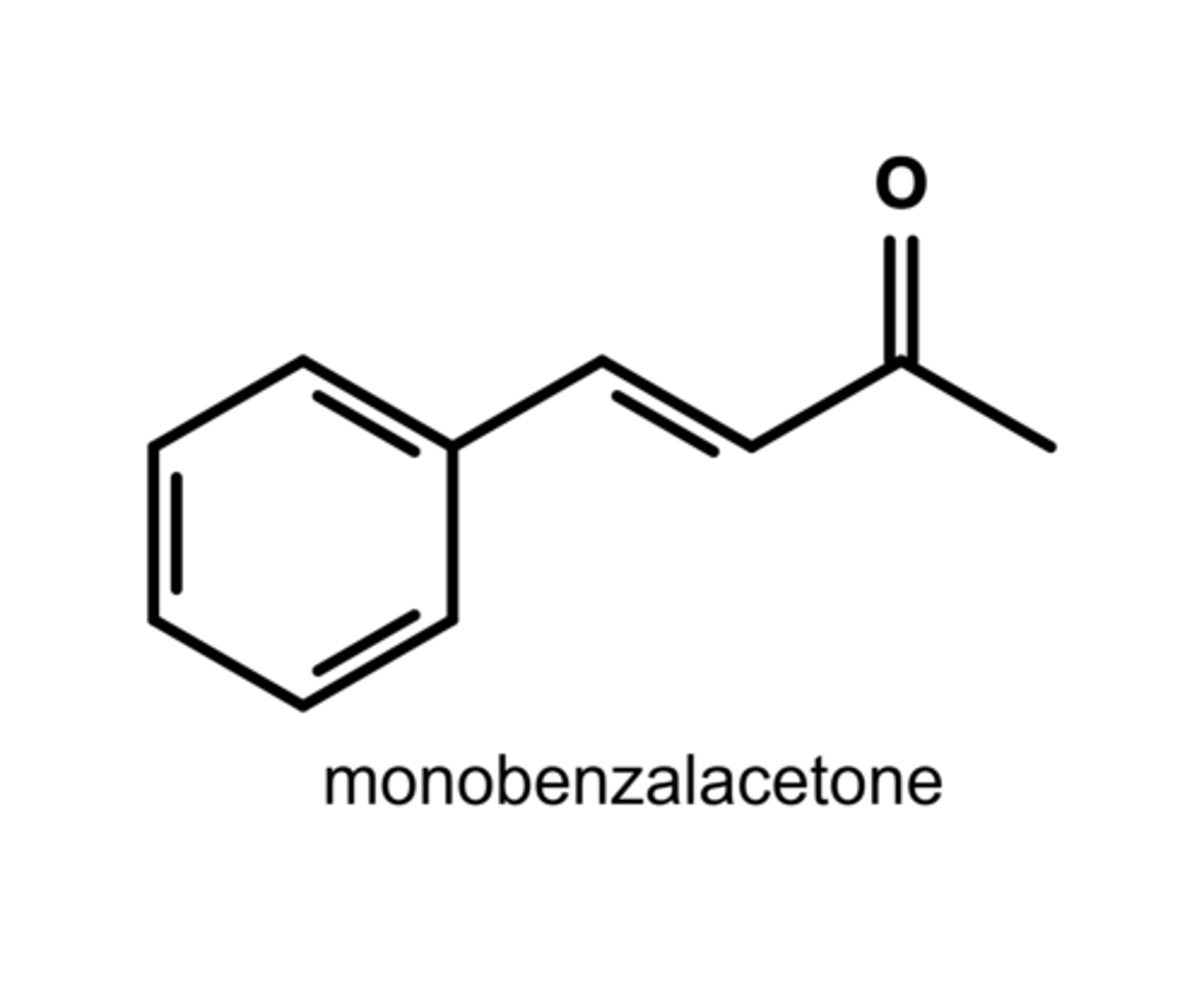
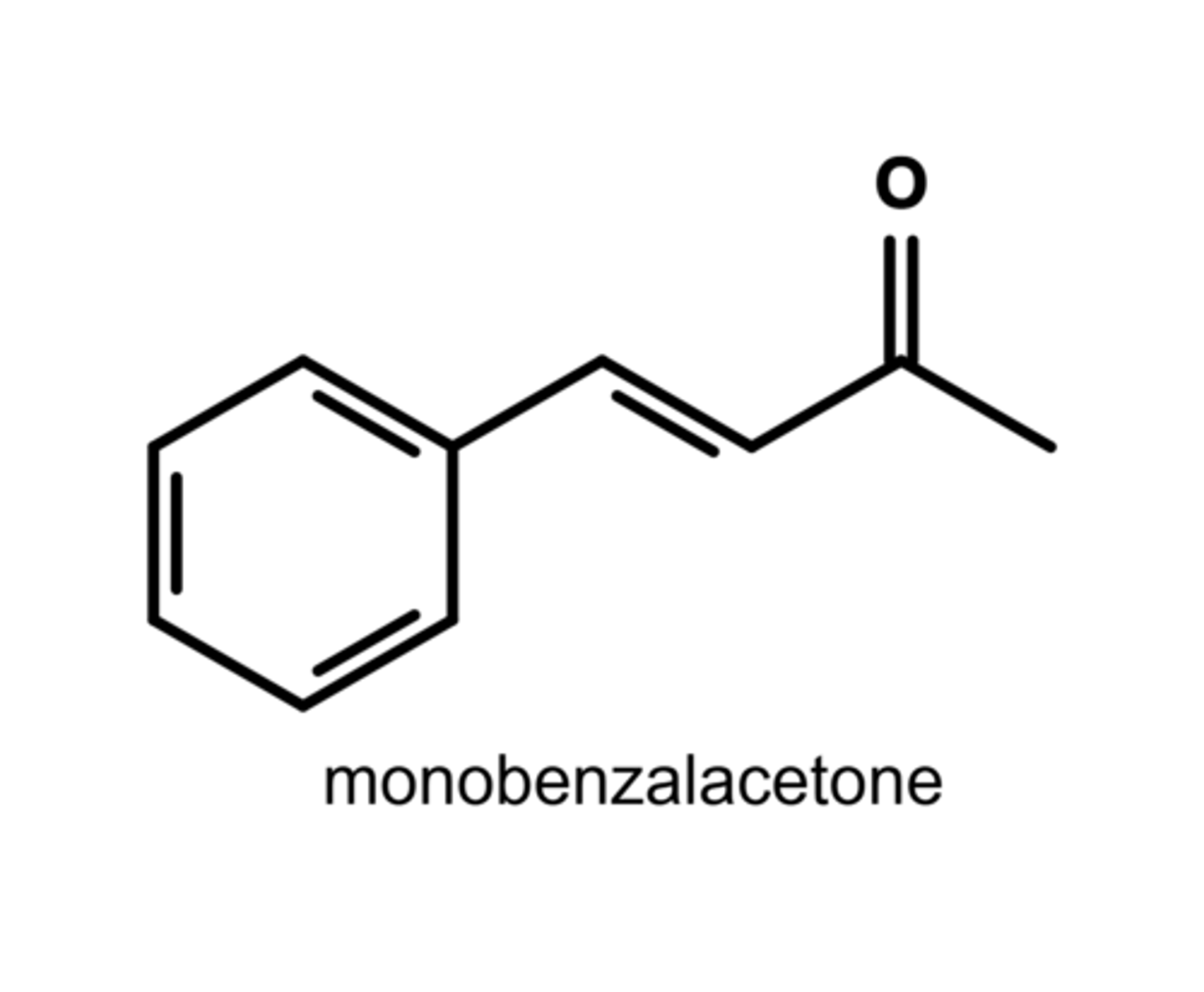
Write the structure of monobenzalacetone.
Explain with a chemical equation the role of NaOH in the synthesis of dibenzalacetone.
(Sourced from Chegg)
What is the purpose of washing the product with cold water?
Removes NaOH and ethanol impurities without dissolving the product itself.
Write the structure of one possible organic side-product during dibenzalacetone synthesis.
Monobenzalacetone is formed due t6o cross-aldol condensation.
(a) What is the safety hazard of each of concentrated sulfuric acid and nitric acids?
(b) Which acid is stronger, sulfuric or nitric? Explain.
(a) Corrosive, eye and airway irritation, skin and clothing burns.
(b) Sulfuric acid is stronger as it has a lower pKa (more willing to donate protons).
Explain with a chemical equation why methyl benzoate dissolves in concentrated H2SO4, but not in water?
Water is too weak to protonate.
"Protonation of carbonyl oxygen of methyl benzoate by concentrated H2SO4 makes particle more polar, permitting it to break down in polar sulfuric corrosive." (Sourced from Chegg)
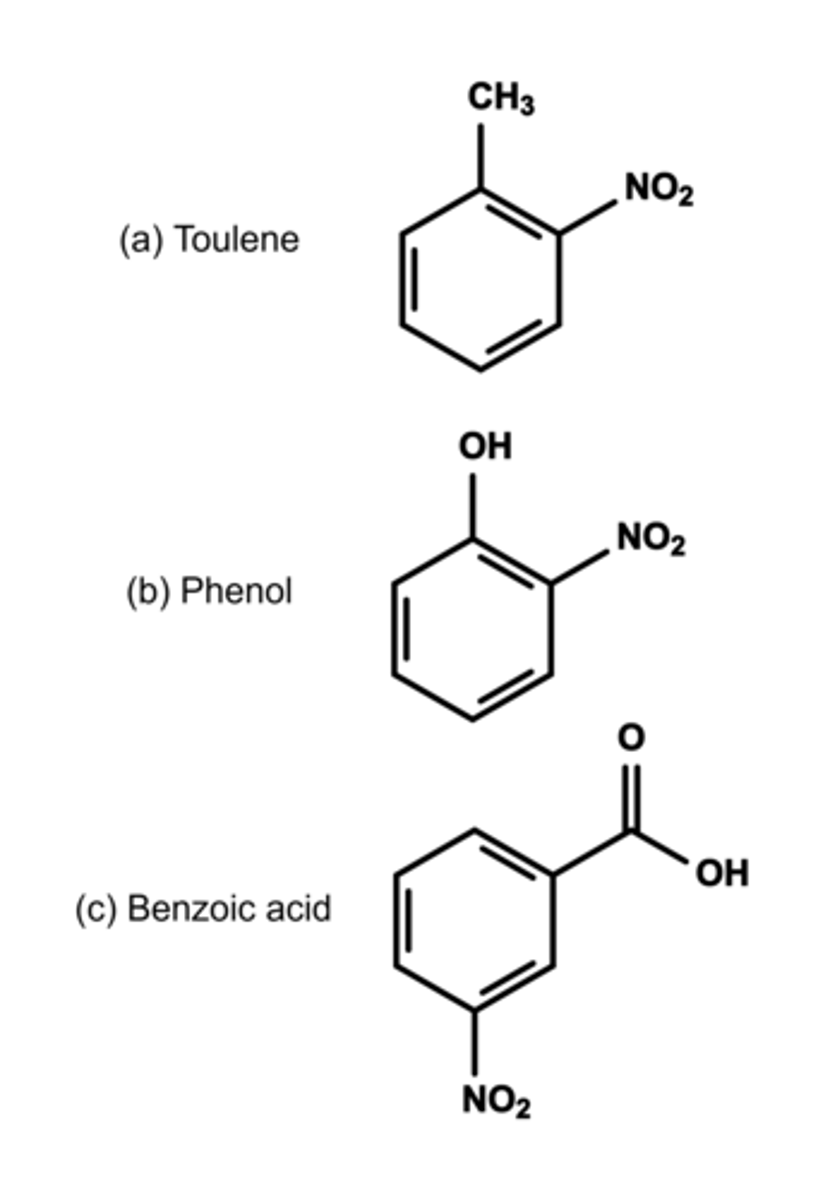
Draw the structure of the major organic product of nitration of the following:
(a) Toulene
(b) Phenol
(c) Benzoic acid
Label each of the following groups as activating or deactivating in electrophilic aromatic substitution (EAS).
(a) -CH3
(b) -NO2
(c) -COOH
(d) -OH
(a) Activating (e- donor)
(b) Deactivating (e- withdrawing)
(c) Deactivating (e- withdrawing)
(d) Activating (e- donor)
Explain why it is important to conduct the nitration of methyl benzoate at low temperature.
To lessen the chance of a dinitration reaction occurring.
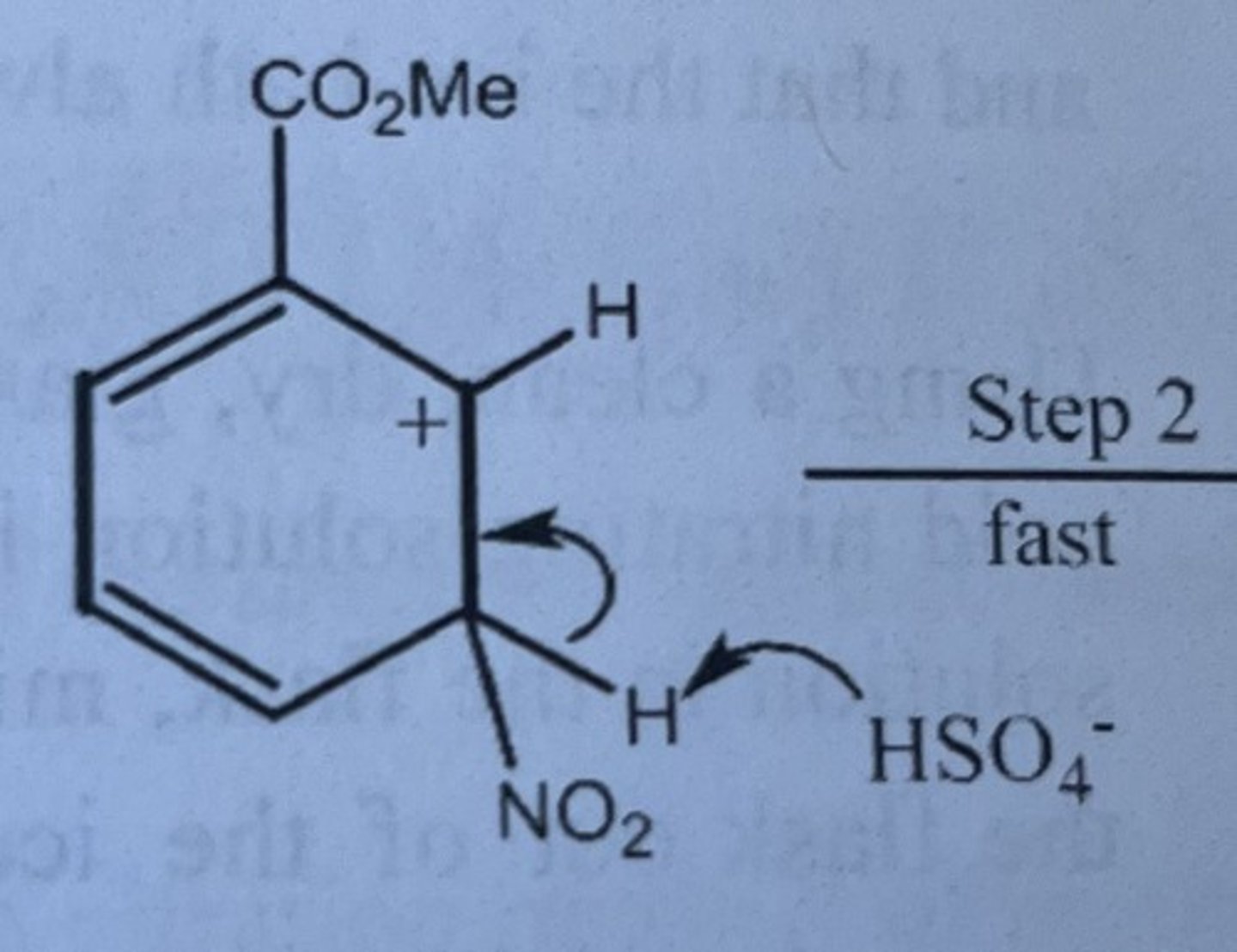
Draw the structure of the arenium ion intermediate involved in the nitration of methyl benzoate.
Draw the structure of the nitration product of methyl benzoate.
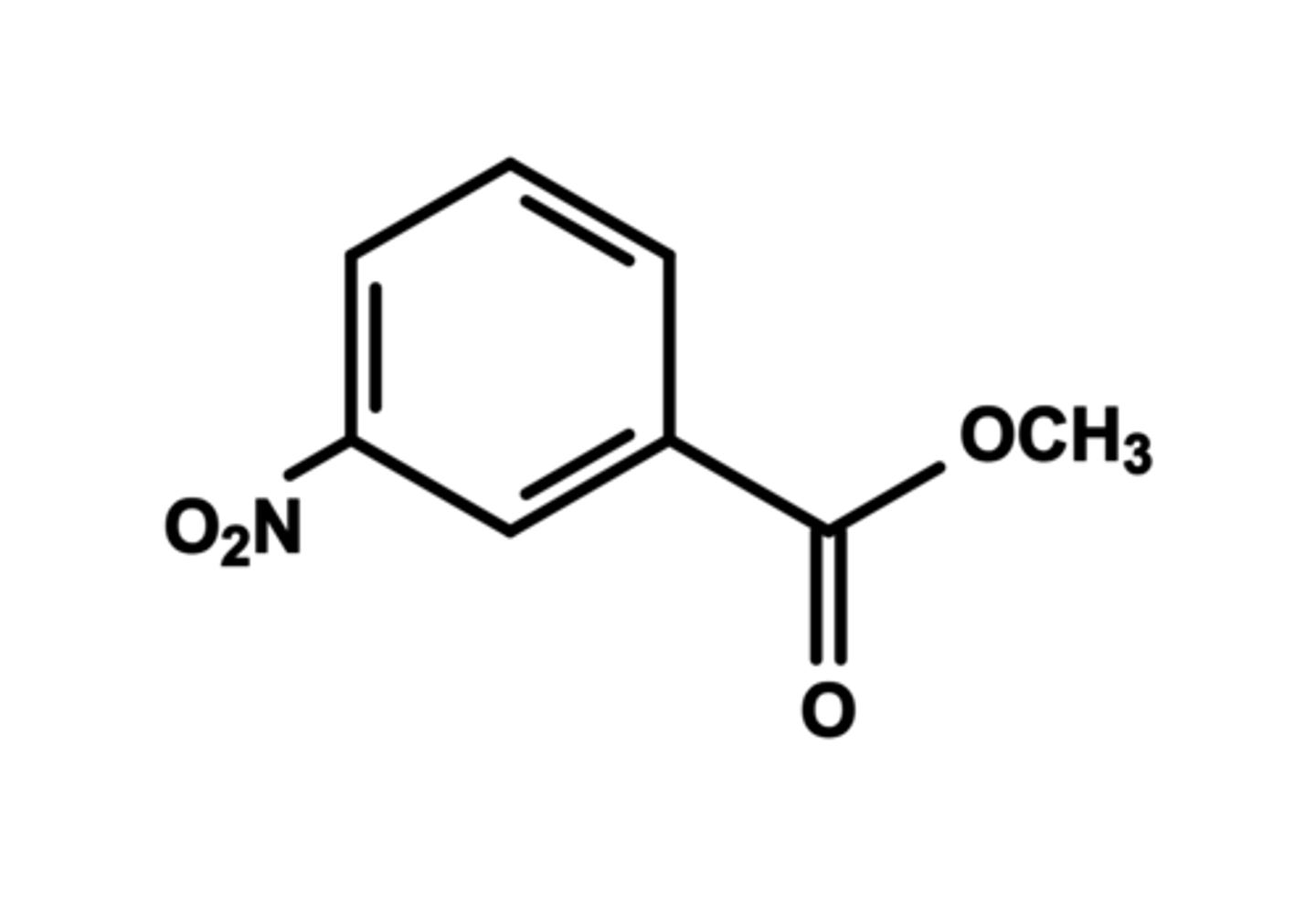
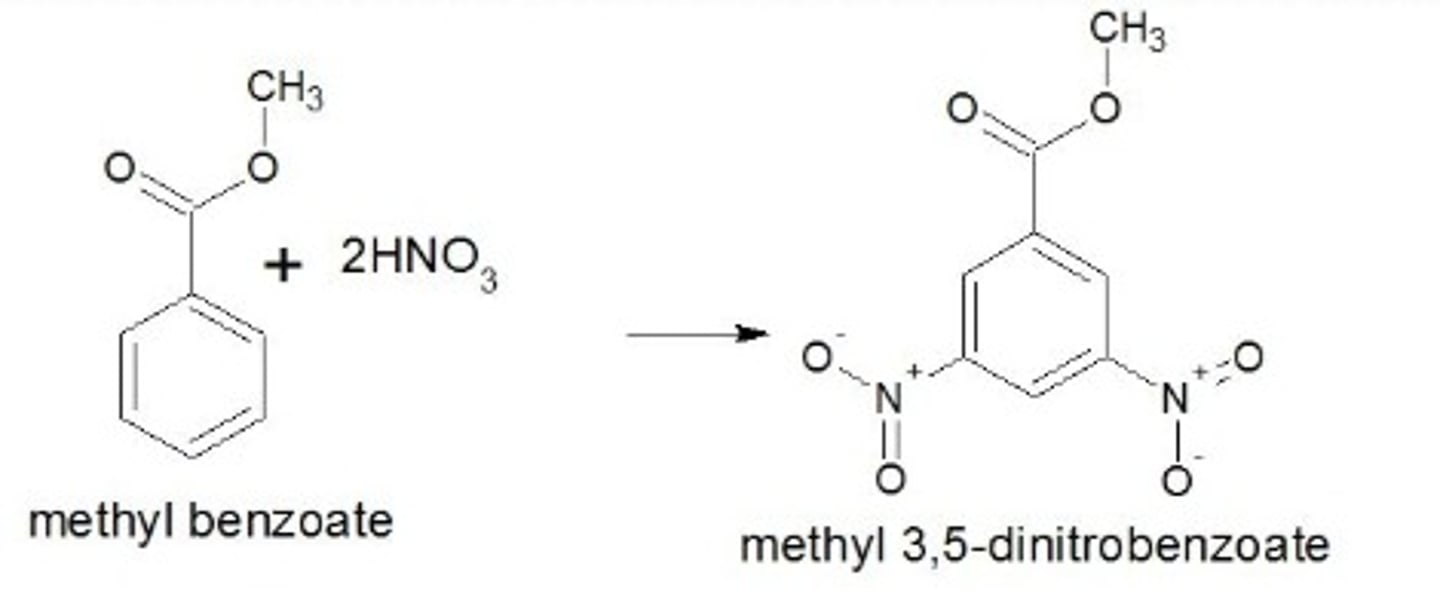
Draw the structure of methyl 3,5-dinitrobenzoate.
(Sourced from Chegg)
Which of the following aromatic compounds undergoes nitration faster than benzene does? Explain.
(a) Ph-CH3
(b) Ph-Cl
(c) Ph-OH
(d) Ph-Br
(e) Ph-NO2
(f) Ph-Ph
(a) Ph-CH3, (c) Ph-OH, (f) Ph-Ph
Explanation: Electron rich substituents allow for easier donation.
In the synthesis of explosive trinitrotoluene (TNT) toluene is nitrated three times. Explain why the first nitration step proceeds much faster than the second & the third nitration steps.
"The first nitration of toluene is faster because the methyl group activates the benzene ring, enhancing its reactivity towards nitration. However, once a nitro group is added, it deactivates the ring, making subsequent nitration steps slower." (Sourced from Chegg)