4. metal alloys and casting
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
alloy
mixture of 2+ metals or metalloids that are mutually soluble in the molten state
mixture of different elements mixed together
Alloying elements are added to alter the (BLANK x3) of a metallic element, thus obtaining properties not found in a pure metal
hardness, strength, and toughness
may be classified on basis of their metal composition as what three categories?
base metal alloy, noble metal alloy, and high noble metal alloys
what are the general requirements of a dental alloy? (5)
Not produce harmful toxicologic or allergic effects
Chemical properties should provide resistance to corrosion and physical changes when in the oral cavity
Physical and mechanical properties (thermal conductivity, melting temperature, coefficient of thermal expansion, and strength) should meet certain minimum values*
The technical expertise needed for fabrication and use should be feasible for the average dentist and skilled technician
The metals, alloys, and companion materials should be plentiful, relatively inexpensive, and readily available
dental metallic material ISO 22674 classification
0–5 based on mechanical properties like strength (proof stress) and elongation. Its purpose is to ensure, through standardized requirements, that alloys have suitable, reliable strength, corrosion resistance, and ductility for specific clinical uses, such as inlays, crowns, or dentures
which type of dental metallic material ISO 22674 classification?
intended use: low stress, single-tooth fixed restorations (small)
example: small one-surface inlay
minimal proof strength w 0.2% non-proportional extension (MPa): N/A
minimum percent elongation after fracture: N/A
minimum young’s modulus (GPa): N/A
type 0
which type of dental metallic material ISO 22674 classification?
intended use: low stress, single-tooth fixed restorations
example: crown w minimal occlusal force
minimal proof strength w 0.2% non-proportional extension (MPa): 80
minimum percent elongation after fracture: 18%
minimum young’s modulus (GPa): N/A
type 1
which type of dental metallic material ISO 22674 classification?
intended use: single tooth, fixed restorations
example: crown w normal occlusal force
minimal proof strength w 0.2% non-proportional extension (MPa): 180
minimum percent elongation after fracture: 10%
minimum young’s modulus (GPa): N/A
type 2
which type of dental metallic material ISO 22674 classification?
intended use: multiple tooth, fixed restorations
example: fixed partial denture (bridge)
minimal proof strength w 0.2% non-proportional extension (MPa): 270
minimum percent elongation after fracture: 5%
minimum young’s modulus (GPa): N/A
type 3
which type of dental metallic material ISO 22674 classification?
intended use: thin sections under high force
example: precision attachment
minimal proof strength w 0.2% non-proportional extension (MPa): 360
minimum percent elongation after fracture: 2
minimum young’s modulus (GPa): N/A
type 4
which type of dental metallic material ISO 22674 classification?
intended use: high stiffness and high stress
example: removable partial denture clasp
minimal proof strength w 0.2% non-proportional extension (MPa): 500
minimum percent elongation after fracture: 2%
minimum young’s modulus (GPa): 150
type 5
what are the noble metals? (8 of relevance)
gold, silver, platinum, palladium, rhodium, ruthenium, iridium, and osmium
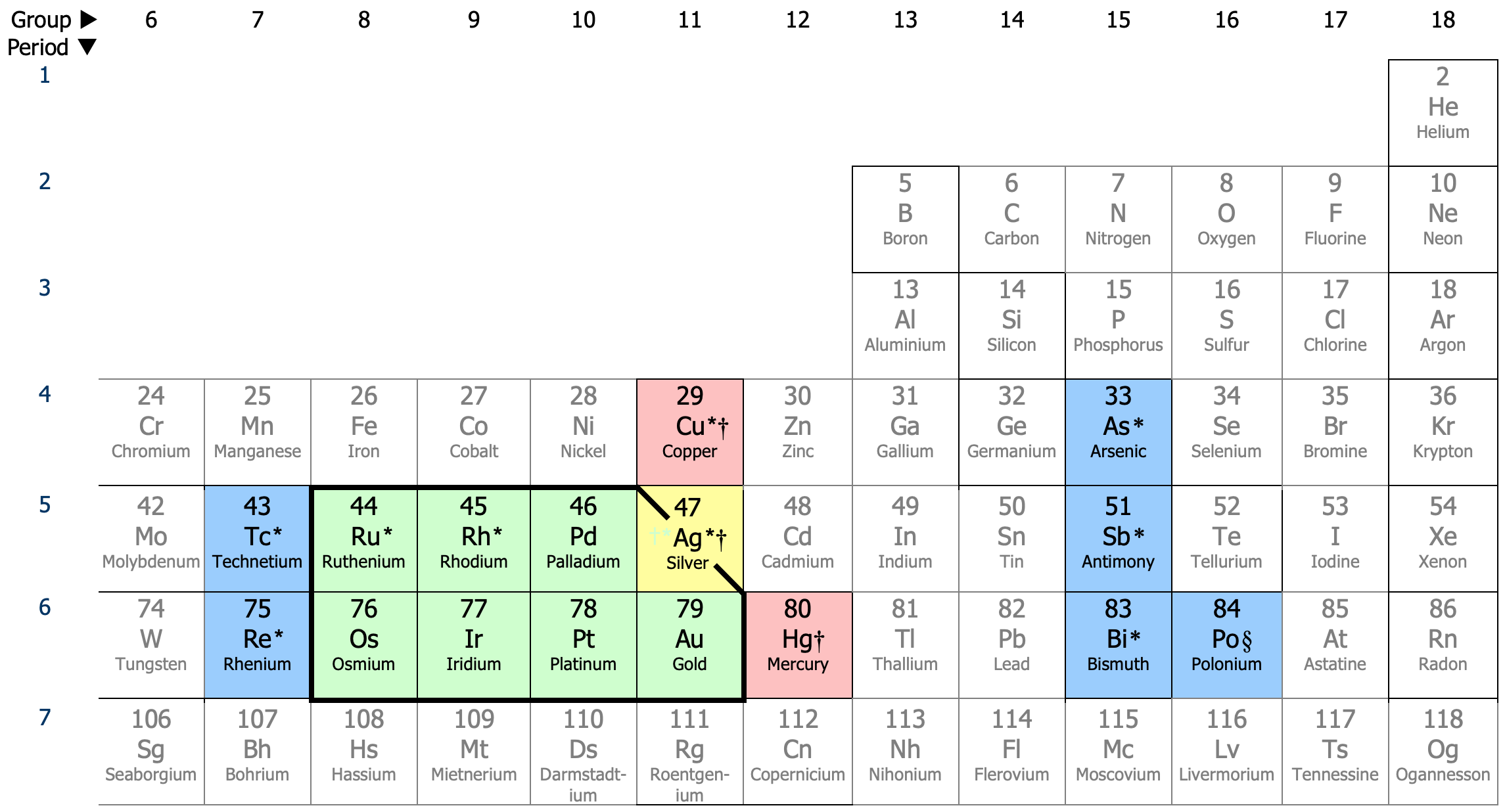
what are the base metals? (13
definition: common and inexpensive metal, as opposed to a precious metal such as gold or silver
nickel, cobalt, chromium, titanium, iron, and copper
also: carbon, silicon, aluminum, gallium, ruthenium, manganese, molybdenum, tungsten, tantalum
noble metals are defined on the basis of their
chemical properties
precious metals are defined based on
cost
(?) was originally used for noble metal alloys containing significant amounts of silver
semiprecious
(?) alloys are composed of (?) ingredients, with most being based on a combo of nickel and chromium
non-precious
which class has this requirement: total noble content ≥60% where ≥40% is gold (IMPORTANT)
high noble alloys
which class has this requirement: titanium content ≥85%
titanium and titanium alloys
which class has this requirement: total noble content ≥25%
noble alloys
which class has this requirement: ≥25%
predominantly base metals
what are five important considerations when choosing a cast alloy? (important to me)
noble metal content → corrosion resistance, internal properties, tarnish resistance, biocompatibility, and classification
hardness → wear resistance, surface
yield strength → load; maximum stress a material can withstand before undergoing permanent deformation
elongation → margin
fusion temperature → soldering
what three properties are important to consider when choosing alloys for PFM restorations?
Thermal expansion, bond strength, and composition
T or F: there should be a slight mismatch between thermal expansion of porcelain and metal (porcelain being slightly lower)
true, this is the porcelain-metal compatibility property of thermal expansion
refers to the Coefficient of Thermal Expansion (CTE) mismatch between porcelain (ceramic) and metal in applications like Porcelain-Fused-to-Metal (PFM) dental restorations or brazed assemblies. To achieve a strong bond, the metal must have a slightly higher CTE than the porcelain, forcing the porcelain into permanent compression as it cools
Because the metal shrinks more during cooling, it squeezes the porcelain, successfully stabilizing the restoration and preventing fractures under pressure
what does porcelain-to-metal bond strength measure?
ensures retention of porcelain to metal in both the oral environment and during thermal processing
composition is important bc some components can affect the (?) of porcelain
color
why is yield strength crucial?
bc of small cross section
what controls the minimum allowable dimensions of critical areas?
stress
what affects the flexibility of a metal framework?
elastic modulus
what affects tarnish resistance, corrosion resistance, and thermal stability?
chemical stability
ANSI/ADA Specification No. 5 for Dental Casting Alloys (!)
stablishes standardized requirements, classification (Types I-IV), and testing methods for noble metal alloys—primarily gold—used in dentistry. It ensures the quality, biocompatibility, composition, and physical properties (hardness, yield strength) of materials used for crowns, bridges, and inlays.
ANSI/ADA Specification No. 5 for Dental Casting Alloys: which class? (!)
85% 20K
description: soft
usage: inlays: low stress applications
yield strength: <140 MPa
elongation (annealed): 18%
vickers hardness: 60-90 VHN
type I
ANSI/ADA Specification No. 5 for Dental Casting Alloys: which class? (!)
75% 18K
description: medium
usage: inlays and onlays: increased stress applications including cusp replacement
yield strength: 140-240 MPa
elongation (annealed): 18%
vickers hardness: 90-120 VHN
type II
ANSI/ADA Specification No. 5 for Dental Casting Alloys: which class? (!)
70% 17K
description: hard
usage: crowns and short-span bridge: high stress applications
yield strength: 201-340 MPa
elongation (annealed): 12%
vickers hardness: 120-150 VHN
type III
ANSI/ADA Specification No. 5 for Dental Casting Alloys: which class? (!)
65% 15K
description: extra hard
usage: long-span bridges, removable partial dentures; high stress, high flexural resistance
yield strength: >340 MPa
elongation (annealed): 10%
vickers hardness: <150 VHN
type IV
what is density effect of alloy elements in dental gold alloys?
how adding metals like copper, silver, or palladium alters the high density of pure gold (specific gravity 19.3 g/cm³) to achieve desired physical and casting properties. Generally, adding base metals or lower-density precious metals reduces the overall density of the alloy, which impacts casting, strength, and tarnish resistance
Specific gravity: 19.32
Melting point: 1063°C (1945°F)
Atomic diameter: 2.88 Å
Space lattice: Face-centered cubic
Chemical activity: Inert
Color: yellow
Approximate content: 50–95%
Density effect: Increases markedly
Effect on color: Lends yellow color
Effect on melting: Raises melting point mildly
Tarnish resistance: Essential to good tarnish resistance
Heat hardening: Contributes importantly with Cu
Gas absorption: —
Castability: —
gold
Specific gravity: 21.45
Melting point: 1769°C (3224°F)
Atomic diameter: 2.77 Å
Space lattice: Face-centered cubic
Chemical activity: Inert
Color: white
Approximate content: 0–20%
Density effect: Increases markedly
Effect on color: Whitens slowly; ~12% required; not pure white
Effect on melting: Raises melting point fairly rapidly
Tarnish resistance: Contributes importantly
Heat hardening: Increases with Cu
Gas absorption: —
Castability: —
platinum
Specific gravity: 12.0
Melting point: 1552°C (2829°F)
Atomic diameter: 2.74 Å
Space lattice: Face-centered cubic
Chemical activity: Mild
Color: white
Approximate content: 0–12%
Density effect: Lowers slightly
Effect on color: Whitens rapidly; as little as 5%
Effect on melting: Raises melting point rapidly
Tarnish resistance: Increases but less than Au/Pt
Heat hardening: Some increase with Cu
Gas absorption: High for hydrogen
Castability: Effects not critical
pallidum
Specific gravity: 8.96
Melting point: 1083°C (1981°F)
Atomic diameter: 2.55 Å
Space lattice: Face-centered cubic
Chemical activity: Very active
Color: red
Approximate content: 0–17%
Density effect: Lowers
Effect on color: Lends red color; dark plate high in Cu
Effect on melting: Lowers melting point (even below its own)
Tarnish resistance: Contributes to tarnish (flame/sulfur)
Heat hardening: Essential if alloy heat hardens
Gas absorption: —
Castability: Effects not critical
copper
Specific gravity: 10.49
Melting point: 961°C (1761°F)
Atomic diameter: 2.88 Å
Space lattice: Face-centered cubic
Chemical activity: Active
Color: white
Approximate content: 0–20%
Density effect: Lowers
Effect on color: Whitens slowly; counteracts Cu redness; green gold
Effect on melting: Slight effect; may raise or lower mildly
Tarnish resistance: Tarnishes with sulfur
Heat hardening: Increases with Cu
Gas absorption: High for oxygen
Castability: —
silver
Specific gravity: 7.31
Melting point: 420°C (787°F)
Atomic diameter: 2.66 Å
Space lattice: Close-packed hexagonal
Chemical activity: Very active
Color: white
Approximate content: 0–2%
Density effect: Lowers
Effect on color: Minimal (too low %)
Effect on melting: Lowers melting point rapidly (solders)
Tarnish resistance: Minimal effect in low %
Heat hardening: Slight with Cu
Gas absorption: Good deoxidizer
Castability: Decreases surface tension; increases fluidity
zinc
Specific gravity: 22.4
Melting point: 2443°C (4429°F)
Atomic diameter: 3.32 Å
Space lattice: Face-centered cubic
Chemical activity: Active
Color: white
Approximate content: 0.005–0.1%
Density effect: Increases slightly
Effect on
Effect on melting: No effect
Tarnish resistance: Increased
Heat hardening: No effect
Gas absorption: No effect
Castability: No effect
iridium

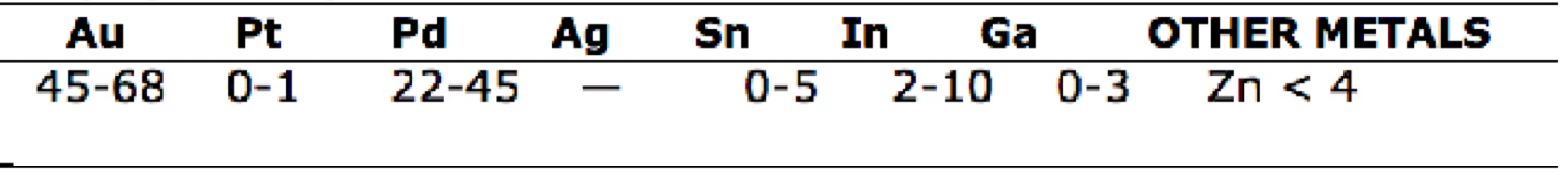
PFM compositions of
high gold
PFM composition of
gold-palladium (no silver)

PFM composition of
gold-palladium-silver

PFM composition of
palladium-copper
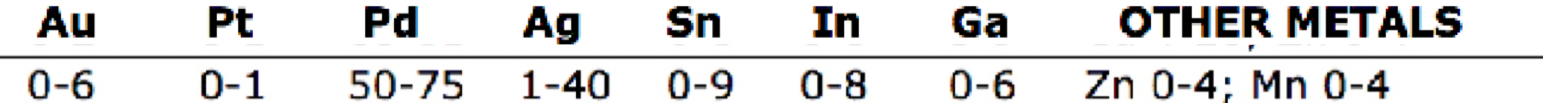
PFM composition of
palladium-silver
the concentrations of the following alloys:
nickel-chromium
nickel-chromium-beryllium
cobalt-chromium
titanium
titanium alloys

which alloy?
Gold content between 78% to 87% by weight
Total noble metal content about 97%
Light yellow in color
Excellent hardness, tensile strength, and corrosion resistance
high gold
what three things are added to high gold alloys for strength and to promote good porcelain bond to metal oxide?
tin, indium, iron
which alloy?
20% to 30% palladium content
10% to 15% silver content
Higher elastic modulus and less susceptibility of dimensional change during the porcelain baking when compared to high-gold alloys
Good clinical working characteristics and corrosion resistance
Silver can change the color of porcelain
gold-palladium-silver
which alloy:
50% to 60% palladium, with the most of the balance being silver
Physical and chemical properties are comparable to other noble metal alloys
Good corrosion and tarnish resistance
Can affect the color of porcelain
palladium-silver
which alloy?
50% gold
40% palladium
Favorable yield strength and hardness
Higher elastic modulus when compared to high-gold alloys
Excellent corrosion resistance
Thermal expansion is incompatible with some higher-expansion porcelains
gold-palladium alloys
which alloy?
70% to 80% palladium
Up to 15% by weight of copper
9% gallium
High hardness and low elastic modulus
Sag resistance is a contra-indication for large-span fixed partial dentures
palladium-copper
(blank) resistance is a contraindication for large-span fixed dentures
sag
preventing the material of the denture from warping under biting pressure, or preventing premature facial sagging (collapsing of the cheeks and lips) caused by jawbone deterioration
which alloy:
88% palladium
4% to 5% cobalt by weight
Have high coefficient of thermal expansion
Forms a dark oxide that may be difficult to cover
More susceptible to hot tearing and embrittlement from carbon when no silver is present
palladiumj-cobalt
palladium-cobalt alloys are (less/more) susceptible to hot tearing and embrittlement from carbon when there is NO silver present
more
base metal alloys are based mostly on what two metals?
nickel and chromium, with a few cobalt- chromium and iron-based alloys available
Corrosion resistance is based on chemical properties

can check for allergies, recalls. not necessarily for PFM but other materials even implants
what is this:
combinations of various types of waxes compounded to provide the desired physical properties. uses include patterns for inlays, crowns, pontics, partial and complete dentures, and for bite registrations
one of several esters of fatty acids with higher alcohols (usually monohydric alcohols)
classified according to their applications into pattern, processing, and impression waxes
dental waxes
what are the three applications of dental waxes?
pattern, processing, impression
what are the four types of pattern waxes?
(blue) inlay for crowns → type I and II
resin
casting
base plate
what are the six types of processing dental waxes?
boxing
sticky
carding
blockout
white
utility
what are the two impression dental waxes?
corrective and bite registration
define:
channel or hole through which plastic or metal is poured or cast into a gate or reservoir and them into a mold
forms a mount for the wax pattern and fix the pattern in space so a mold can be made
creates a channel for elimination of wax during burnout which allows for molten metal to enter such mold
spruing
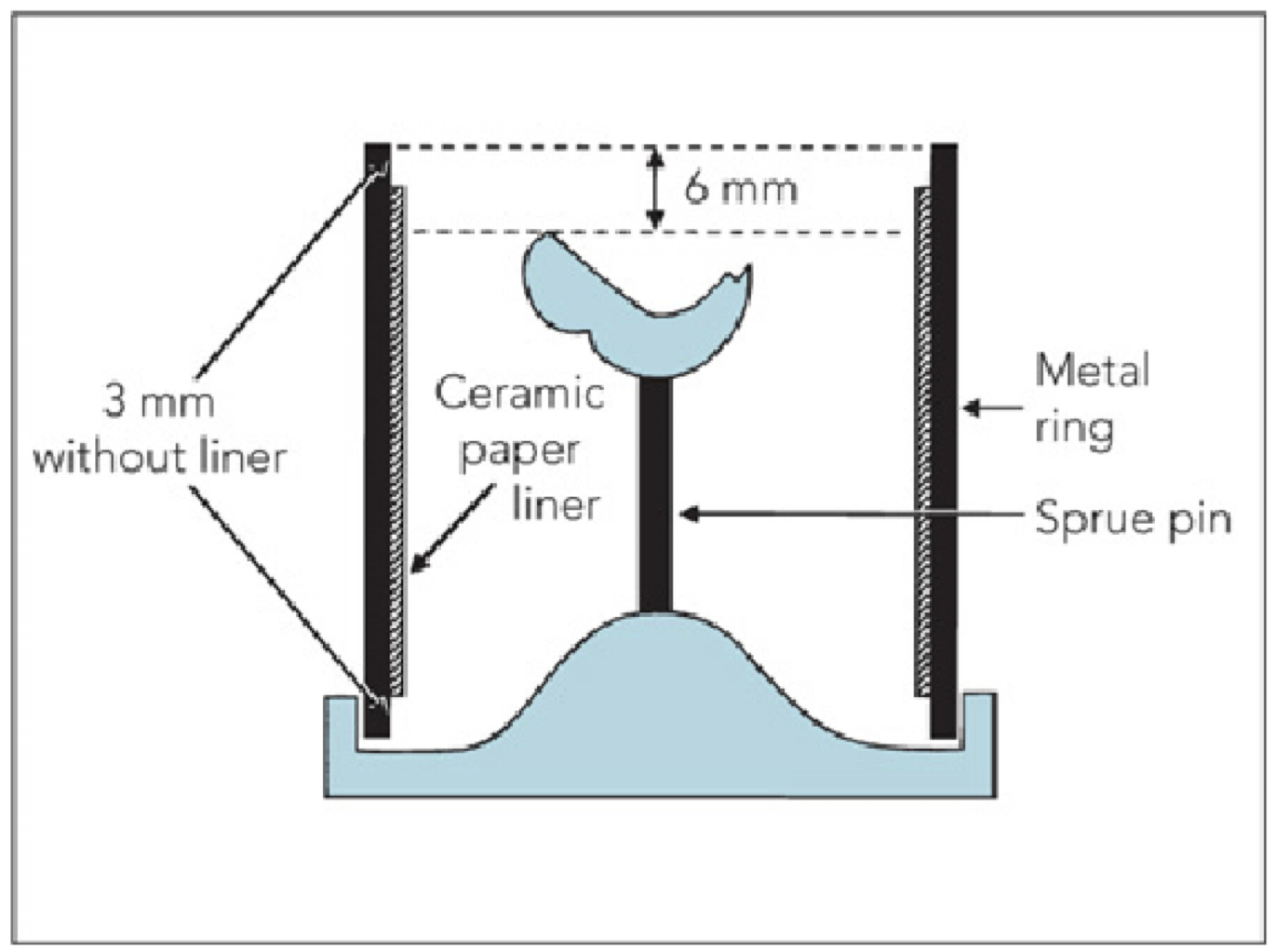
define: the excess material remaining at the base of the mold after dental casting
sprue button
the sprue should be small and long OR large and short?
the sprue must be large enough so it remains open until the casting solidies and short enough to allow rapid filling of the mold
sprue should always attach to the (bulkiest/slimmest) part of the pattern?
bulkiest