Oncolytic viruses - specificity
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
what are the 3 major parts of cancer?
tumour cell (rogue cell) → build complex environment (tumor microenvironment) → evade immune cells
all of these aspects must be present for it to produce cancer
mutations in what kinds of genes lead to uncontrolled growth?
oncogene: normally a ‘go’ step that is ‘control’, mutation leads to ‘uncontrolled go’
eg. CDKs, Ras, E2F → overactive induces cancer
tumor suppressor: normally a ‘stop’ step to stop ‘uncontrolled go’, mutation leads to ‘uncontrolled go’
p53 → inactive p53 induces cancer
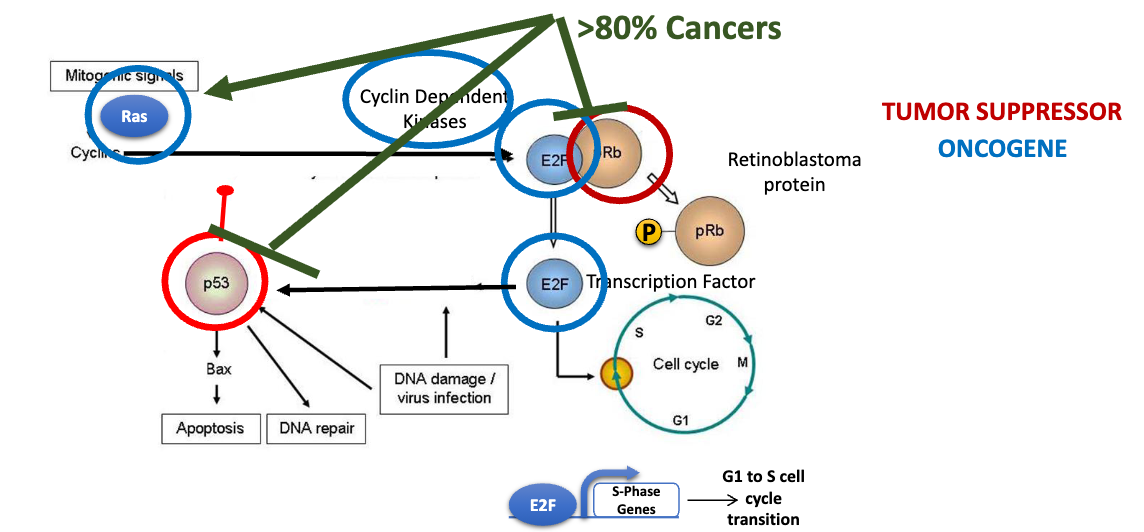
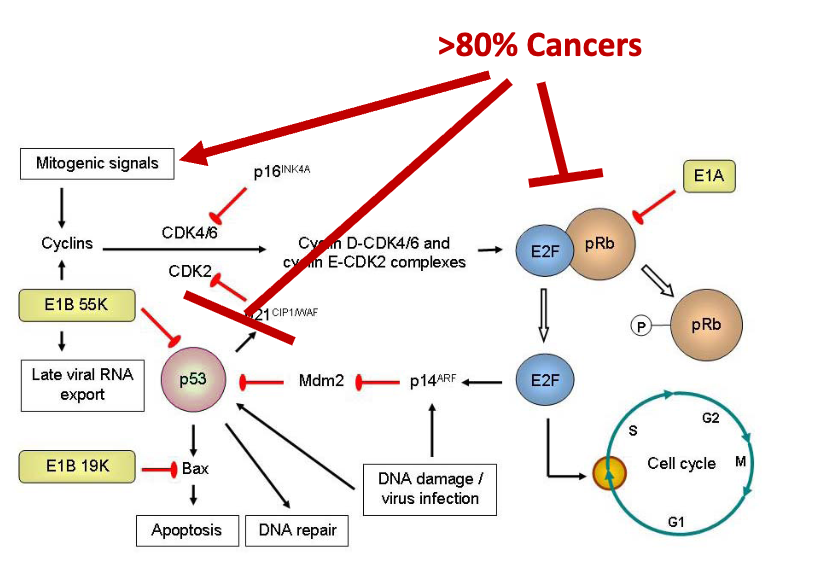
describe the pathway of proteins involved in >80% of cancers
normally: Ras (oncogene) activates E2F (oncogene) by turning on cyclin dependent kinases (CDKs, oncogenes) that phosphorylate Rb (retinoblastoma protein, tumor suppressor), which is bound to E2F to keep in off state. Pi-Rb detaches from E2F transcription factor, which is then able to initiate S phase. E2F also acts in feedback loop, where high levels of E2F (eg from damage) activate p53 (tumor suppressor), which induces cell death
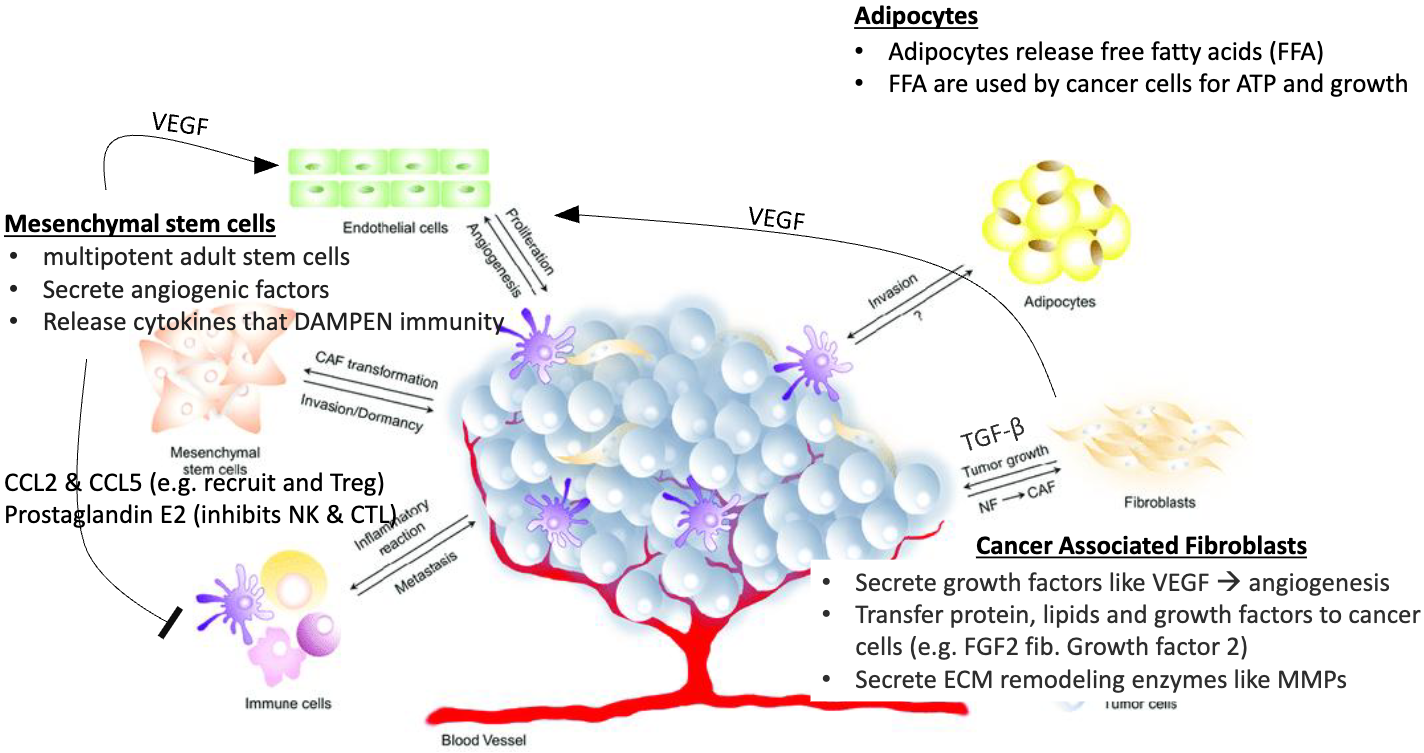
describe the parts of a tumor microenvironment that are required
ECM: provides structure, must constantly be reconfigured/remodeled as tumor grows
ECM-modifying proteases/enzymes
blood vessels: feed tumor
MANY cell types → non-tumor cells that are necessary for tumor development
endothelial cells: supplies nutrients and growth factors
describe the various cell types in the tumor microenvironment that are required to support cancer progression
Adipocytes (fat cells)
release frr fatty acids (FFA) which is ‘food’ for constantly dividing cells
FFA used by cancer cells for ATP and growth
Mesenchymal stem cells
multipotent adult stem cells → can become CAFs
secrete angiogenic factors
release cytokines that dampen immunity → eg, CCL2 & CCL5 (recruit Tregs), and Prostaglandin E2 (inhibits NK & CTLs)
Cancer Associated Fibroblasts (CAFs)
like normal fibroblasts but are in cancer
secrete growth factors like VEGF → angiogenesis
transfer protein, lipids, and growth factors to cancer cells (eg. FGF2 fib Growth factor 2) → help keep everything going
secrete ECM remodeling enzymes like MMPs
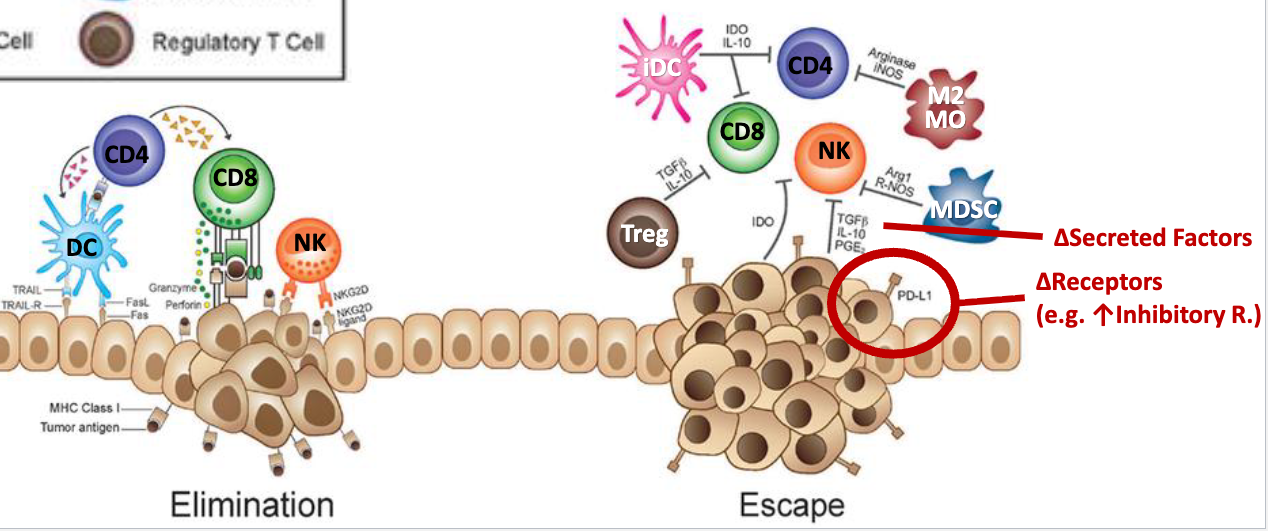
why is it a requirement for cancer that the tumor evade the immune system?
immune system looks for mutations, self in wrong place, etc and can be activated and clear tumor. for tumor to grow, needs to dampen immune response
describe the immunosuppressor cells found in tumors and how tumors suppress the immune system
immunosuppressor cells like myeloid derived suppressor cells (MDSCs), immature DCs (iDCs), Tregs, and M2 macrophages suppress activity of helper and cytotoxic T cells, and NK cells
tumor supporting cells secrete cytokines that inhibit immune cells and support immunosuppressor cells
secrete TGFB, IL10, PGE2
tumor cells upregulate inhibitory receptors → eg. PD-L1 on tumor cells
PD-L1 is an inhibitory receptor that binds PD1 on T cells and inhibits T cell activation
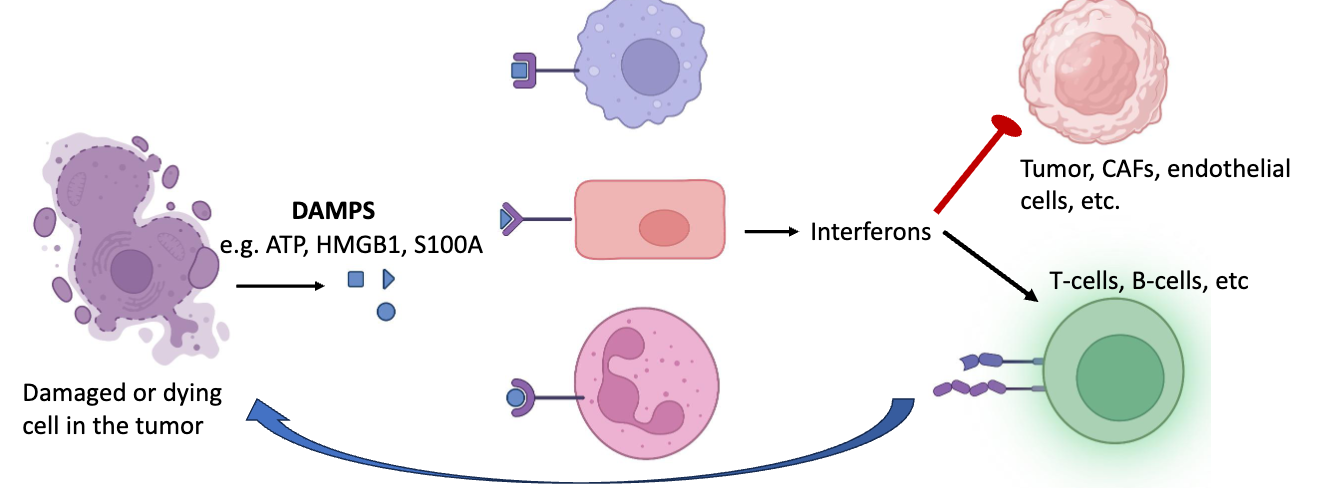
describe how even at the cellular level, ‘antiviral/anti-damage’ pathways are dampened
DAMPs like ATP, HMGB1, S100A are released from damaged and dying cells → frequent collateral of fast growing tumor environment. similar to PAMPS, DAMPs stimulate IFN expression
IFNs have negative effects on cell growth and cancers, and prompt anti-tumor immune cells
this pressure slects for dysregulated antiviral/anti-damage signalling pathways → pressure of IFN leads to pressure for tumor cells to evolve and dampen IFN production/response
how does the tumor and tumor microenvironment relate to viruses?
increased cell division/signalling, vasculature, and immune/IFN suppression make tumors a great environment for viruses
describe how oncolytic viruses work
kill cancer cells, dismantle tumor microenvironments, and stimulate anti-tumor immunity → are also self-dosing
is cleared in healthy cell, but can replicate in tumor cell
tumor cell replication leads to direct tumor cell death, local inflammation (destruction of tumor microenvironment), release of virus progeny (infect more tumor cells), and systemic anti-tumor immune response
self dosing: doses up in tumor cells, doses down elsewhere
changes cyto/chemoines in tumor microenvironment, promoting immune activity
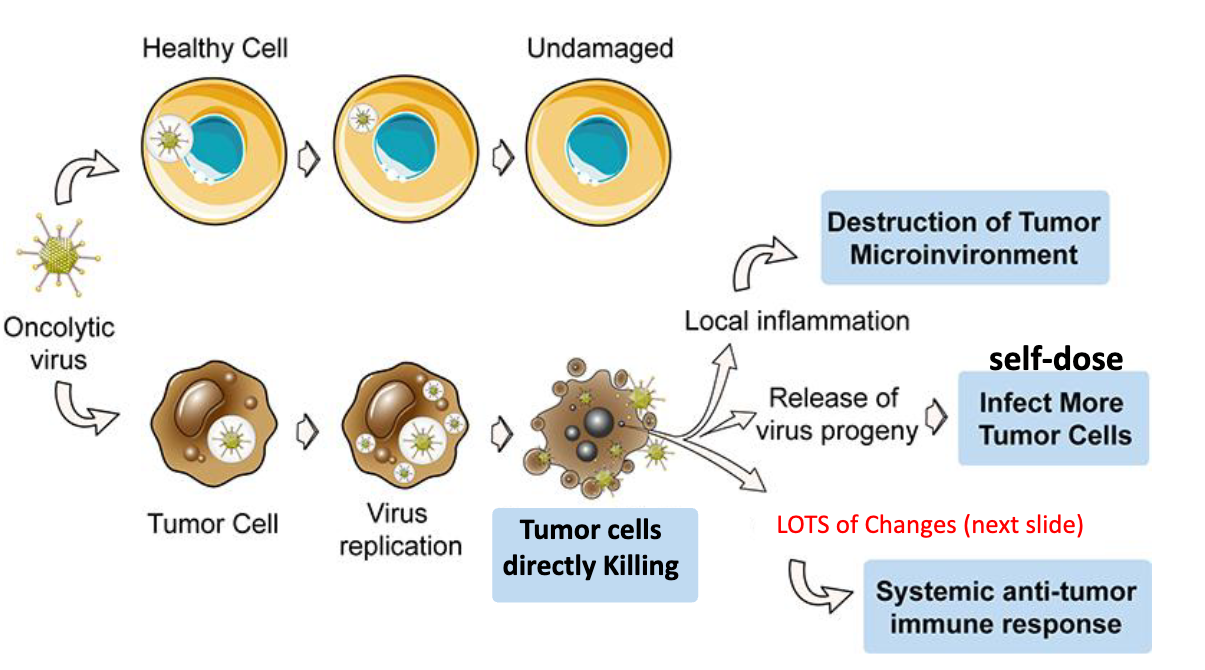
how do OVs promote anti-tumor immunity?
increases PAMPs/DAMPs → immune cell stimulation
increase proinflammatory cytokines → more recruitment of immune cells
increase tumor antigens → DCs now present and activate tumor-specific T cell and B cell responses
but can also induce more immunosuppression?
what are the 3 general ways to make oncolytic viruses specific for tumor cells?
re-targeting by modulation → eg. adenovirus
re-targeting by deletion → eg. HSV, VACV
“wrong host or niche” → VSV, NDV, reovirus
describe adenovirus
non-enveloped DNA virus, 35kb, ~25 genes
replicates in nucleus
57 serotypes → most cause mild respiratory infections, conjunctivitis, tonsilitis
fiber needed for Ad infection → fiber is trimer that attaches to cells
E2F and cell cycle needed to Ad replication → E2F drives expression of viral E1A/E1B, which pull Rb away from E2F → more E2F = increased cell replication
E2F transcribed E2 viral genes, also drives cell cycle providing Ad DNA replication, NTPs, lipids
E1B 55K and E1B 19K block p53 pathway
describe an example of making a virus specific for cancer cells by modulation
transductional targeting → decide what it binds to, modify Ad to make specific for cancer cell
making a virus selective by changing their cell receptor binding specificity towards receptors common in cancer cells
Ad fiber normally interacts with coxsackievirus-adenovirus (CAR) receptor, BUT, CAR is low on cancer cells
modify fiber specificity to make it specific for cancer cells → using biochemical or genetic targeting
describe the biochemical vs genetic targeting in transductional targeting example
make Ad fiber specific for cancer
biochemical targeting: virus can be directed to specific receptors with ‘adaptors’ → put adapter b/w fiber & something specific on tumour cell (eg. Ab)
genetic targeting: fiber knob domain (binding part) can be substituted with receptor binding domains from: other human serotypes, xenotype switching (other species), pseudotyping (other viruses), add binding domains
eg. Ad5/49K
what are the advantages/disadvantages of biochemical vs genetic transductional targeting?
biochemical: only works on incoming viruses (not new progeny) so adapters not self-dosing, but is easier
genetic works on incoming & progeny virus, but is very difficult to achieve and just might not be able to add on
describe another way to make virus selective by modulation
transcriptional targeting → make virus selective by driving expression of virus’ essential genes (like E1A/B) by transcription factors expressed in cancer cells
eg. oncogene Survivin (BIRC5), a member of the inhibitor of apoptosis gene family is overexpressed in most cancers, but not in normal cells. Engineer virus so E1A/B is under Survivin promoter → showed specifically reduced viability of lymphoma cells but not healthy PBLs
what are challenges of using transcriptional.transductional targeting as the major basis for specificity to cancer cells?
harder than you think → might not get something
tumour cells acquire more & more mutations → select for cells that have that protein downregulated
cancers are very different from each other, not many “common” receptors or transcription factors
describe how to make a virus specific for cancer cells by deletion
viruses evolve many ways to interrupt pathways in normal cells → viruses need to turn on/off key processes, such as apoptosis, antiviral state, cell cycle and nucleotide synthesis, nuclear import etc
cancer cells turn on/off similar key processes
if you delete these viral regulatory genes, virus will not replicate in a ‘normal’ cell BUT will replicate in a cancer cell where the process is circumnavigated already by cell
describe the specificity by deletion example 1
disable Ad E1A so that it can’t turn on cell cycle through Rb and p53 modulation, which will disable Ad from replicating in normal cells. But, since most cancers have the same pathways dysregulated, Ad will still replicate in cancer cells
E1A normally interferes with Rb, E1B 55K binds p53, E1B 19K blocks Bax
BUT E1A has many essential roles, so can’t just delete whole protein → just delete the E1A Rb-binding domain
what modifications does the DNX-2401/Delta -24-RGD Ad virus have to confer its specificity?
deletion: small deletion to stop Rb interactions
transductional targeting: RGD (Arg-Gly-Asp) (non-specific, bind integrins, many on cancer) mediates CAR-indpendent binding to cellular integrins is added to fiber protein to add specificity
describe the second example of specificity ‘by deletion’
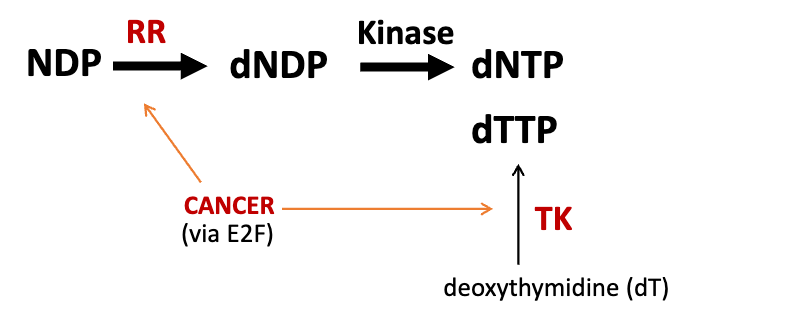
large DNA viruses (HSV and VV) express ribonucleotide reductases because they need large pools of dNTPs for DNA synthesis
nucleotide synthesis requires ribonucleotide reductase (RR) & thymidine kinase (TK)
HSV encodes RR subunit (UL39 gene = ICP6), and TK (UL23)
VACV encodes RR subunit (F4L gene) and TK (J2R) → deletion of F4L RR makes virus non-pathogenic but still replicate efficiently in tumors and prolongs survival in mice with bladder cancer → even higher survival when both F4L and J2R are deleted
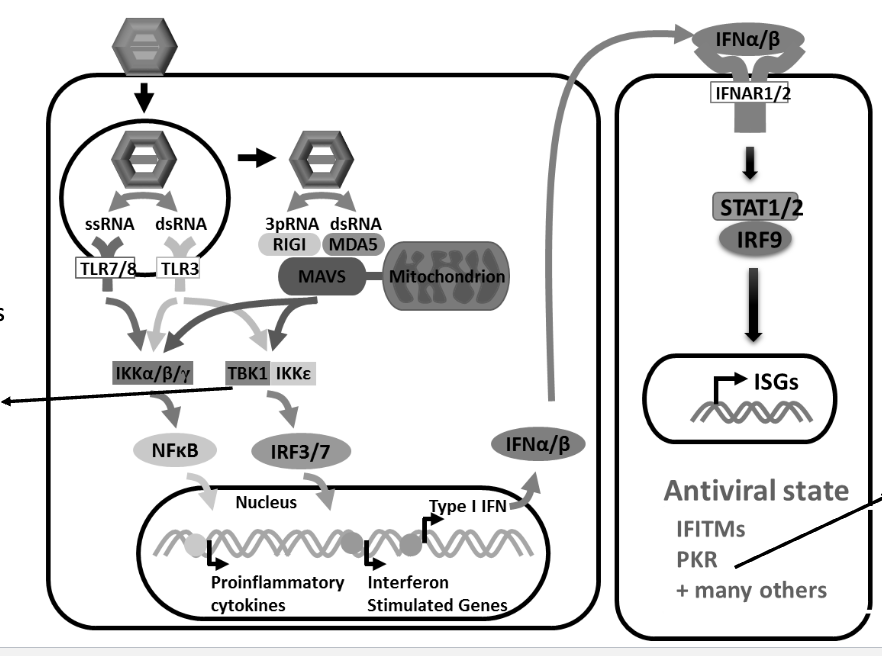
briefly describe the antiviral signalling pathways activated by virus PAMPs
TBK1 is a kinase activated by cytoplasmic receptors that detect viral PAMPs. TBK1 phosphorylates the transcription factor IRF3 that stimulates expression of interferons (IFNs)
when IRF3 phosphorylated, moves into nucleus to increase IFN expression
When PKR binds dsRNA from viruses, it auto- phosphorylates to
become active. Then it stops virus replication by phosphorylating eIF2a and stopping translation imitation
describe the specificity ‘by deletion’ example 3
Herpesvirus g34.5 gene recruits host phosphatase PP1a to dephosphorylate and inactivate TBK1 (blocks IRF3/7 activation) and PKR, blocking antiviral response → if g34.5 is deleted, the virus can only replicate in cancer cells which naturally have these pathways downregulated
Oncovex (T-VEC) → deleted ICP34.5 and ICP6 (RR) to make it specific for cancer cells
describe specificity ‘by insufficiency’
animal-tropic viruses can be specific to human tumor cells without modification → not compatible with human proteins but can be supported in tumor
NDV, VSV, and Minute parvovirus do not have virus genes capable of stopping human antiviral signalling and therefore these viruses are sensitive to antiviral signalling in normal cells. They can replicate in cancer cells with subdued antiviral signalling.
viral genes of these ‘non-human’ viruses are not compatible with human pathways anyhow, so even without deleting anything, they depend on transformed cells to provide these processes
what is an example of a virus with specificity ‘by insufficiency’?
Newcastle disease virus (NDV) → in humans is usually asymptomatic, possible mild symptoms → mild conjunctivitis and flu-like symptoms. In birds, have virulent and non-virulent strains
NDV, like other paramyxoviruses, uses viral protein “V” to stop IRF3 nuclear translocation and prevent IFN production. However, this is species-specific, and can overcome IFN production in chicken cells but not human cells
in chicken cells, V necessary for virus rep bc deleting V reduces virus replication by a lot → in human cells, even with V, NDV replicates poorly, and ± V doesn’t have much impact on rep
know IFN is involved bc if flu NS1 added (known IFN inhibitor in both chickens and humans), dV rep is restored in chicken cells, and both wt and dV rep restored in human cells
NDV therefore can’t repliacte efficiently in IFN-competent normal cells, but can replicate well in IFN-deficient cancer cells
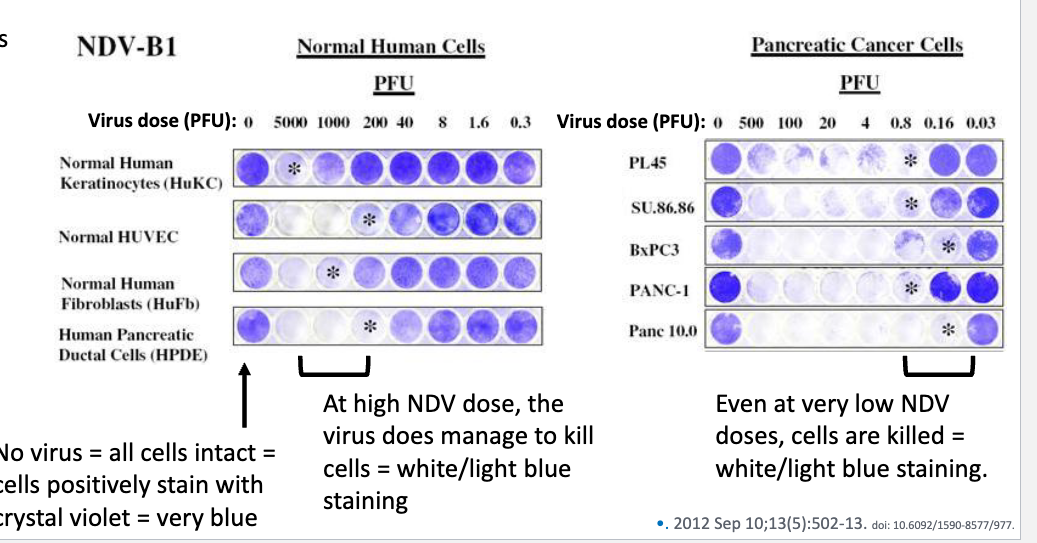
describe the second example of specificity by ‘insufficiency’
reovirus → very stable in enviro, decades in water, ubiquitous everywhere → so stable that doesn’t need to stop immune system
non-pathogenic virus that infects most mammals including humans - many hosts
naturally infects replicating gut enterocytes where it sheds 10000s new viruses into enviro
naturally cleared by immunity rapidly - didn’t ‘bother’ collecting virus genes to modulate antiviral signalling → cancer being immunosuppressed (less IFN, less cellular immunity) permit reovirus infection
didn’t ‘bother’ collecting genes to modulate cell signalling since gut crypt cells provide Ras signalling, p38 signalling, and Pl3K signalling to mediate various stages of virus replication → cancer cells also have these paths ‘on’ while normal cells do not have them constitutively active