6. organisms respond to changes in their internal and external environments
1/161
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
162 Terms
give the stages of urine formation:
formation of glomerular filtrate
reabsorption of glucose and water by proximal convoluted tubule
maintainance of a Na+ gradient in the medulla by the loop of Henle
reabsorption of water by distal convoluted tubule and collecting ducts
antidiuretic hormone (ADH) binds to V receptors found in cell-surface membranes in 2 parts of a nephron - name the 2 parts of a nephron where V receptors are found (1)
distal convoluted tubule and collecting duct
a decrease in blood pressure stimulates the release of ADH - give the location of the receptors that detect a decrease in blood pressure and explain how the release of ADH will affect blood pressure (3)

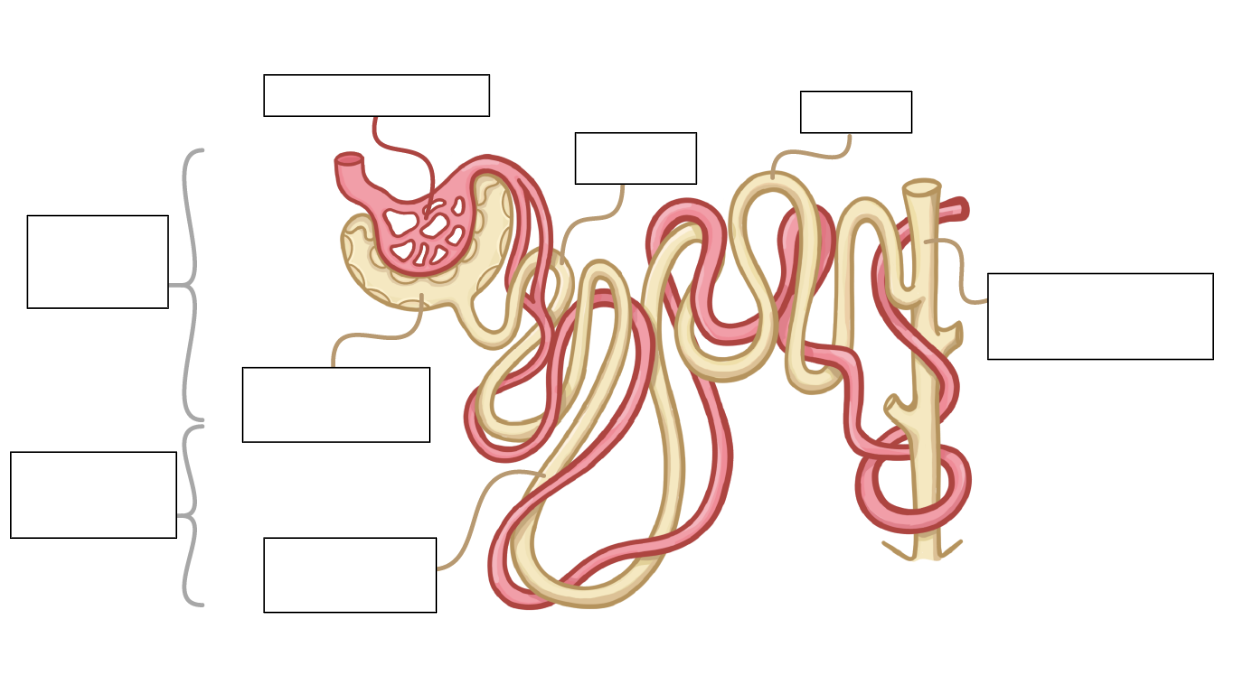
can you label this diagram of a nephron?
PCT = proximal convoluted tubule - closest to Bowman’s capsule
DCT = distal convoluted tubule - further from Bowman’s capsule
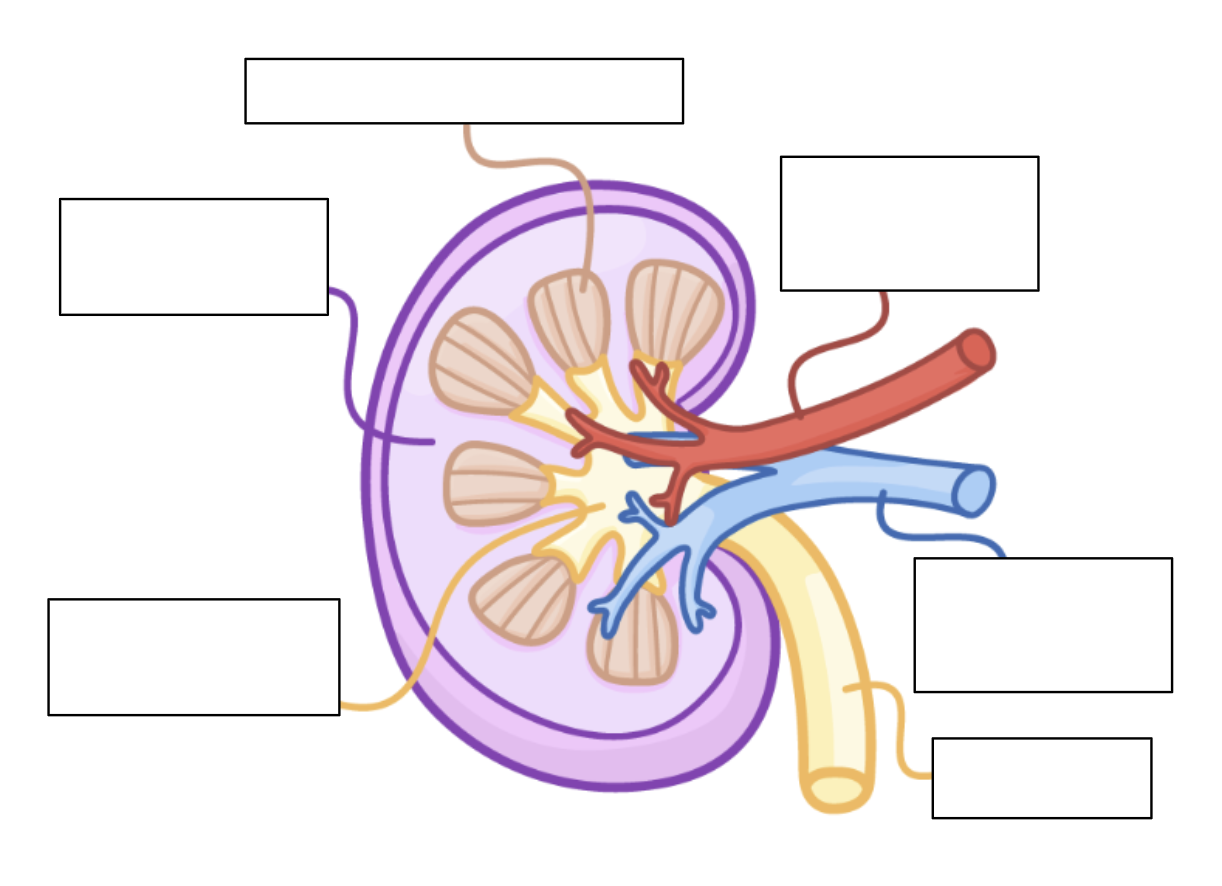
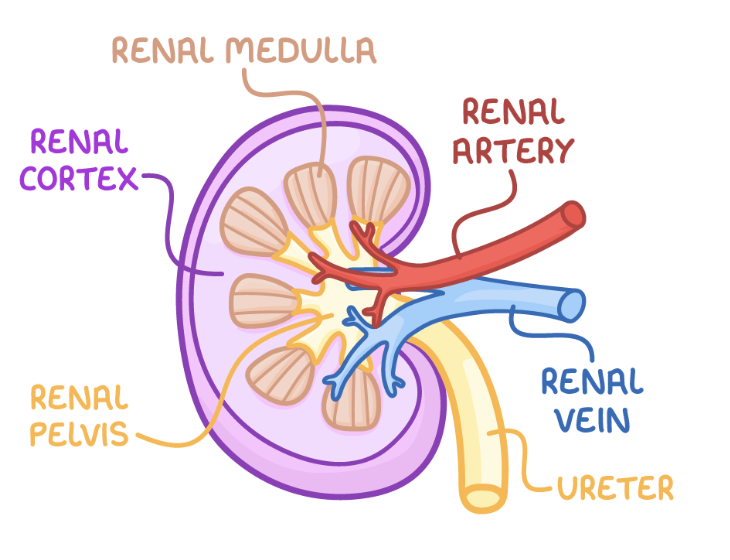
can you label this diagram of the kidney?
what does it mean if the loop of Henle is longer/medulla is deeper?
lower ψ in medulla so steeper conc gradient
more water reabsorbed
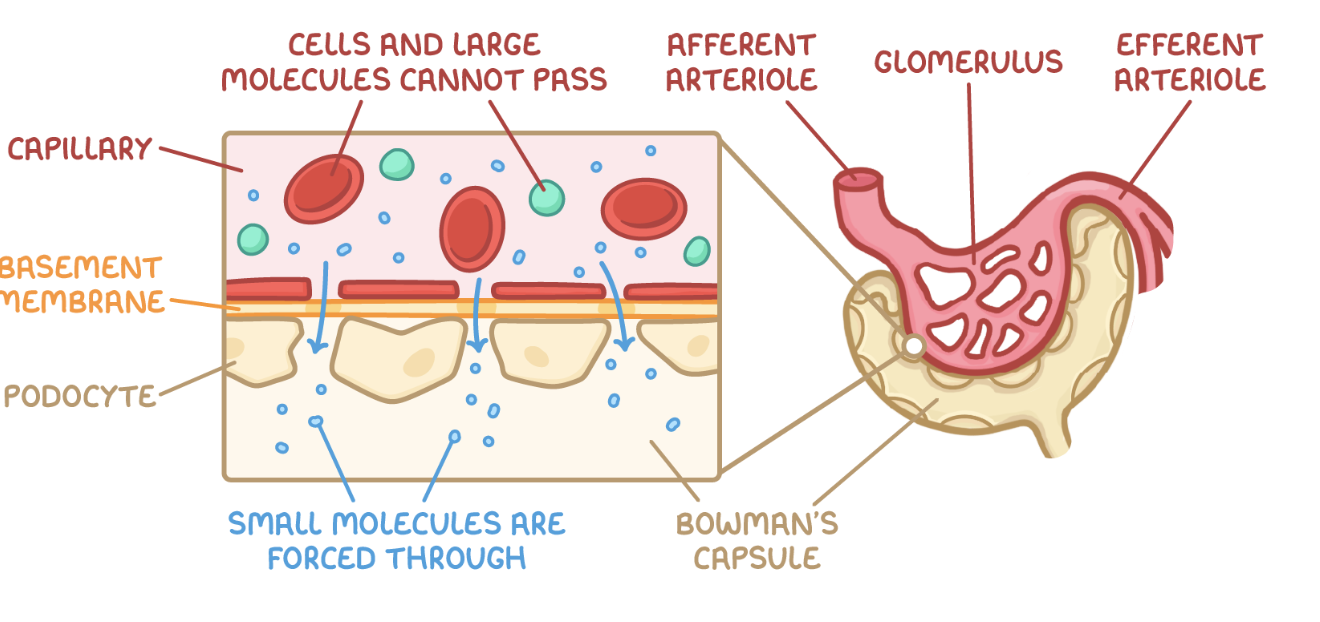
describe the formation of glomerular filtrate:
blood enters the glomerulus through the afferent arteriole
blood leaves the glomerulus through the efferent arteriole, which is smaller so maintains a higher hydrostatic pressure
this high pressure forces smaller molecules e.g. water, glucose, urea out through gaps (fenestrations) in the capillary endothelium
the molecules move through the basement membrane, which acts as a selective filter, preventing larger molecules such as RBCs and proteins into the Bowman’s capsule
smaller molecules move through the Bowman’s capsule epithelium through podocytes (specialised cells)
filtered fluid collects in the Bowman’s capsule
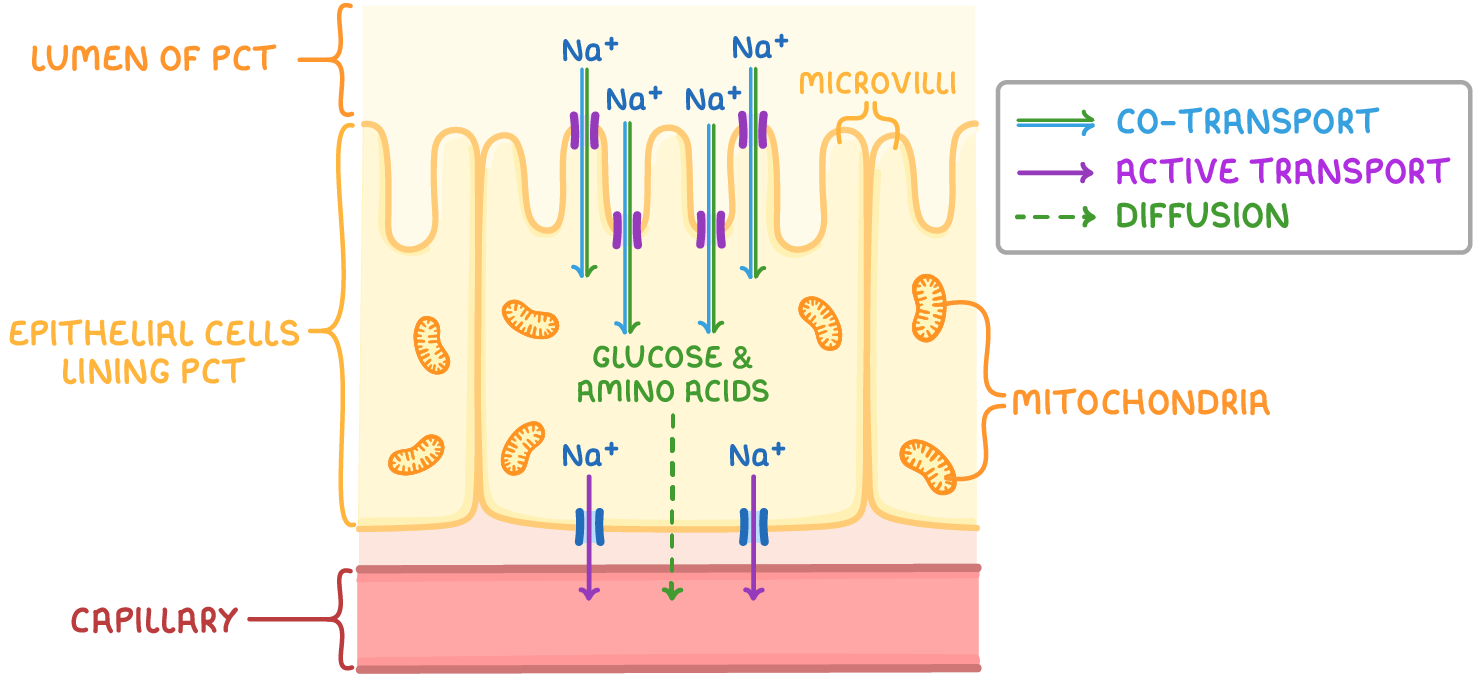
describe and explain how 3 features of the cells in the proximal convoluted tubule allow the rapid reabsorption of glucose into the blood (3)

give 4 adaptations of the distal convoluted tubule:
many microvilli so SA increases
many mitochondria so higher R rate
ADH receptors
selective permeability
what is the purpose of the distal convoluted tubule?
makes final adjustments to pH balance and water content in the blood
adjusting water reabsorption determined by ADH
describe the reabsorption of glucose and water in the proximal convoluted tubule:
Na-K pump actively transports 3Na+ out of the epithelial cell into the capillary - this creates a concentration gradient of Na+ into the epithelial cell as the Na+ conc in the cells lining the PCT decreases
glucose and Na+ enter the epithelial cell by facilitated diffusion - this is cotransport
glucose diffuses out of the epithelial cell into the capillary by facilitated diffusion and is transported in the blood
what happens to urine if the ψ of the blood decreases?
change detected by osmoregulators in hypothalamus which shrink
posterior pituitary gland secretes more ADH
collecting duct and DCT water permeability increases
less dilute urine as more water reabsorbed
what happens to urine if the ψ of the blood increases?
change detected by osmosregulators in hypothalamus which shrink
posterior pituitary gland secretes more ADH
so water permeability of collecting duct and DCT increases
less dilute urine as less water reabsorbed
describe how the loop of Henle maintains a Na+ gradient:
water leaves descending limb by osmosis into interstitial fluid as descending limb permeable to water
lowest ψ at tip of medulla - water reabsorbed by surrounding capillaries by osmosis
at the bottom of the ascending limb, which is impermeable to water, Na+ and Cl- diffuse out due to low concentration of filtrate - this increases the concentration of ions in interstitial space so ψ very low
at top of ascending limb, Na+ and Cl- leave by active transport and ion concentration in filtrate decreases as it ascends
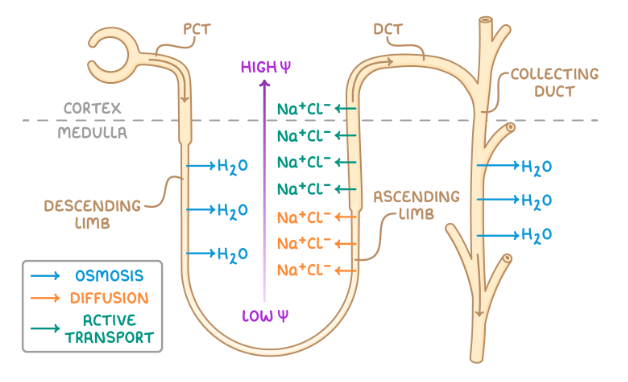
where is the loop of Henle? describe its function:
found in medulla - maintains Na+ grad
ensures that urine produced is more concentrated than blood
functions as a countercurrent multiplier - creates conc grad in surrounding medulla
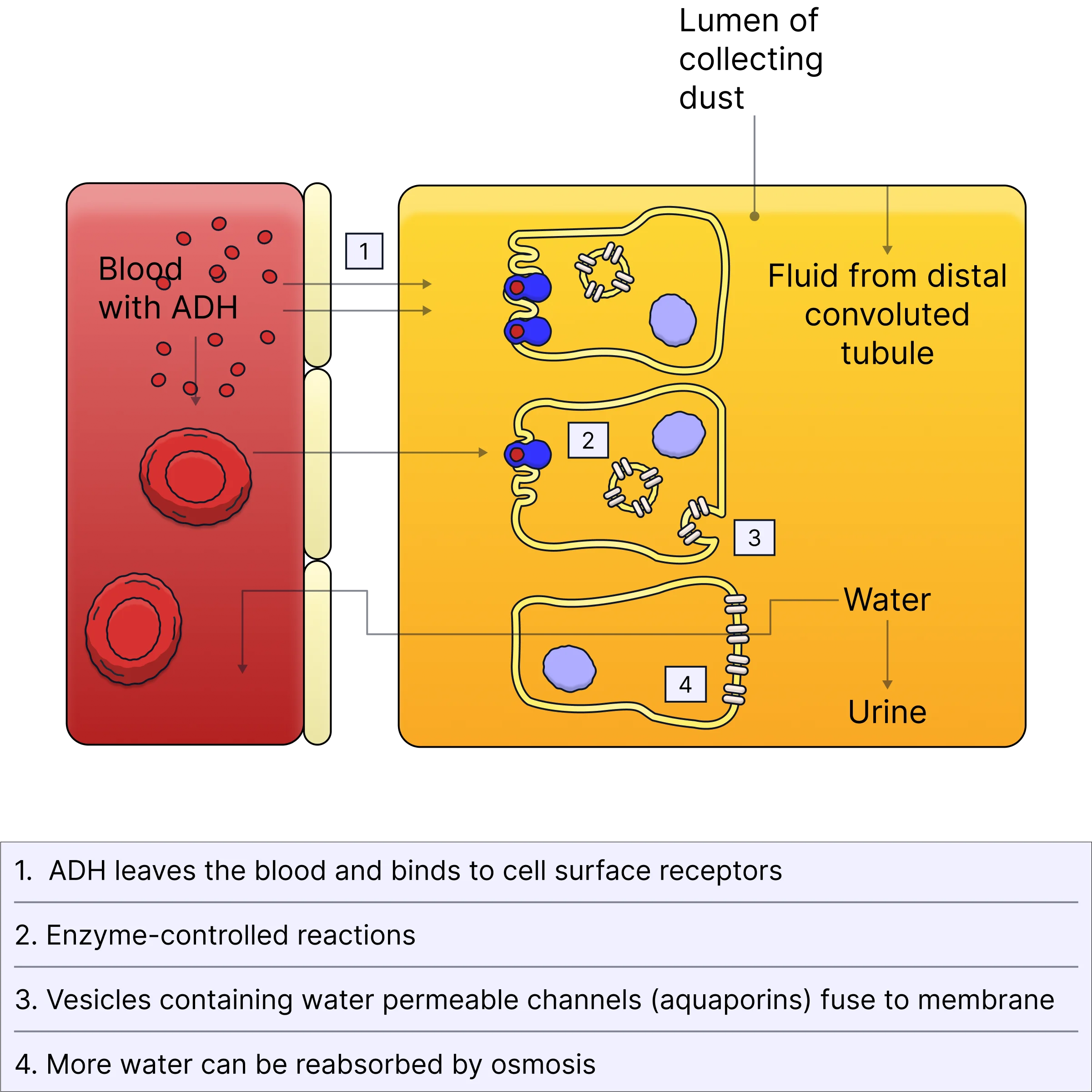
describe the reabsorption of water by the distal convoluted tubule and collecting ducts:
hypothalamus detects low water potential in blood and produces ADH which is secreted into the blood by the posterior pituitary gland
ADH binds to receptors on cells lining the collecting duct on lumen
causes vesicles containing aquaporins to be inserted into cell membrane
water enters cell through aquaporins by osmosis down ψ grad, then moves by osmosis from cell to capillary via interstitial fluid
can you label this diagram?
yes :)
why does the heart need to contract from the base upwards?
contraction starts from the apex of the heart to move blood upwards to arteries, out of the ventricles
empties as much blood as possible from the ventricles
what does it mean for the heart’s contraction to be myogenic?
the heart beats at a baseline rate w/o any input from the nervous system
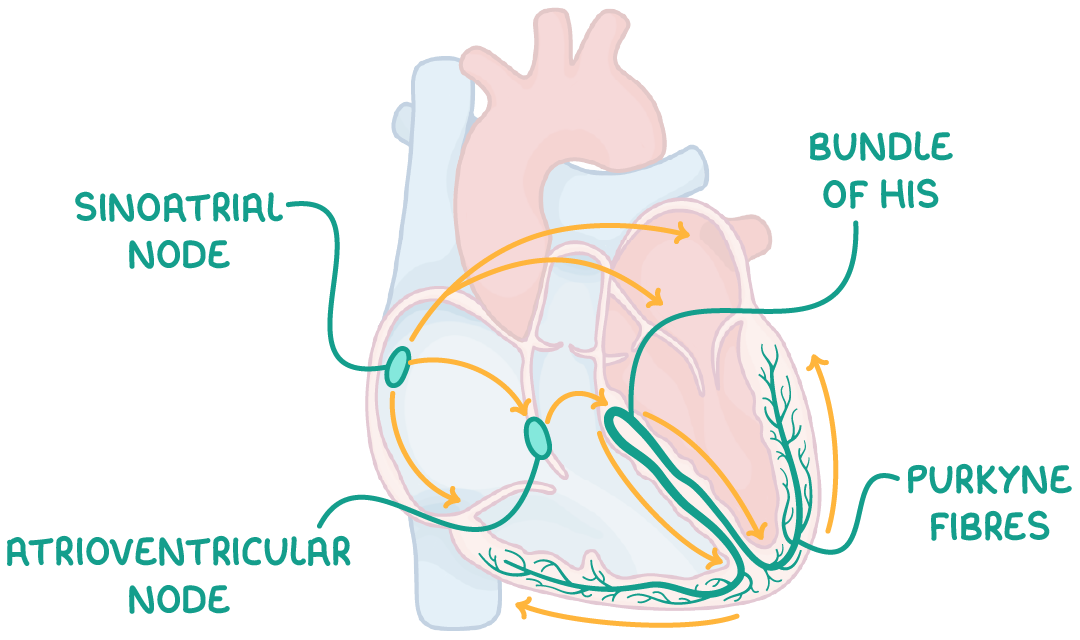
what is the function of the sinoatrial node (SAN)? where is it located?
sends a wave of electrical activity across the atria, depolarising it, causing atrial contraction (i.e. acts a pacemaker)
located in the wall of the right atrium
what is the function of non-conducting collagen tissue?
prevents ventricles contracting at the same time as the atria
what is the function of the atrioventricular node (AVN)?
delays electrical activity, allowing atria to fully empty
sends a wave of electrical activity down the bundle of His and up the Purkyne fibres, depolarising them and causing the ventricles to contract from the apex upwards
explain why there is a short delay between the impulses generated by the SAN and those passing through the AVN (2)
allows atria to contract and empty blood
before ventricles contract
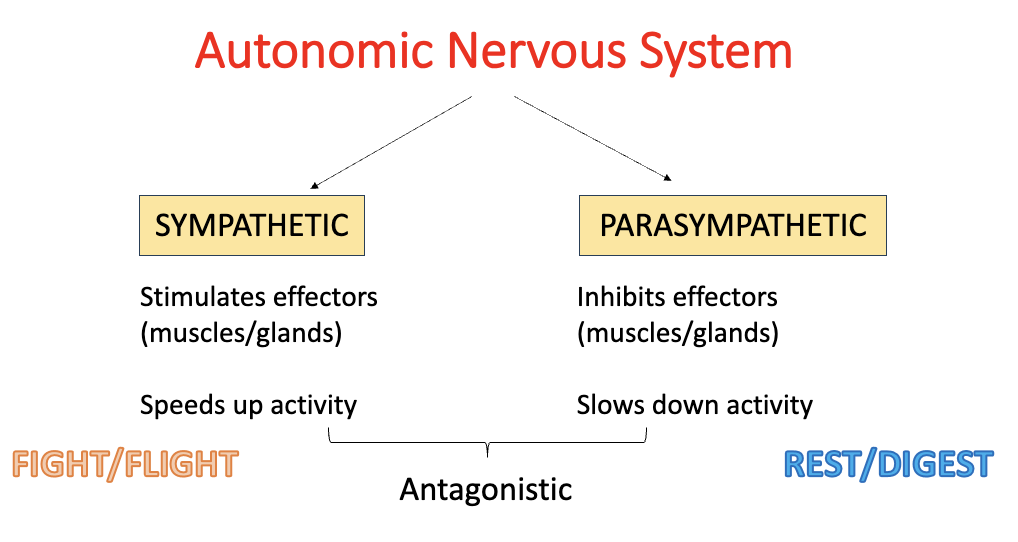
what is the autonomic nervous system?
part of the nervous system which controls involuntary activities
e.g. heart rate, blood pressure, digestion
/ ed into sympathetic and parasympathetic nervous system
name and explain the divisions of the autonomic nervous system:
sympathetic:
stimulates effectors (i.e. increases heart rate)
speeds up activity
(aka. ‘fight/flight’)
parasympathetic:
inhibits effectors (i.e. decreases heart rate)
slows down activity
(aka. ‘rest/digest’)
sympathetic and parasympathetic nervous system are antagonistic - this means they have opposite effects at target tissues
which part of the brain controls changes to the heart rate?
medulla oblongata
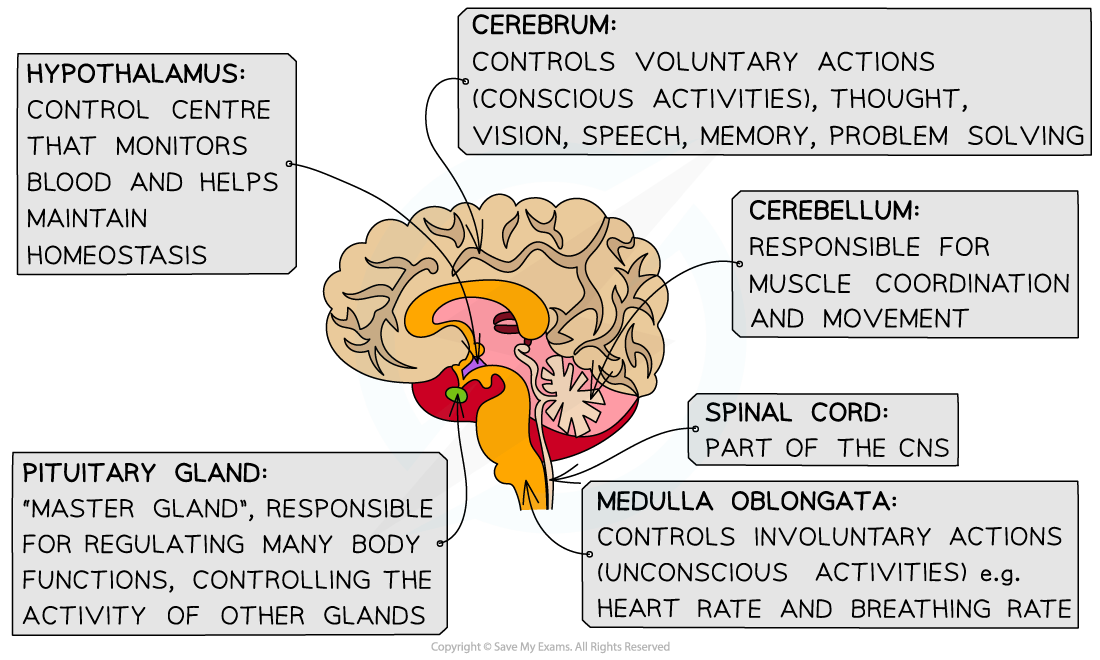
what are baroreceptors? where are they located?
blood pressure receptors - located in walls of aortic and carotid arteries
what are chemoreceptors? where are they located?
chemical receptors - located in walls of aortic and carotid arteries
what happens when blood pressure increases?
baroreceptors in aortic and carotid body are stretched
baroreceptors send increased freq of nerve impulses to medulla oblongata
increased freq of impulses across sympathetic pathway ls stimulation of the SAN by acetylcholine
decreased freq of waves of electrical activity spread across atria and ventricles to decrease heart rate

what happens after increased muscular/metabolic activity?
increased muscular/metabolic activity → increased rate of resp
CO2 conc increases (O2 conc decreases), causing pH of blood to decrease and H+ conc to increase
chemoreceptors in the walls of aortic and carotid arteries detect the decrease in pH
increase in freq of impulses to the medulla oblongata to increase heart rate
increased freq of impulses along the sympathetic pathway to the SAN and noradrenaline is secreted
an increased freq of waves of electrical activity spread across the atria and ventricles to increase heart rate

describe the myogenic stimulation of the heart and how the regular contraction of the atria and ventricles is coordinated - do not include the autonomic nervous system in your answer (5)
SAN releases (wave of) electrical activity
(so) atria contract (at the same time)
AVN relays/passes electrical activity after a (short) delay
(via) Purkyne tissues in/and bundle of His
(so) ventricles contract (at the same time from bottom upwards)

name the cells in the pancreas and give their function:
islets of Langerhans = clusters of specialised cells:
alpha cells - secrete glucagon
beta cells - secrete insulin
why is it important to regulate blood glucose concentration in the bloodstream?
extreme blood glucose levels cause changes in water potential, potentially causing cell lysis
what is glycogenesis? when does it occur?
literally: glycogen synthesis
conversion of glucose → glycogen in condensation reactions
occurs when blood glucose conc is higher than normal
describe what happens when blood glucose conc is too high:
beta cells in the pancreas detect high blood glucose levels, secreting insulin into the bloodstream
insulin binds to receptors on target cells
there are more glucose channel proteins in the target cell membrane, increasing permeability, as vesicles containing these proteins fuse w/ the membrane so more glucose diffuses into the target cells
insulin also activates enzymes that convert glucose → glycogen via condensation reactions in glycogenesis
the glucose concentration in cells decreases, creating a diffusion gradient for more glucose to diffuse in, decreasing the glucose concentration in the blood
what is glycogenolysis?
literally: glycogen hydrolysis
hydrolysis of glycogen → glucose
occurs when blood glucose conc is too low
what is gluconeogenesis?
literally: glucose new synthesis
conversion of AAs and lipids → glucose
occurs when blood glucose conc is too low
describe what happens when blood glucose levels are too low:
alpha cells in pancreas detect low blood glucose conc and secrete glucagon
glucagon binds to specific protein receptors on the surface membranes of liver cells
activates enzymes that hydrolyse glycogen → glucose in glycogenolysis
this activates enzymes that convert AAs and lipids → glucose in gluconeogenesis
glucose then leavers the liver cells by FD, increasing glucose conc in blood
describe how adrenaline acts via the second messenger model:
secreted by adrenal glands to increase blood glucose conc during times of excitement/stress
adrenaline binds to a complementary receptor on the cell surface membrane of a liver cell
the binding of adrenaline causes the protein to change shape, activating a G protein
this activates the enzyme adenylyl cyclase
the activated adenylyl cyclase converts ATP → cAMP
cAMP acts as a 2nd messenger, binding to and activating many protein kinases via phosphorylation, amplifying the signal from adrenaline
protein kinases activate enzymes that catalyse the breakdown of glycogen into glucose in glycogenolysis
glucose moves out of liver cells by FD and into the blood through channel proteins
this increases blood glucose conc so more glucose can be delivered to body cells for R
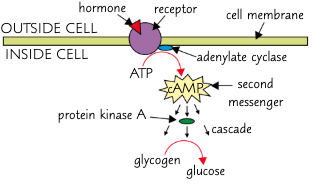
in the second messenger model, what are the first and second messengers? how do they affect each other and what is the rseult?
1st messenger = hormone e.g. adrenaline triggers the formation of the 2nd messenger
2nd messenger = cAMP activates enzymes to carry out extracellular signalling
what is an action potential?
rapid impulse that travels along a neurone, causing changes in membrane potential
what are the 3 main stages of generating an action potential?
depolarisation
repolarisation
hyperpolarisation
describe and explain how an action potential is generated:
resting neurone at resting potential - some K+ voltage gated channels are open, all Na+ channels are closed
stimulus arrives at neurone, causing voltage-gated Na+ channels to open ∴ Na+ diffuses into the axons down an electrochemical gradient, making it less -ve
if the membrane potential reaches the threshold potential of -55 mV, more voltage-gated Na+ channels open - this influx of Na+ causes depolarisation
when enough Na+ enters the axon, membrane potential reaches +40 mV - this is action potential
when action potential has been reached, all voltage-gated Na+ channels close and voltage gated K+ channels open - this means that K+ diffuse down the electrochemical gradient out of the axon
the diffusion of K+ out causes a temporary overshoot of the resting potential - hyperpolarisation - as part of the refractory period
to restore resting potential, voltage-gated K+ channels close and Na-K pump actively transports 3Na+ out and 2K+ in
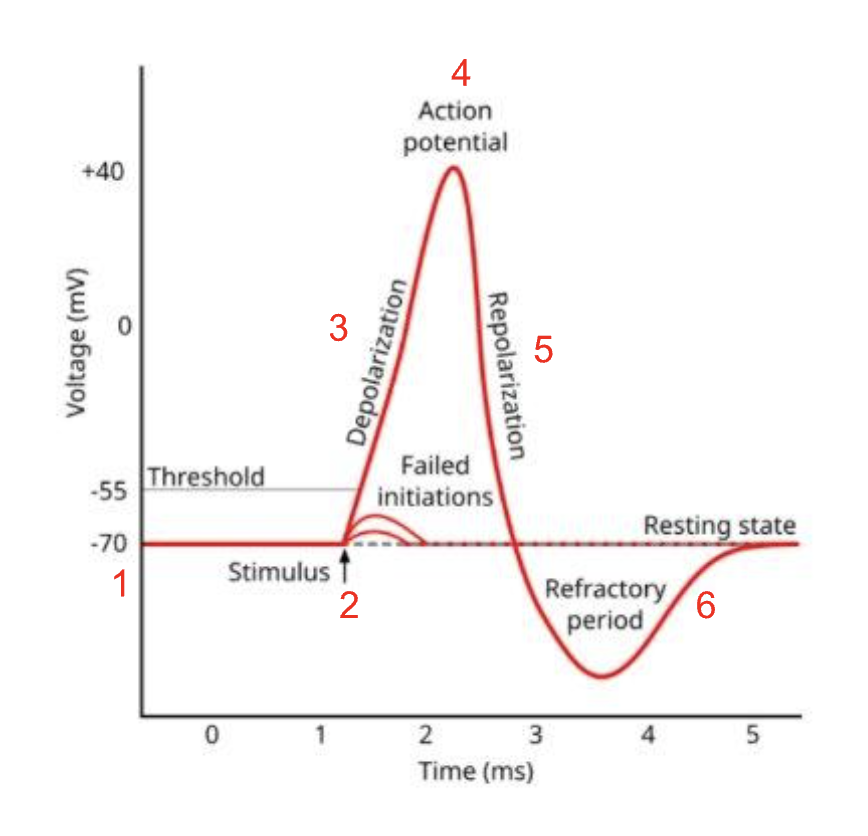
what is depolarisation?
a reversal in membrane potential
what voltage is action potential?
+40 mV
what voltage is the threshold potential?
-55 mV
how does the action potential move along the neurone?
as a wave of depolarisation
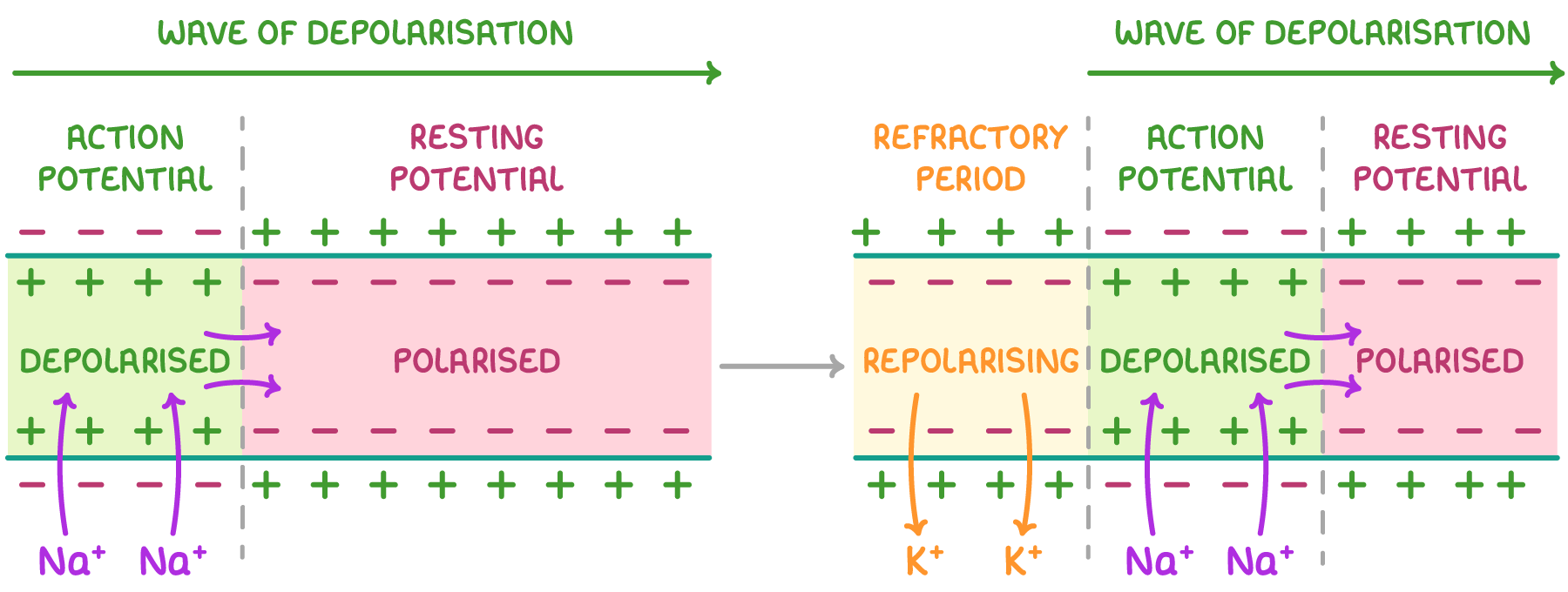
how does the speed of action potential transmission change with axon diameter?
larger axon diameter means there is less resistance to ion flow
∴ wave of depolarisation travels faster
how does the speed of action potential change with temperature?
higher temp → faster diffusion of ions
∴ faster action potential transmissions
over 40oC - proteins denature → slower action potential transmission due to membrane damage
explain the importance of the refractory period:
ensures action potentials are discrete (i.e. don’t overlap)
limits the freq of impulses by setting a minimum time period between action potentials
ensures impulse travels in 1 direction
describe and explain the all-or-nothing principle:
once the threshold is reached, an action potential will always fire w/ the same change in voltage, no matter how big the stimulus is
if the threshold isn’t reached, an action potential won’t fire
a bigger stimulus won’t cause a bigger action potential, but it will cause them to fire more frequently
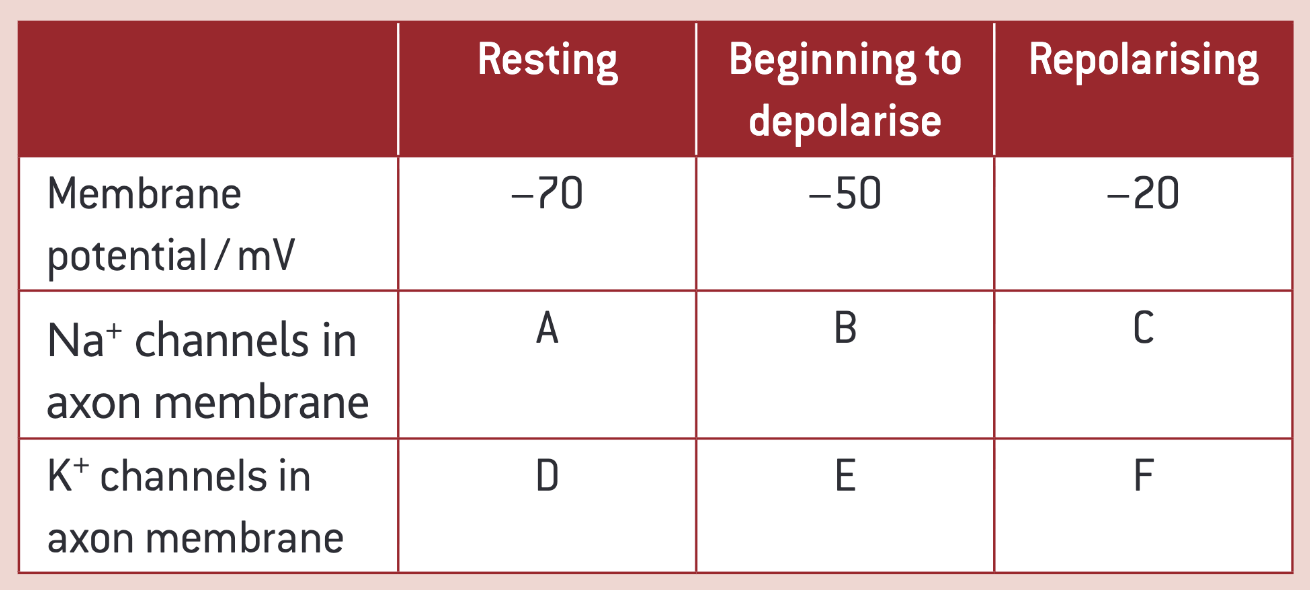
complete this table:
A = closed
B = open
C = closed
D = some are open
E = closed
F = open
describe how resting potential is maintained:
Na+ channels are voltage gated and closed to prevent Na+ diffusing into the neurone
membrane more permeable to K+ ions and less permeable to Na+ ions
K+ leak channels allow FD of K+ out of the neurone
Na-K pump actively transports 3Na+ out and 2K+ into the neurone
this forms an electrochemical gradient as +ve ions accumulate in the extracellular space
this makes the axon cytoplasm -vely charged so the membrane is polarised
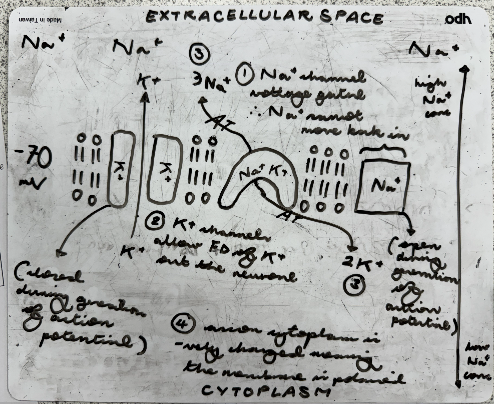
what is the function of the Na-K pump?
actively transports 3Na+ out and 2K+ into the neurone
restores resting potential after action potential
what is the resting potential?
diff in electrical charge across cell surface membrane when a neurone is not transmitting an impulse - 70 mV (the inside of an axon has a charge that is 70 mV more -ve than the outside)
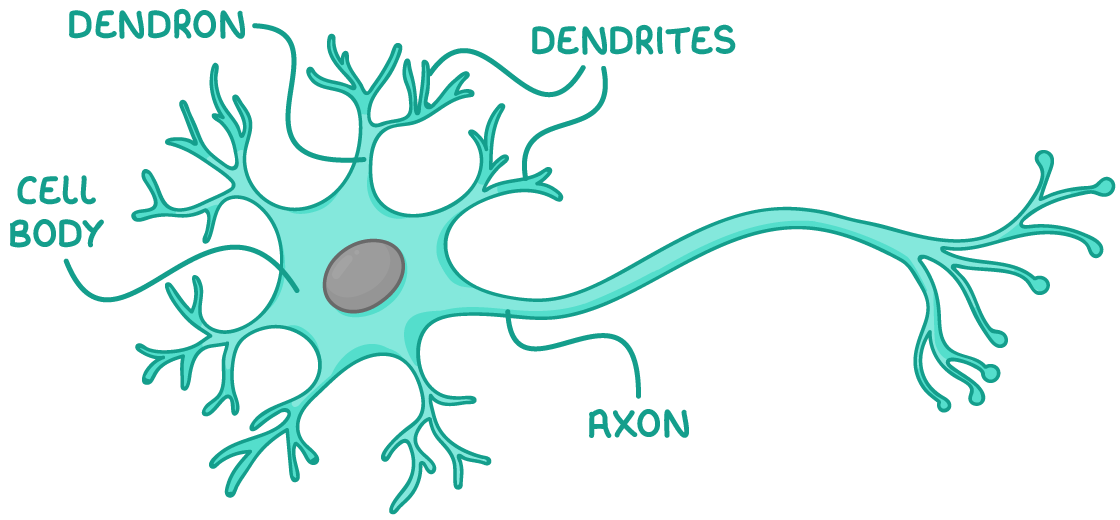
give and explain the features of this neurone:
cell body - contains nucleus and other organelles e.g. mitochondria and ER
dendrons - short branches extended from the cell body which further / into dendrites
axon - single nerve fibre which carries impulse away from cell body to other neurones/effectors
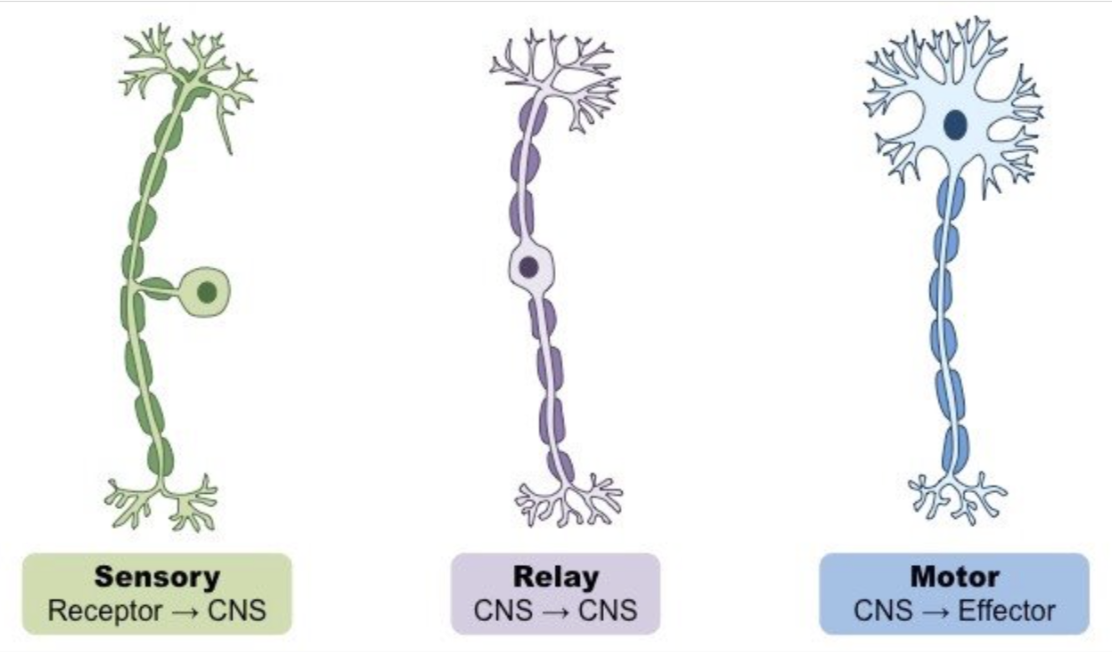
give the order of travel for a reflex arc:
sensory neurone → relay neurone (in CNS)→ motor neurone → effector
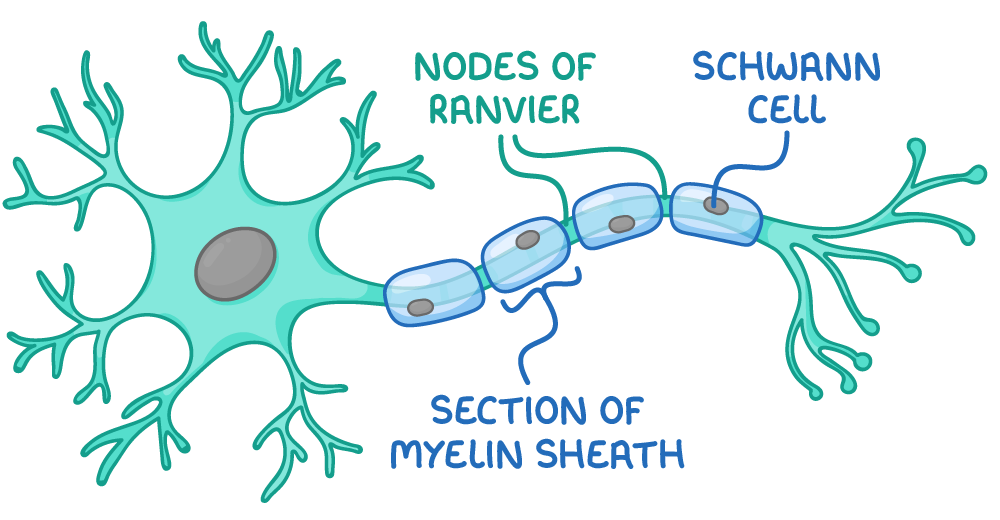
what are the functions of Schwann cells?
membranes form myelin sheath
remove debris via phagocytosis
aid regeneration
suggest 2 advantages of simple reflexes:
any 2 from:
rapid
protect against damage to body tissues
do not have to be learnt
help escape from predators
enable homeostatic control

in this investigation, it is not possible for a student to react in less than 45 ms - suggest one explanation for the value recorded in Trial 3 in Table 1 (1)


in response to touch, nerve impulses can be transmitted at speeds of 76.2 m s-1 - suggest 3 reasons why in this investigation, the estimated speed of student A’s impulse transmission was less than 76.2 m s-1 (3)

a scientist investigated the effect of inhibitors on neurones. she added a respiratory inhibitor to a neurone and the resting potential of the neurone changed from -70 mV to 0 mv - explain why (3)

explain why different proteins are required for the diffusion of different ions through the membrane (2)
each protein has a specific 3o structure
diff ions have diff structures/shapes
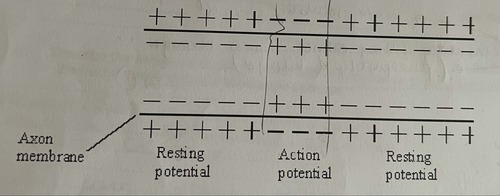
describe how the change shown in the diagram occurs when an action potential is produced (2)
Na+ channels open
Na+ ions enter the axon
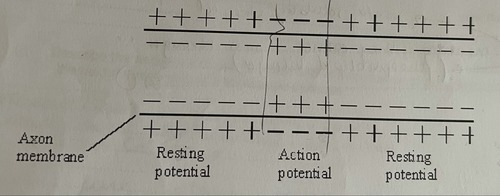
explain what causes the conduction of impulses along a non myelinated axon to be slower than along a myelinated axon (3)
myelinated - ion movement only at nodes of Ranvier
impulse jumps from node to node via saltatory conduction
non myelinated - more depolarisation over whole length of neurone and no saltatory conduction
what is a synapse?
junction between 2 neurones
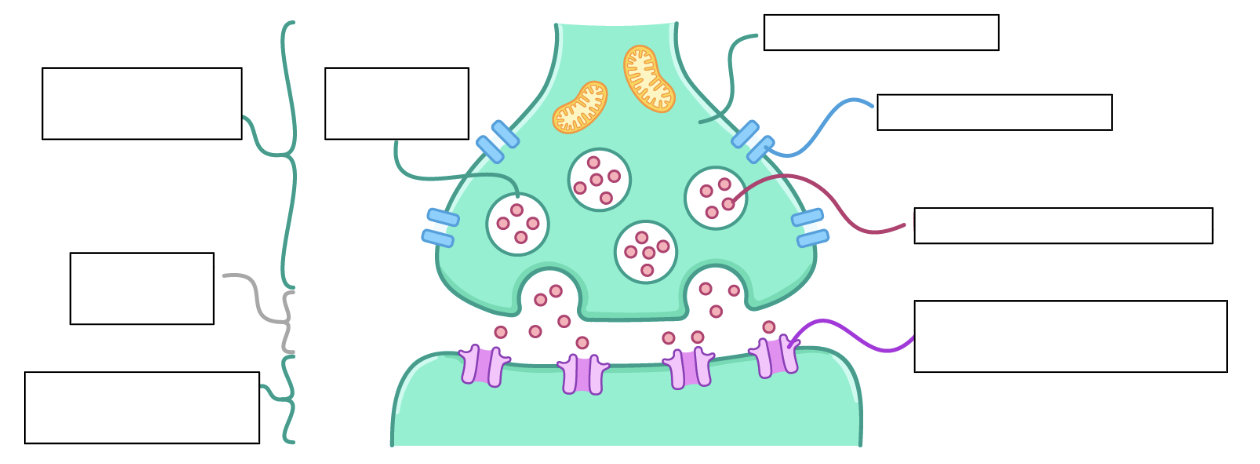
can you label the parts of the synapse?
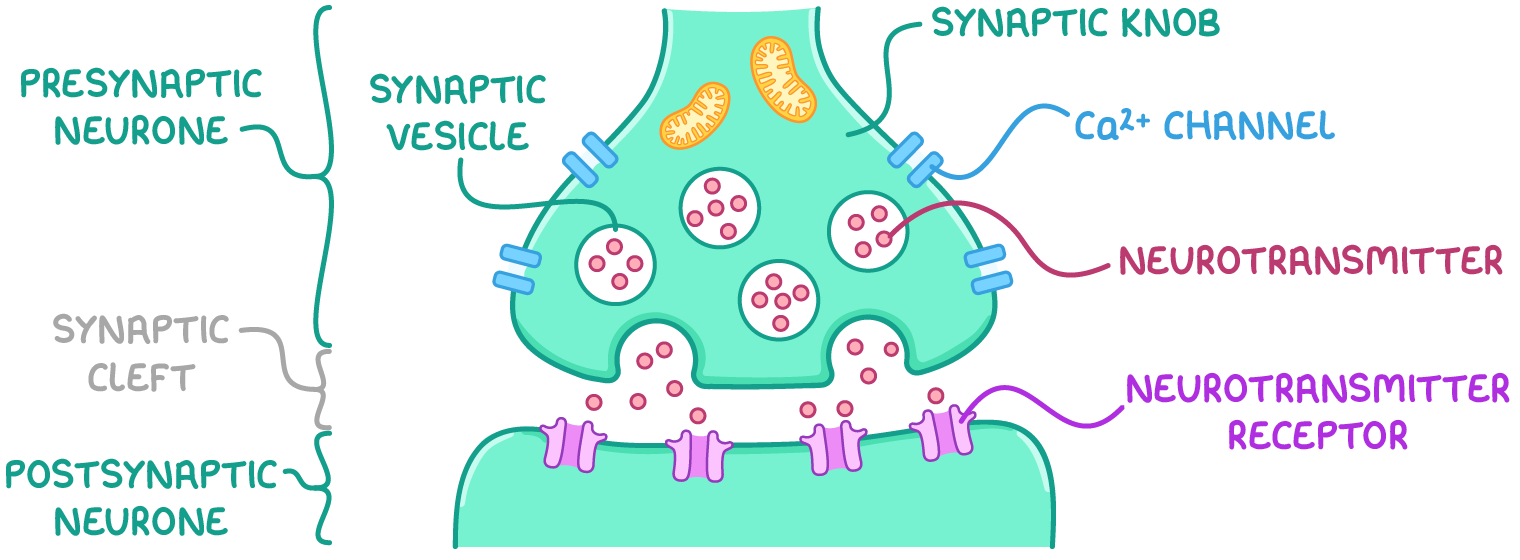
describe the process of synaptic transmission:
at a cholinergic synapse, an action potential arrives at the presynaptic membrane, causing it to depolarise
this triggers the opening of Ca2+ ion channels, allows Ca2+ ions to enter the pre-synaptic knob by FD
the influx of Ca2+ ions causes synaptic vesicles containing the neurotransmitter acetylcholine to move towards and fuse w/ the presynaptic membrane
the neurotransmitter is then released into the synapse/synaptic cleft - it diffuses along the gap and binds to specific receptors on the post-synaptic membrane
this binding causes Na+ ion channels to open, allowing Na+ ions to diffuse into the post synaptic neurone
if enough Na+ ions enter, the membrane will reach the threshold, causing depolarisation and generating a new action potential
what is acetylcholinesterase?
enzyme which catalyses the hydrolysis of acetylcholine into acetate and choline
these products are reabsorbed (endocytosis) into the presynaptic neurone where acetylcholine is regenerated using E from ATP
if neurotransmitter not removed, keeps binding to receptors and keeps causing action potentials
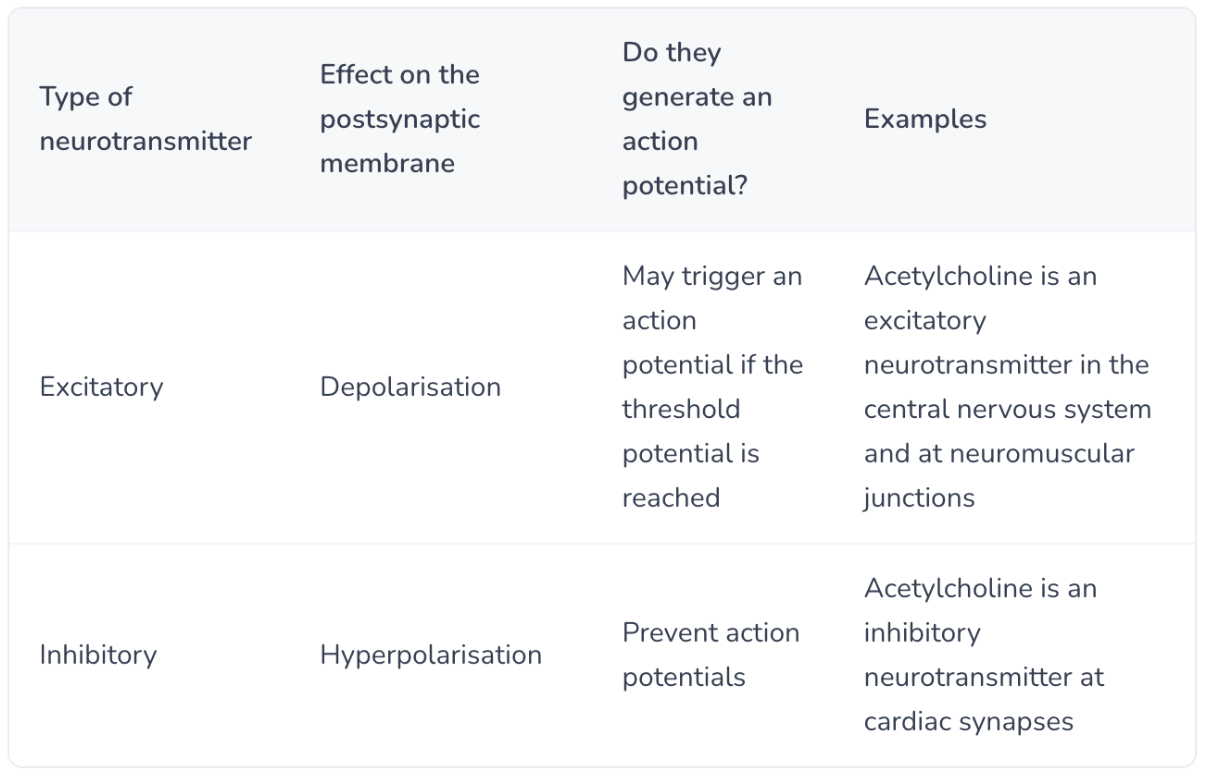
is the cholinergic synapse stimulatory or inhibitory?
stimulatory
give an example of an inhibitory synapse and describe what happens at an inhibitory synapse:
e.g. GABA:
inhibitory neurotransmitters released into the synaptic cleft and bind to Cl- channels on the postsynaptic membrane
Cl- channels open, allowing an influx of Cl- into the postsynaptic neurone by FD
(K+ channels open, allowing K+ ions to leave the postsynaptic neurone)
→ the postsynaptic membrane is hyperpolarised, so action potential not produced and depolarisation does not occur
∴ more sodium ions required to reach threshold for depolarisation/action potential

can you fill in this table?
what is summation?
the process in which the effects of multiple neurotransmitters are combined to produce a response
describe and explain temporal summation:
repeated firing in short succession from the same presynaptic neurone (as neurotransmitters released from 1 vesicle alone not enough to reach the threshold)
threshold is reached and action potential is more likely due to the increased amount of neurotransmitter
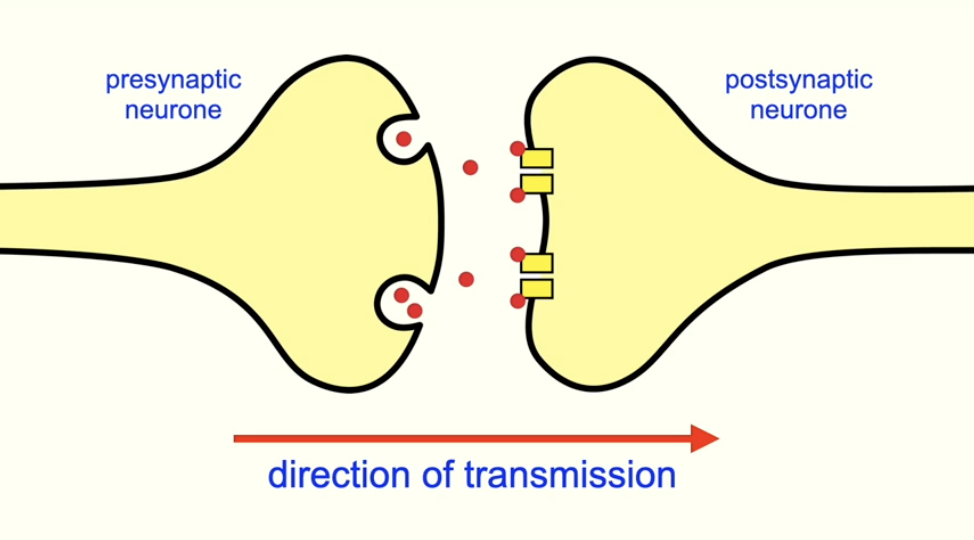
in temporal summation, why must the repeated firing occur in short succession?
if the second firing does not occur until a while after the first firing, the action potential from the first firing gets broken down (as it does not reach the threshold)
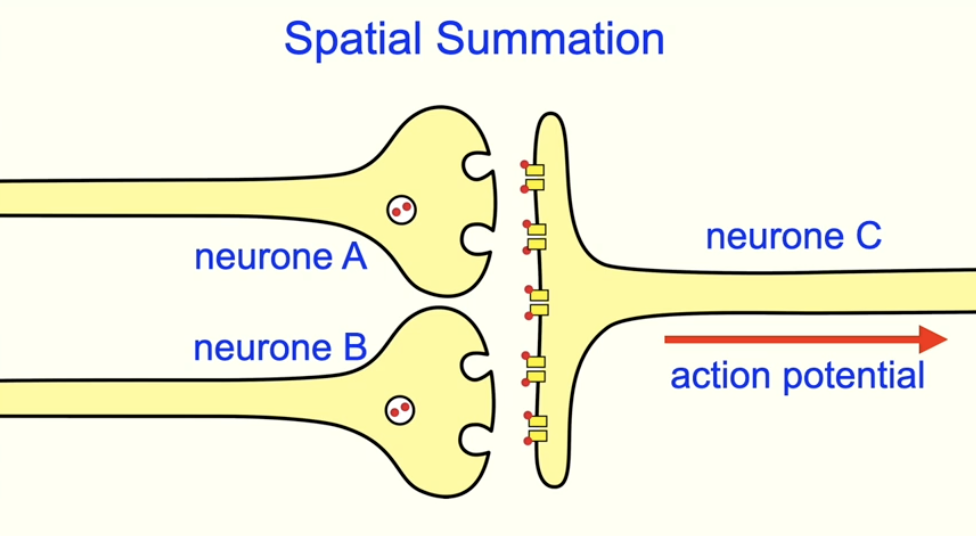
describe and explain spatial summation:
multiple presynaptic neurones converge on 1 postsynaptic neurone
individually, the neurones do not release enough neurotransmitter to reach the threshold
but the combined effect of all neurotransmitters is enough for the postsynaptic neurone to reach the threshold and trigger an action potential
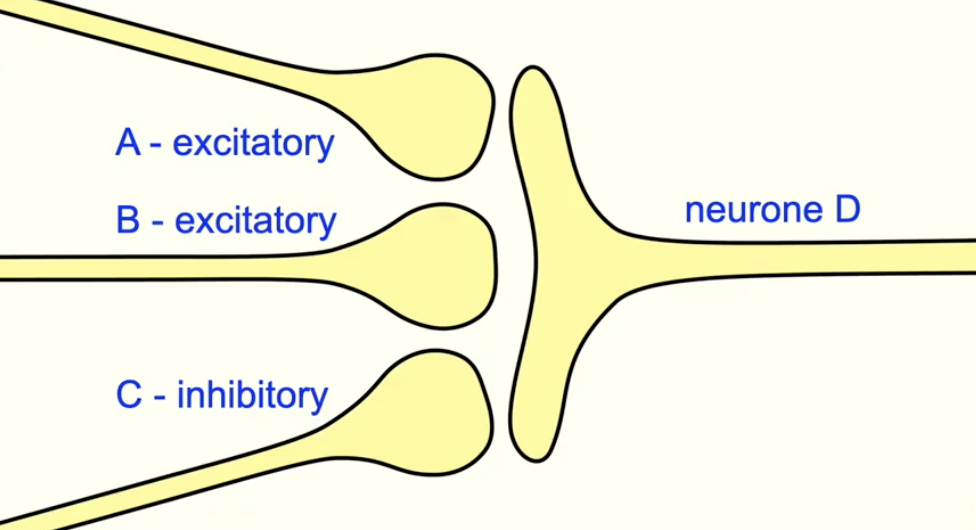
why may summation occur when an inhibitory synapse is present?
if the threshold is reached, an action potential will fire
if multiple presynaptic neurones converge on 1 postsynaptic neurone and some are excitatory and others are inhibitory:
for an action potential to occur, the excitatory neurones must summate
to overcome the hyperpolarisation caused by the inhibitory neurones
S is a similar shape to acetylcholine - suggest how anaesthetic S stops the transmission across the synapse (3)
complementary to receptor for acetylcholine
binds to receptor
on postsynaptic membrane
prevents acetylcholine from binding
∴ no action potential in postsynaptic neurone as neuronal activity is inhibited
give 2 key features of receptors:
receptors respond only to specific stimuli
stimulation of a receptor leads to the establishment of a generator potential
what is a generator potential?
the initial nervous impulse that is generated
can you label this Pacinian corpuscle?
(sensory neurone) axon
(sensory) neurone ending
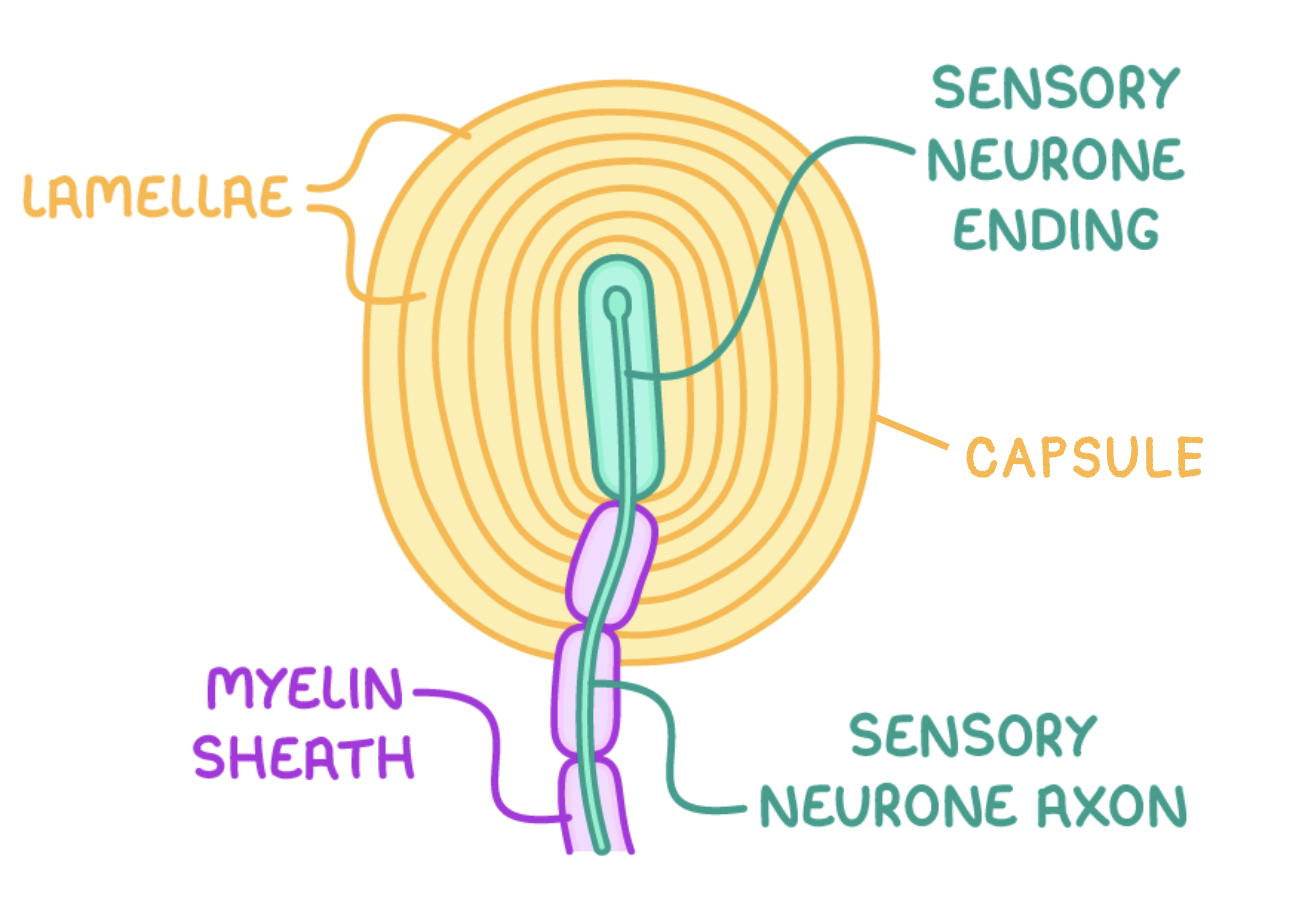
give an example of a receptor - what does it respond to and what does it consist of?
mechanoreceptor found in the skin
responds to pressure/vibrations
consists of the end of a sensory neurone wrapped in layers of connective tissue
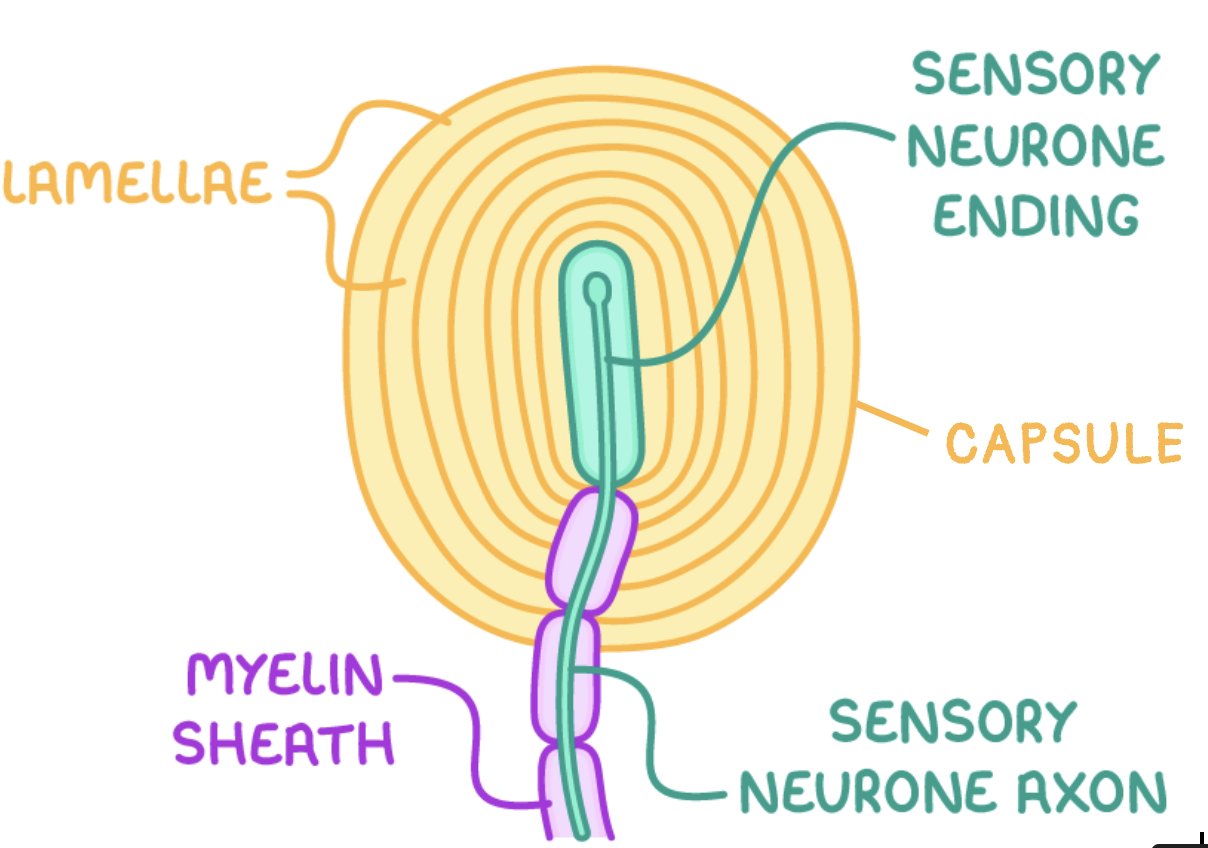
what are stretch-mediated ion channels? what do they respond to?
ion channels present in Pacininian corpuscles - Na+
respond to mechanical forces along the plane of the cell membrane (membrane tension) but not to hydrostatic pressure perpendicular to it
describe what happens when the Pacinian corpuscle is stimulated:
pressure causes the lamellae to become deformed
increase in pressure deforms the stretch mediated Na+ ion channels in the sensory neurone’s plasma membrane
Na+ ion channels in membrane open
Na+ ions diffuse in, depolarising the nerve ending
this leads to a generator potential, which if the threshold is met, generates an action potential
the increase in pressure causes more Na+ channels to open so more Na+ ions can enter
give the order of connections between photoreceptors and the CNS:
rods and cones (synapses) bipolar neurones → ganglion cells → optic nerve → CNS
what are the 3 types of cone cells?
red sensitive cone cells
green sensitive cone cells
blue sensitive cone cells
where are the photoreceptors found on the eye? describe and explain their distribution:
in the retina:
rod cells - across entire retina except fovea
cone cells - on fovea
as cone cells only respond to high light intensities and the fovea receives the highest intensity of light as this is where the lens focuses light
this means that rod cells can be located further from the fovea as they can respond at lower light intensities
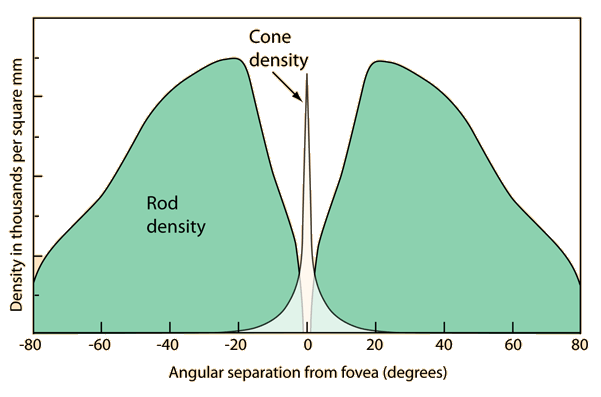
compare and contrast rod and cone cells:
rod cells found across entire retina except fovea, whereas cone cells only found on fovea
rod cells highly sensitive to light, whereas cone cells are less sensitive to light
rod cells can only generate B&W images, whereas cone cells can generate images in colour
rod cells provide low resolving power, whereas cone cells provide a higher resolving power
the optical pigment in rod cells is rhodopsin, whereas the optical pigment in cone cells is iodopsin
how do rods and cones detect light?
light causes the chemical breakdown of optical pigment inside rods and cones
change in membrane potential causes Na+ to diffuse in, establishing a generator potential
if the generator potential reaches the threshold then an action potential is sent along a bipolar neurone to the optic nerve
can rod cells detect light at very low intensities? why?
yes! retinal convergence - many rod cells connect to 1 bipolar neurone so the threshold is more likely to be met as spatial summation occurs
do rod cells have a high or low visual acuity (resolving power)?
low: many rod cells connect to 1 bipolar neurone - the brain is unable to distinguish between separate light sources as multiple signals are sent to the brain
do cone cells have high or low visual acuity (resolving power)? why?
high:
1 cone cell connects to 1 bipolar neurone, sending separate signals to the brain
this allows points close together to be distinguished
can cone cells detect light at very low intensities? why?
no:
cone cells connected to 1 bipolar neurone
threshold unlikely to be reached to produce an action potential

after the student had stared at a purple square, he saw a green afterimage - suggest why (3)

describe how the pupil constricts:
in the iris:
circular muscles contract
radial muscles relax
decreasing the amount of light that can enter the eye
describe how the pupil relaxes:
in the iris:
circular muscles relax
radial muscles contract
increasing the amount of light that can enter the eye
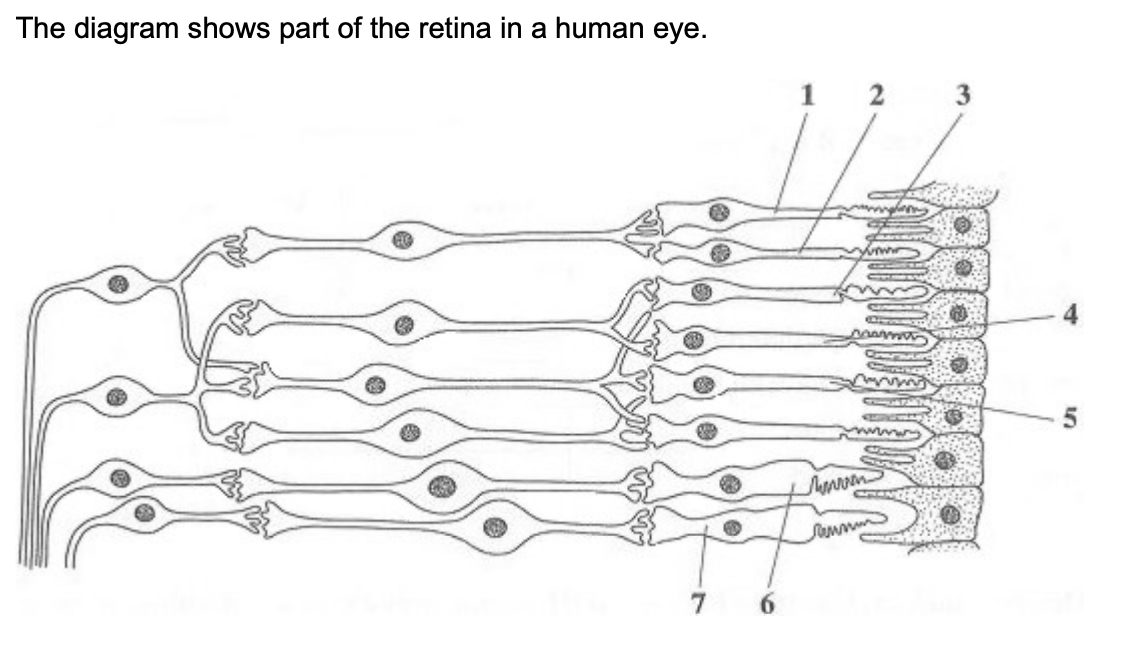
when one unit of light energy falls on cell 3, no light is seen. but when one unit of light energy falls on cell 3, one unit falls on cell 4 and one unit falls on cell 5, light is seen - explain why (3)
1 unit is sub-threshold/3 units are above threshold/give sufficient depolarisation
(1 unit) no impulses/no action potential/in (sensory) neurone/does not stimulate (sensory) neurone/3 units → impulses;
spatial summation/sufficient neurotransmitter released from 3 receptors/insufficient neurotransmitters from 1

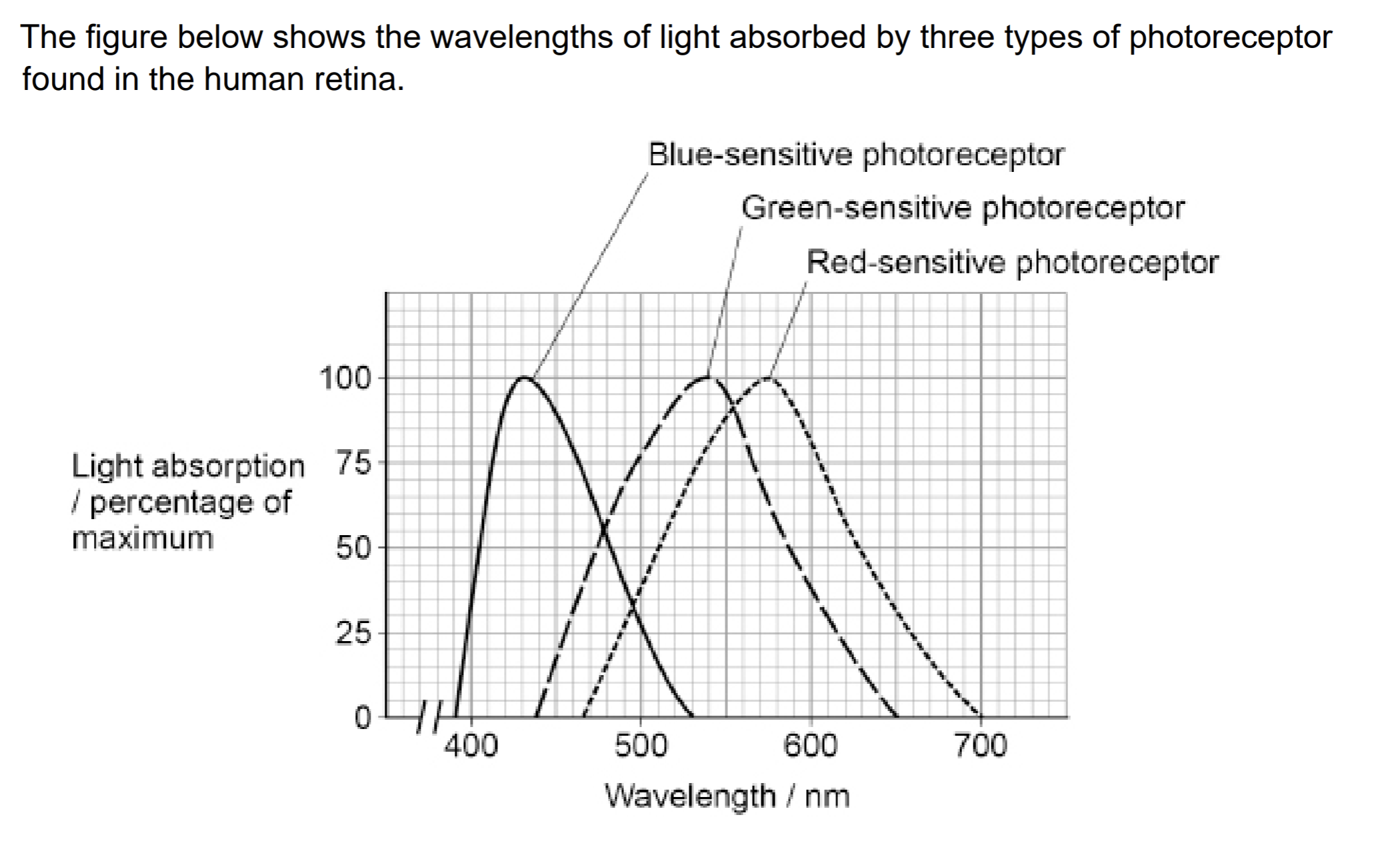
use the figure and your knowledge of colour vision to explain how an orange colour is seen at 600 nm (3)
colour vision involves cones
each photoreceptor has diff pigment/absorbs diff wavelengths
80% max absorption

describe how an action potential is generated at a neuromuscular junction:
arrival of action potential at presynaptic neurone causes Ca2+ channels to open and Ca2+ to diffuse in
vesicles containing acetylcholine fuse w/ the presynaptic membrane
acetylcholine is released into the synaptic cleft and diffuses across
acetylcholine binds to specific receptors on the sarcolemma
Na+ diffuse in → depolarisation
Ca2+ released by sarcoplasmic reticulum
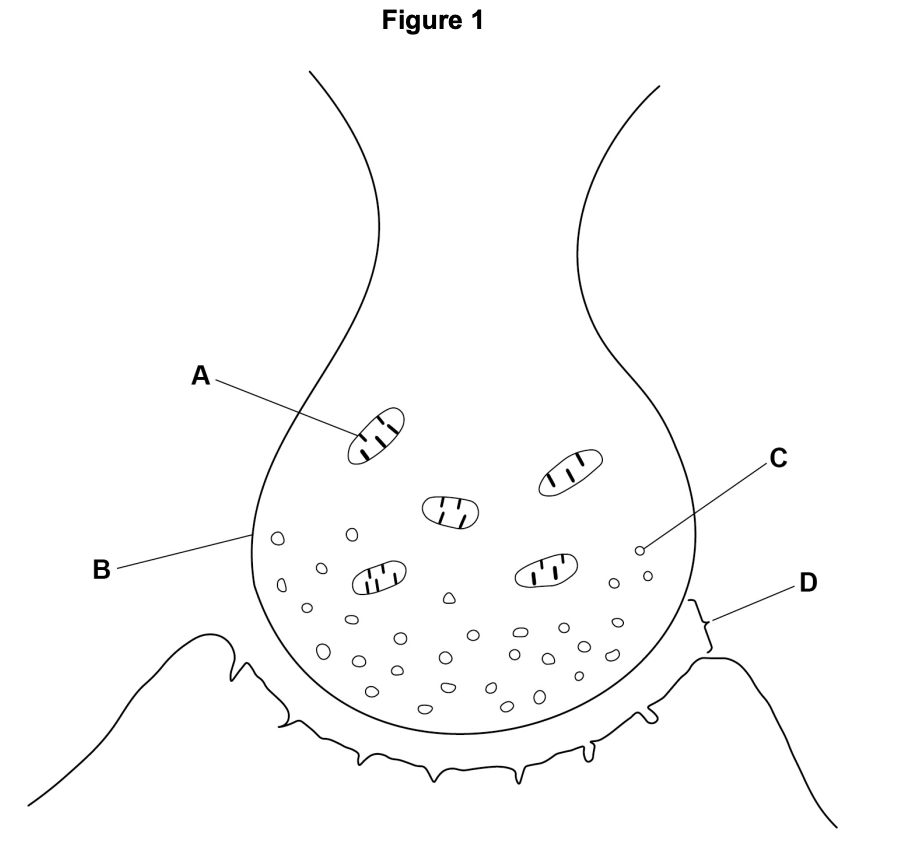
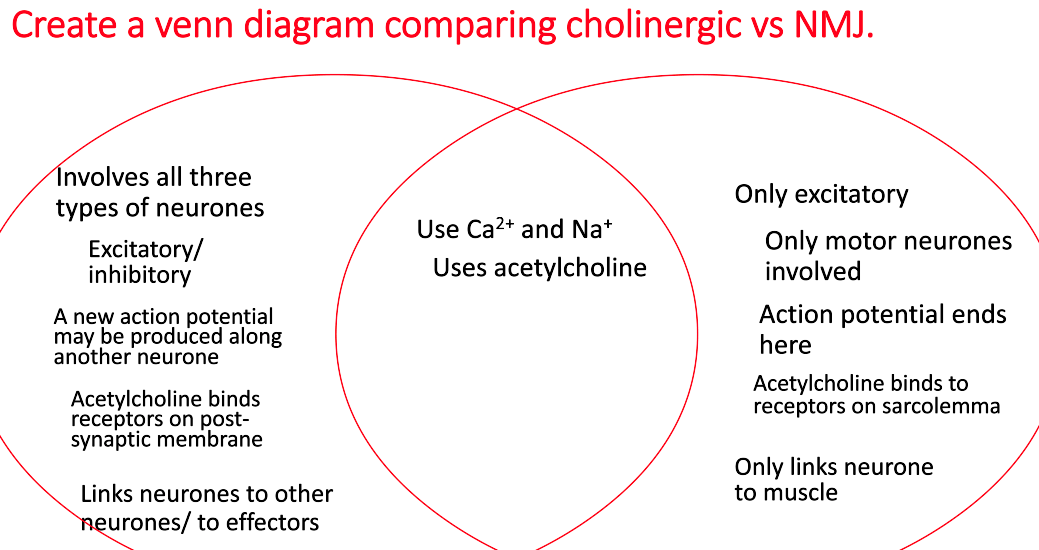
give the similarities and differences between an action potential arriving at a cholinergic synapse and a neuromuscular junction:
similarities:
both involve Ca2+ and Na2+
both use acetylcholine
differences:
all 3 neurone types involved in cholinergic whereas only motor neurones involved in neuromuscular junction
cholinergic can be excitatory/inhibitory but neuromuscular junction is only excitatory
cholinergic may produce a new action potential along another neurone but action potential ends at the neuromuscular junction
cholinergic - acetylcholine binds to receptors on postsynaptic membrane whereas after neuromuscular junction - acetylcholine binds to receptors on sarcolemma
cholinergic links neurones to other neurones/effectors but neuromuscular junction only links neurone to muscle