PHARM ANA LEC FINALS 1.2
1/118
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
119 Terms
regulated the development,
manufacture and marketing of medical products by a number of laws and
guidelines to assure the safety, protection and well being of the patient.
Government health authorities
based on a correctly implemented system of
Quality Assurance incorporating ___ and ___ which involves performing various tests and analysis to elucidate
conformance to set standards and specifications.
Authorized manufacture
Good Manufacturing Practice
Quality
Control
broad term which includes
suitability of drugs and products for their utilization which is decided by
their efficiency and safety, according to label claim, or as promoted or
publicized, their conformity to specifications about identity, purity and
other characteristics.
quality
The quality of any product must be built all throughout the product life
cycle, including the?
testing, inspection, labelling, storage and
distribution.
It is part of Good Manufacturing Practice
● Involves operational techniques and activities that are used to fulfill
requirements for quality
● Aims to eliminate errors and production of end product of given
specifications
● In-Process Quality Control (IPQC) & Finished Product Quality Control
(FPQC)
QUALITY CONTROL
Monitor and adaptation of manufacturing processes in order to comply
to the given specifications
● Should be carried out before the manufacturing process is complete
● Physical parameters and its quality attributes, and may involve control
of equipment and environment
IN PROCESS QUALITY CONTROL
Qualitative and Quantitative Analysis of Product
● Determines the test and procedures and acceptance criteria/limit
● The product must comply with the acceptance limit for the approval of
complete batch manufactured.
FINISHED PRODUCT QUALITY CONTROL
IDENTIFICATION TESTS?
Particle size
● Colour and odor
● Unique identification marking (e.g. emboss, engraving, printing)
measured using a vernier caliper
Thickness
measured using a specialized tool (monsanto type)
Hardness tester
affected by compression of tablet and compressive
force, and method of granulation.
Hardness
physical condition that describes the tendency of tablets to
break into smaller pieces or to detach a percentage of powder or
powder loss from the tablet’s outer surface under mechanical and
physical stress. Capsules do not undergo this test.
Friability
Friability equipment
Roche Friabilator
Vanderkaamp friabilator
Friability Standard setting
25 rpm for 4 minutes (100 revolutions)
Friability samples condition
650 mg or greater - 6.5g dedusted sample
< 650 mg - 20 tablets
Friability formula
% Friable = [(initial weight - final weight/initial weight) x 100
Friability acceptance criteria
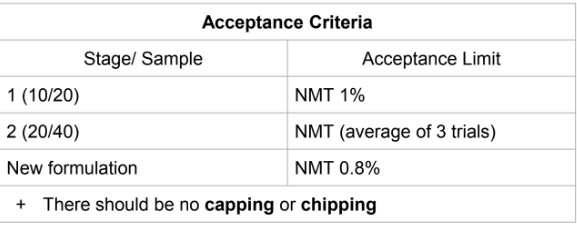
Separation of the
crown of the tablet
Capping
Splitting of the tablet
into layers
Lamination
Breaking of edges
Chipping
mechanical break up of
compressed tablets into smaller particles
Disintegration
Disintegration equipment
Basket Rack Assembly
Disintegration requirements
Six (6) cylindrical tubes; 10 mesh wire cloth at bottom portion and
disks
Disintegration temp
37 ± 2 oC; 29-32 cycles/min
medium and acceptance criteria of Plain, uncoated tablet
Sugar coated
Capsules
Distilled Water30 minutes
medium and acceptance criteria ofEnteric coated tablet
Distilled water (5 mins) | (None)
● Simulated Gastric Juice TS
pH 2.5 (1 hour) | (None)
● Simulated Intestinal Juice
TS pH 6.8 (1 hour) | (1 hour)
medium and acceptance criteria of Buccal tablet
Distilled Water
4 hours
medium and acceptance criteria of Sublingual tablet
Distilled Water
3 minutes
medium and acceptance criteria ofChewable tablet
None
None
process in which a substance forms a solution, and
an important parameter to assess bioavailability and therapeutic
effectiveness. Measures the extent and rate of
solution formation from a dosage form.
Dissolution
Dissolution media temp
35 ± 0.5 oC
Dissolution Aliquot
Midway from surface of the medium; 1cm from vessel wall.
Apparatus I in dissolution
Basket
Apparatus II in dissolution
Paddle
Apparatus III in dissolution
Reciprocating cylinder
Apparatus IV in dissolution
Flow through cell
Apparatus V in dissolution
Paddle over disk
Apparatus VI in dissolution
Revolving cylinder
Apparatus VII in dissolution
Reciprocating holder
Height and inside diameter (mm) of dissolution test vessel 1 L
160 - 210
98 - 106 mm
Height and inside diameter (mm) of dissolution test vessel 2 L
280 - 300
98 - 106 mm
Height and inside diameter (mm) of dissolution test vessel 4 L
280 - 300
145 - 155 mm
Dissolution test formulas

Dissolution test acceptance criteria
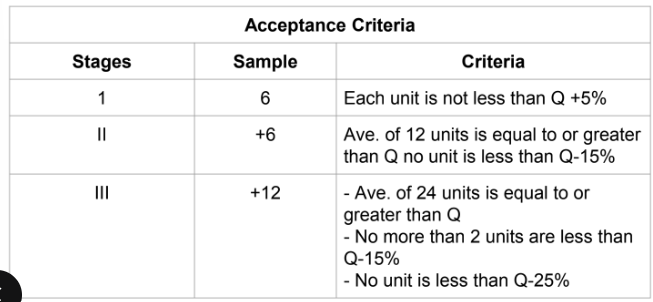
degree of uniformity in the amount of drug
substance among dosage units ( dosage forms containing a single dose or
a part of a dose of drug substance in each unit)
Uniformity of dosage units
a method used to determine drug content
uniformity of drug distribution (based from USP XX).
Weight Variation Test
formula of Weight Variation Test
Ave. weight = Total wt. of 20 tablets/ 20
● Weight variation = [(individual weight - Average weight)/ average
weight] x 100
acceptance criteria of weight variation test
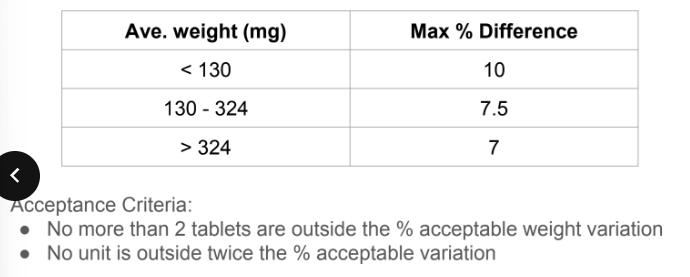
uniformity of dosage forms

content uniformity old acceptance criteria
10 dosage units are assayed individually
and requirements for content uniformity are met if amount of active
ingredient in each unit lies within the range of 85%-115% of Label
claim, with SD <6%
content uniformity new acceptance criteria
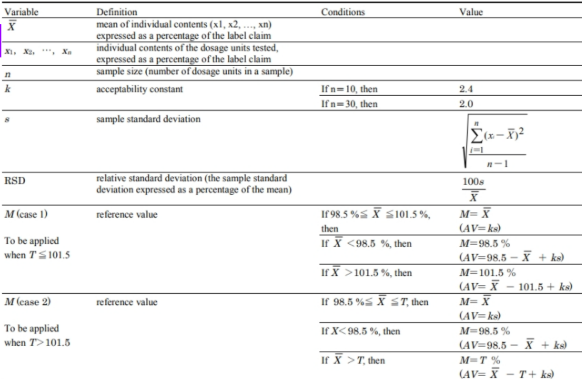
Acceptance Value (AV) calculated must
be less than or equal to L1 (maximum allowed AV; 15) for the first
10 units, if not met, retest.
content uniformity retest acceptance criteria
test 20 more units and the AV calculated must be equal to
L1 for 30 units
uniformity of dosage forms acceptance value formula
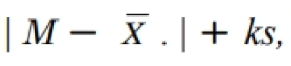
Variable letter symbols
Intermediate of both solid and liquid dosage forms which can be
used therapeutically and cosmetically.
SEMI-SOLIDS
SEMI-SOLIDS examples
Pastes
○ Jellies
○ Ointments
○ Creams
IDENTIFICATION TESTS of semi-solids
Physical appearance
● Texture
● Color – early signs of degradation
● Phase separation and homogeneity – instability
plays a necessary role in the administration of medicated
formulation in a standard dose into the skin
Spreadability
Spreadability is done by spreading ___ of sample between ___ (___times)
1gm
two horizontal glass plates
three
Spreadability types in adults
Face and neck: 2.5
○ Front of trunk: 6.8 (~7)
○ Back of trunk: 6.7 (~7)
○ Hand: 1.2 (~1-2)
○ Arm and forearm: 4
○ Leg and thigh: 5.8 (~6)
○ Foot: 1.8 (~2)
should be calibrated using prepared standard buffer solutions before each use.
pH meter
In pH, 1 gram of each formulation is dispersed in ___, and
the pH is determined with ___ measurements
25 mL deionized water
triplicated
Procedure for non-aerosols (melting range)
Remove any labeling, and clean and dry containers
● Weigh individually
● Remove contents from each container and dry
● Weigh each empty container
● Compute for the next content of each container
○ Net content = initial weight - final weigh
● Compute for the % labeled content
○ % labeled content = net content/ label content x 100
Acceptance criteria of non-aerosols
the % LC of any single container is NLT 90% where the
labeled amount is 60 g or 60mL or less; NLT 95% where the labeled amount is >60
g or 60mL but NMT 150g or 150 mL
Procedure for aerosols
Remove any labeling, and clean and dry containers
● Weigh individually
● Remove contents from each container by employing safe technique (e.g.,
chill to reduce internal pressure, remove the valve then pour)
● Remove any residual contents with suitable solvents, then add a few
portions of methanol
● Retain as a unit the container, the valve, and all associated parts, and heat
them at 100°C for 5 minutes, and then cool and weigh the empty container
● Compute for the net content and %LC
acceptance criteria for the Procedure for aerosols
Net content is NLT labeled amount
METAL PARTICLES IN OPTHALMIC OINTMENTS If for single use
must pass Sterility
METAL PARTICLES IN OPTHALMIC OINTMENTS for multiple use
must pass Antimicrobial effectiveness testing unless it is
bacteriostatic itself.
METAL PARTICLES IN OPTHALMIC OINTMENTS procedure
Extrude all the contents in a petri dish
2. Heat at 85°C for 2 hours and cool
3. Inert petri dish on the stage microscope
4. Examine the petri dish for metal particles
5. Count the number of of metal particles that are 50μm or larger in any
dimensions
6. Acceptance criteria: total number of particles in all 10 tubes does not exceed
50, and if not more than 1 tube is found to contain more than 9 such particles.
The presence of certain microorganisms in nonsterile preparations may
have the potential to reduce or even inactivate the therapeutic activity of
the product and has a potential to adversely affect the health of the
patient
● For non-sterile oral/ topical products
MICROBIAL CONTENT
used to measure Total Aerobic Microbial
Count (TAMC), what is the incubation?
Tryptone Soy Agar
30 - 35°C for 2 -3 days
measure Total Yeasts and Molds
Count (TYMC) what is the incubation?
Potato Dextrose Agar
20 - 25°C for 5 - 7 days
microbial content list with specified organisms
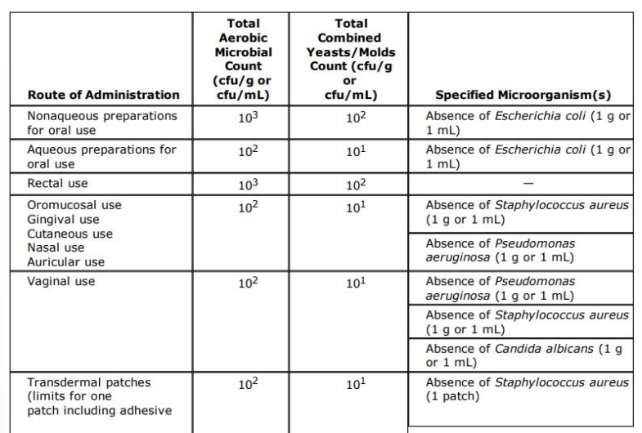
microbial content list with specified organisms p2
Significance of other microorganisms recovered should be evaluated in
terms of the following:
The use of the product; hazard varies according to ROA
● The nature of the product
● The method of application
● The intended recipient
● Use of immunosuppressive agents, corticosteroids
● The presence of disease, wounds, organ damage
pourable pharmaceutical formulations which
contain a mixture of active drug components and nondrug components
(excipients) dissolved or suspended in a suitable solvent or mixtures of
solvents.
● These preparations designed to give the max. therapeutic response
and produce rapid therapeutic effects.
Liquid dosage forms
visualization or using a homogenizer in liquids
Homogeneity Test
tests in liquid
Homogeneity Test
Viscosity and Specific Gravity Test
Leaker test
pH considerations in liquids
The effect on the body when the solution is administered
○ The effect on stability of the product
○ The effect on container-closure system pH measurement
(often used for ampoules and vials)
○ Test container is immersed in a dye bath. Pressure and vacuum is
applied for some time. From the dye bath the container is removed
and washed. The container is then analyzed for the presence of
dye either by means of UV-Spectroscopy or visually.
Leaker test or Dye bath test
provide assurance that oral liquids will, when
transferred from the original container, deliver the volume of dosage
form that is declared on the label of the article
Delivered Volume
Delivered Volume tests are applicable to products labeled to contain NMT
___, whether supplied as liquid preparations, or liquid
preparations that are constituted from solids upon the addition of a
designated volume of a specific diluent
250mL
Sample size in liquids
10 containers initially; 20 containers on retest
Delivered Volume
Procedure:
Gently pour the contents of each container into separate dry
graduated cylinders of a rated capacity not exceeding two 1/2 times
the volume to be measured, and calibrated to contain. Avoid the
formation of bubbles in doing this.
● Allow each container to drain for a period not to exceed 30 minutes
for Multiple-unit containers, and 5 seconds for Single-unit
containers
Sedimentation volume in suspension
settled volume / total or initial volume of suspension
Ideal SV
1
measurement of repulsion between particles
Zeta Potential Determination
Zeta Potential Determination is Related to the electrostatic forces which could affect the attraction
(aggregation) and/or repulsion of
colloids
Zeta Potential Determination is Determined by
microscopic electrophoresis
lead to the
attractive forces exceeding the repulsive forces, creating flocs (loose
agglomerates of particles)
Low zeta potential (or reduced to a certain value)
Degree of Flocculation
β = Ultimate sedimentation volume of flocculated suspension (Vu) /
Ultimate sedimentation volume of DEflocculated suspension
Ease of Redispersibility in suspension
Solute should be 100% redispersible with minimum agitation
Particle Size Determination in suspension
Optical Microscopy
○ Sedimentation Rate - based on Stoke’s Law
should pour readily and evenly from its container
Viscosity
A liquid disperse system that uses small globules of a liquid (dispersed
phase) distributed through another liquid in which it is immiscible
forming spherical bodies called ___.
micelles
EMULSIONS
Emulsifying agents
(gum arabic, lecithin, carrageenan, CMC,
polysorbate)
Based on nature of dispersed phase:
Oil in water (O/W)
○ Water in oil (W/O)
Based on globule size of emulsions
Microemulsons (0.01 um)
○ Fine emulsions (0.25 to 25um)
Tests for types of emulsions
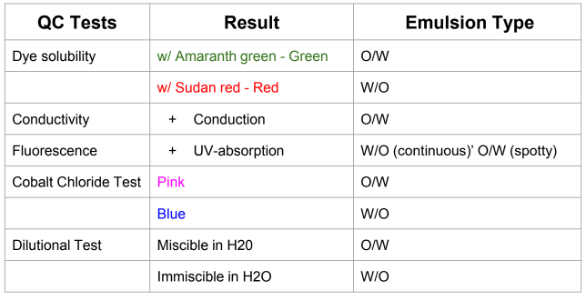
Test for creaming
Upward creaming (O/W); Downward creaming (W/O)