semi-microscale Amide Synthesis of Phenacetin Lab 9
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
mechanism of the formation of phenacetin
acetic anhydride gets protonated and then p-phenetidine will come and the amine group will come and attach to the carbonyl carbon, causing the electrons to get pushed up to the oxygen and make it an alcohol. Now the NH2 has a positive charge but then the second carbonyl from the acetic anhydride will come and take it and now that oxygen is protonated. The lone pair from the alcohol of the acetic anhydride will push down and cause the other half of the anhydride to leave and then the electrons will get pushed up to the carbonyl and make it neutra again. Then we get acetic acid and phenacetin.
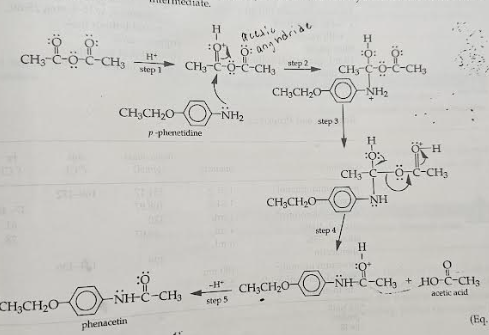
how to control the pH of the solution
so when the acetic anhydride gets protonated the pH of the solution might get too low, so then the p-phentidine will get protonated and turn to its conjugate acid. So to combat this a buffer of acetic acid and its conjugate base sodium acetate is added to control the pH.
what was the reaction
activated carbon was added to the solution of p-phenetidine and HCl to decolorize it. Sodium acetate was added to the p-phenetidine and HCl solution to be turned into the conjugate acid of acetic acid and perform as a buffer solution. Acetic anhydride was then added and precipitate formed.
what is the value of doing a mixture melting point from the phenacetin of williamson synthesis and aide synthesis
With a mixture melting point it can be observed if the reaction went properly and if both are the same product. If the melting point is lower than expected then something is impure.