Water Pollution W1
1/24
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Ecologists’ view
Intro of foreign materials into env should be kept at a level which no detectable biological change results
Economists’ view
Discharge/other action which occasions economic damage to man
USA Env protection agency definition
“Any substances in water, soil, or air that degrade the natural quality of the environment, offend the senses of sight, taste, or smell, or cause a health hazard. The usefulness of the natural resource is usually impaired by the presence of pollutants and contaminants”
Characteristics of pollution events
The pollutant
The source of the pollutant
The transport medium (air, water, soil)
The target (organism, ecosystem)
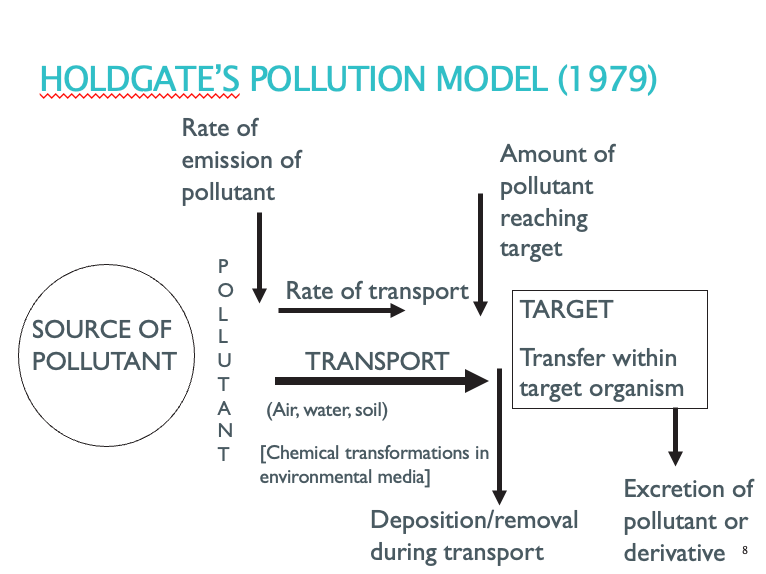
Holdgate’s pollution model (1979)
Notes:
No reason why pollutant can’t travel through air/water/soil at some point.
Chemical transformations may amplify/reduce impact.
Not all pollutants reaches target (deposition).
Discrete point source
e.g., sewage leak
Easier to identify + control
Diffuse source
e.g., acid rain, fertilisers
Harder to track causes + more likely to be affected by physical/biological factors that make it harder to deal with.
Example sources of pollution
Agricultural, e.g., fertilisers
electricity generation e.g., water vapour greenhouse gases
Derelict gas works
Metal Mining e.g., relicts on landscape, uses a lot of water + draining releases lots of metals.
Metal industries e.g., atm pollutants
Urban + industrial sources e.g., litter, car tyres, vehicle exhausts
Waste disposal + transport e.g., spillages
Effect generating properties
Toxicity e.g., consumption affects organisms
corrosion of metals e.g., impact on building from acid rain
Pathway determining properties
Affects distance + rate of dispersion in env.
e.g., organic material dissolved in water is controlled by its solubility
Primary pollutants
exert harmful effects in form in which they enter the environment
e.g., nitrates from fertilisers
secondary pollutant
pollutants are synthesised as a result of chemical processes in the env, often from less harmful precursors
e.g., ozone , which is formed when hydrocarbons and NOx combine in sunlight
Advection
(gravity) - movement with water mass
Diffusion
(turbulence + particle motion) - spreading out of material in water mass
Dispersion key factors
vertical movement restricted by:
depth
stratification
inputs without loss lead to accumulation of pollutants in:
lakes + seas
Lakes + sea sediments
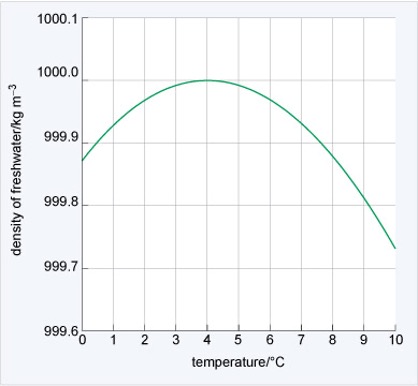
Stratification
density of water depends on temperature
—> affects where pollutants sit in water
During stratification there is a limited exchange of water between strata.
Seasonal stratification
Winter
less dense cold water over warmer, more dense water
Spring
wind mixing plus low solar heating means less stratification
Summer
solar warming makes surface water less dense than deeper, colder water
Autumn
wind mixing plus low density difference, low solar heating
Epilimnion
Upper strata
Hypolimnion
Lower strata
(have less access to oxygen)
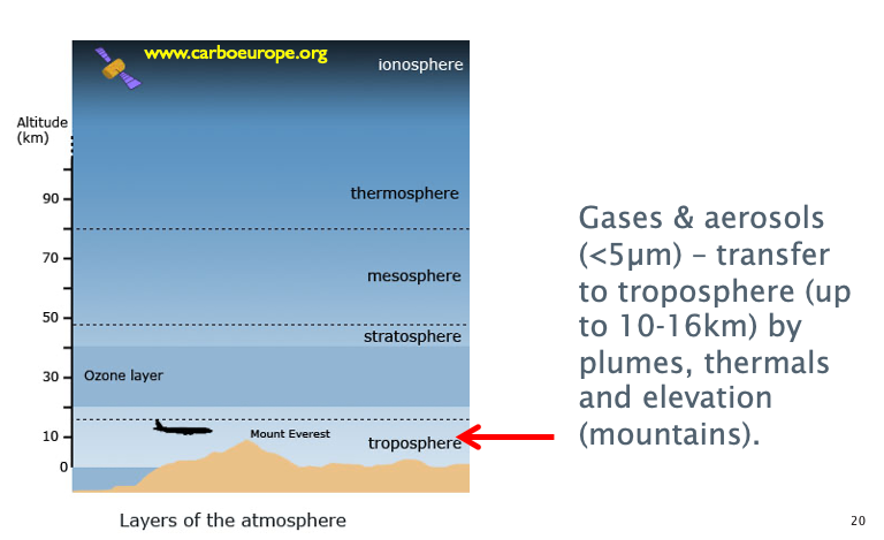
Air pollution
Move air movement at higher boundaries - so pollutants higher up will experience diffusion + advection faster.
There is some exchange between troposphere + stratosphere at equator.
Gases in stratosphere don’t wash out unless degraded by strong radiation above ozone.
Dispersion in horizontal plane
Generally less restricted dispersion.
Transport tends to be with wind in turbulent boundary layer.
Turbulence + thus dispersion is controlled by solar radiation, wind speed, cloud cover + topography (roughness of landform).
Important factors in soil pollution
soil composition + structure (wet, dry, anaerobic, aerobic, acid etc.)
pollutant properties (polar, non-polar, hydrophobic, hydrophilic, liquid, gas)
Soil properties of reactions
Soil at surface is more organic, so has more reactions.
Bottom soil more mineral.
Dry fields with cracks - rain flows through gaps + doesn’t come into contact with soil. If fields ploughed rain will percolate.
Exposure pathways of pollutants for humans
Air - inhalation
water - consumption of water or ingestion of aquatic organisms
Soil - plants being ingested
Indoor pollution
fabric of building e.g., asbestos
importing substances e.g., lead workers (but windows in these factories means pollutants can be diluted)