GEN CHEM EXAM 5
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
what is a transition metal complex?
A transition metal complex is formed between a metal ion and a ligand
What is a coordination compound?
A coordination compound is a transition metal complex plus any counter ions (with an opposite charge to the complex ion) — these ensure that the compound is neutral
Which configurations are inherently stable?
ones in which all orbitals are filled or in which degenerate orbitals are half-filled
When transition metals form ions, they lose electrons from the _____ orbital before the _____ orbital.
s, d
Monodentate ligands:
ligands that bind to the metal atom via one electron pair
Bidentate ligands:
can simultaneously bind to the metal atom via electron pairs on two different atoms.
Polydentate ligands:
can bind via multiple lone pair
Coordination Number
number of ligands bonded to the metal ion
Different complexes can have the same coordination numbers (number of ligands bonded to central metal atom) but may contain….
mono, bi, or polydentate ligands
when naming coordination compounds, a metal that forms part of the anion (comes second) has what at the end?
-ate
When writing out coordination compound formulas from the name, list the neutral compounds in alphabetical order-_______
by chemical symbol
Diamagnetic:
all paired electrons, total spin of 0
Paramagnetic:
has unpaired electrons
In the presence of ligands, the d-orbitals are NOT_____
degenerate but split into two sets
the energy gap between the e and t orbital sets is known as_____
the crystal field splitting energy
If the plotting between the e and T orbitals is large for a transition metal complex, it is referred to as _______
a strong-field complex
If the splitting between the e and t orbitals is small for a transition metal complex, it is referred to_______
as a week-field complex.
the higher the charge on the metal ion, the larger the____
value of the crystal field splitting energyt
The higher the charge on the metal ion, the more ____ electrons pull, which result sin more repulsion.
tightly
Transition metal complexes are colored because they can absorb electromagnetic radiation in the visible region, resulting in the promotion of an electron from the _____ to the _____ orbitals.
t2g to eg
The wavelength of light absorbed by a transition metal complex depends on the size of the _________
crystal field splitting energy
Diamagnetism:
atom/molecule is repelled by a magnetic field because it has NO unpaired electrons
Paramagnetism:
atom/molecule is attracted to a magnetic field because it HAS unpaired electrons
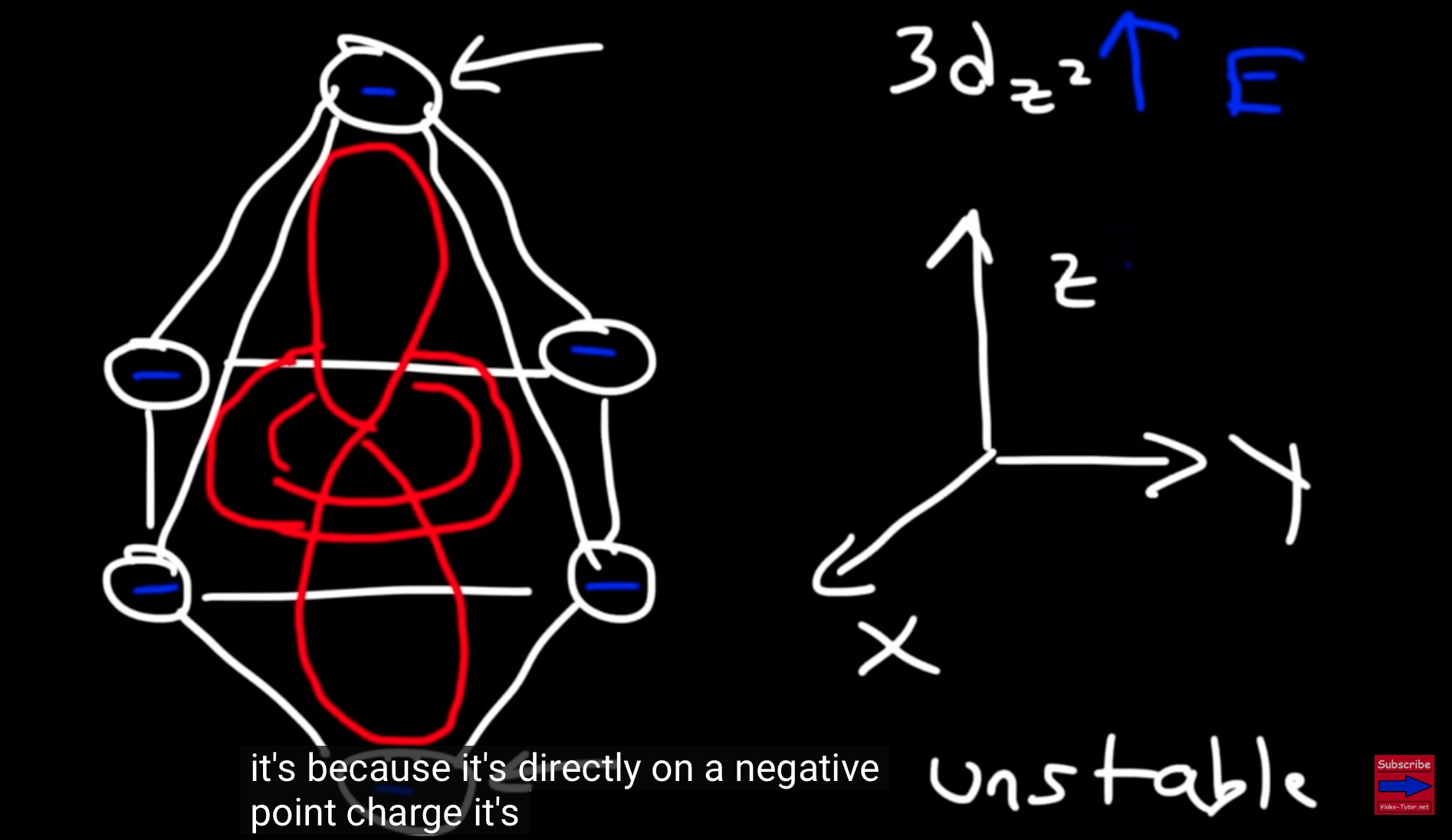
3dz orbital goes up in energy because it is directly on a negative point charge and this is a highly UNSTABLE situation
Hydrocarbons only contain ______
hydrogen and carbon
Carbon makes ____ bonds
4
Alkanes:
only single bonds between C atoms
Alkenes:
one or more double bonds between C atoms
Alkynes:
one or more triple bonds between C atoms
Aromatics:
contain a benzene ring
What are the steps for naming hydrocarbons?
identify the longest chain, the base functional group, and whether it is an alkane, alkene, or a alkyne
Name branches as alkyl groups
number the chain from the end closest to a branch
locate branches on the chain, but list alphabetically in name
count branches, if more than one of the same group present then use di, tri tetra
1 carbon in parent chain:
meth-
2 carbons in parent chain
eth-
3 carbons in parent chain
prop-
4 carbons in parent chain:
but-
5 carbons in parent chain
pent-
6 carbons in parent chain
hex-
–CH3
methyl-
–CH2CH3
ethyl
-CH2CH2CH3
propyl
-CH2CH2CH2CH3
butyl
Suffix indicates the ____
max number of bonds between carbons
Alkane Suffix:
-ane
Alkene suffix:
-ene
Alkyne suffix:
-yne