UF BRUNNIG Nutrition Exam 3
1/339
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
340 Terms
proteins contain?
carbon, hydrogen, oxygen, nitrogen
amino acids are made of?
carbon, hydrogen, amino group, acid group, side group or side chain
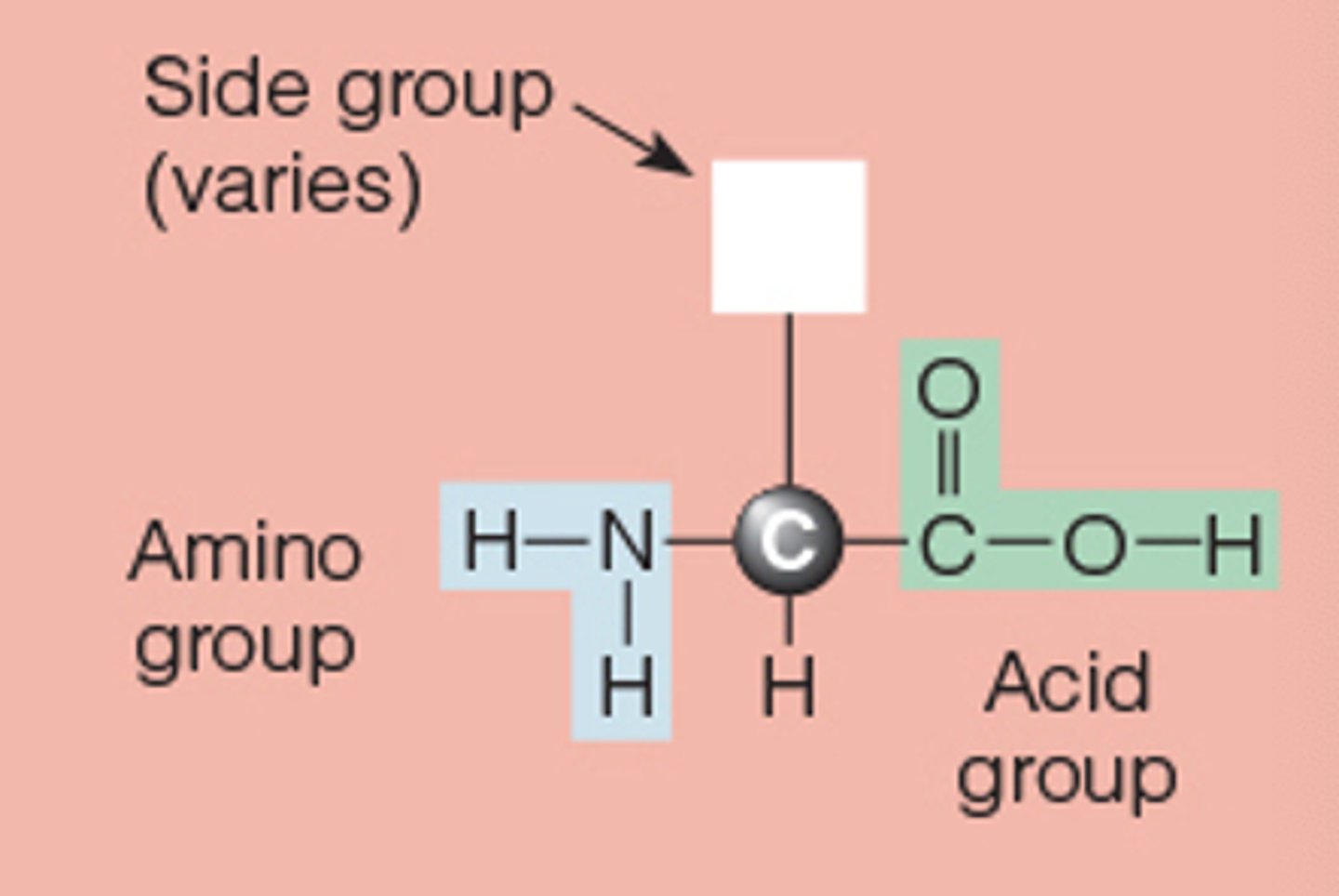
all amino acids have a common
chemical structure
but each amino acid has a different
side group
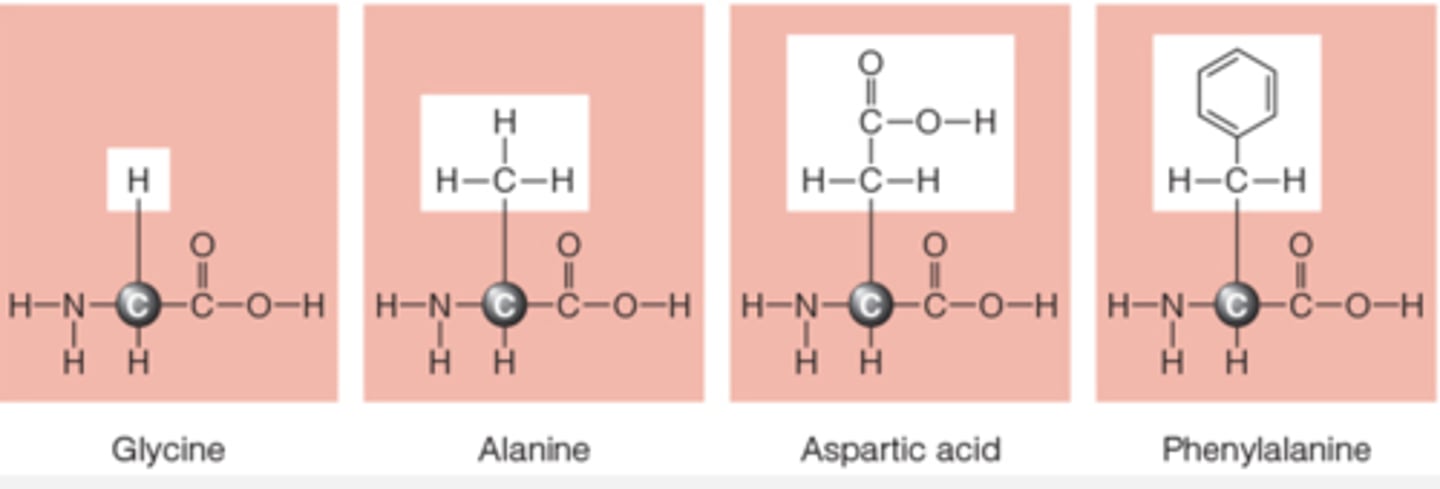
amino acids can be categorized by
essential, nonessential, and conditionally essential
essential amino acids
9
histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, valine
our body must ingest these because we cannot make enough in quanity
non-essential amino acids
11
alanine, arginine, asparagine, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, tyrosine
our body can make these
conditionally essential amino acids
arginine, cysteine, glutamine, glycine, proline, tyrosine
sometimes a nonessential amino acid can become essential
what links amino acids?
covalent peptide bonds created through condensation reactions
Dipeptide
Two amino acids bonded together
tripeptide
three amino acids bonded together
polypeptide
ten or more amino acids bonded together
protein structure
primary, secondary, tertiary, quaternary
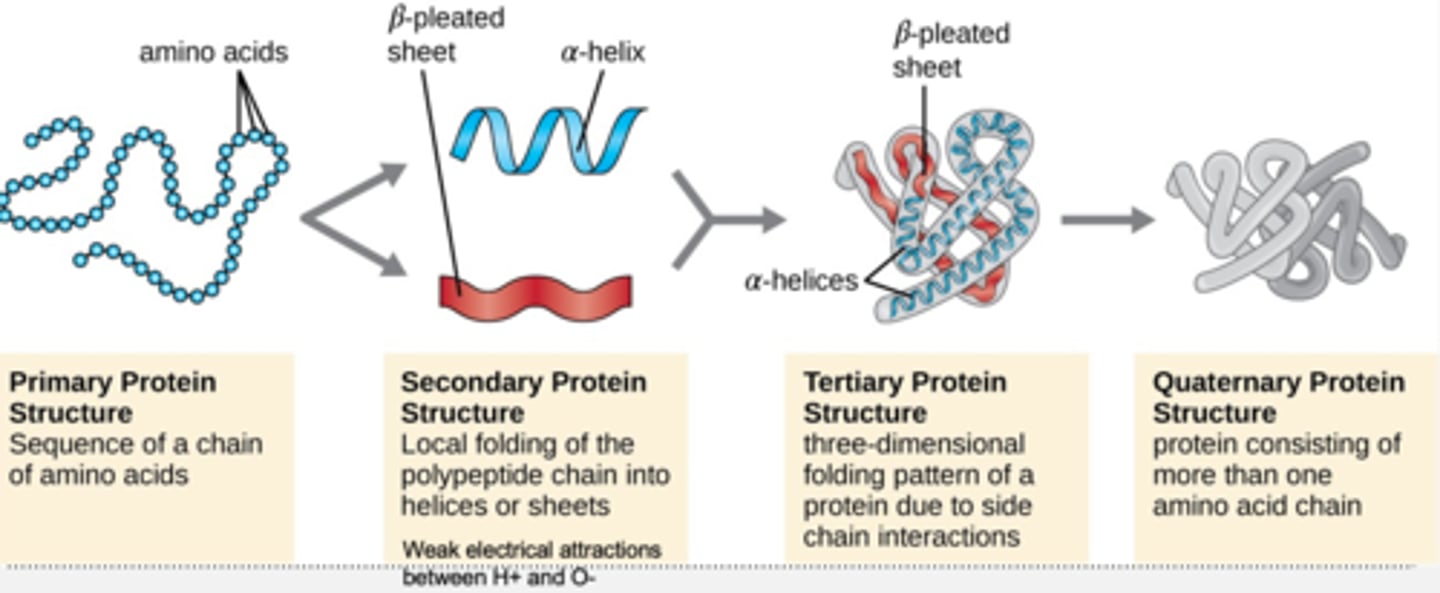
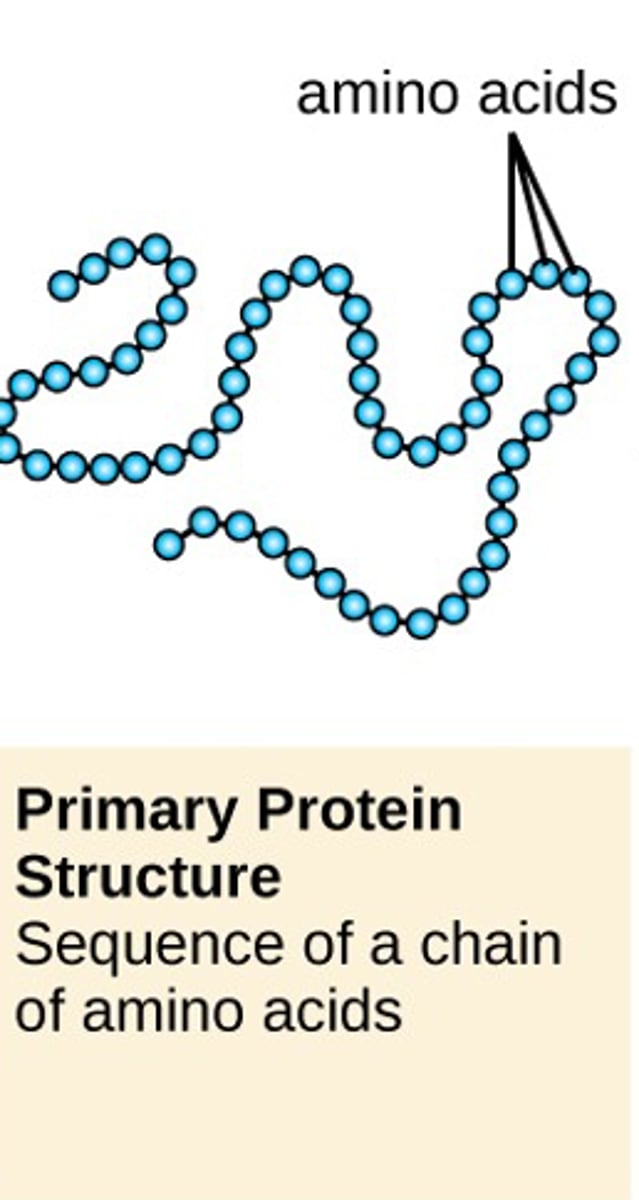
primary protein structure
sequence of a chain of amino acids
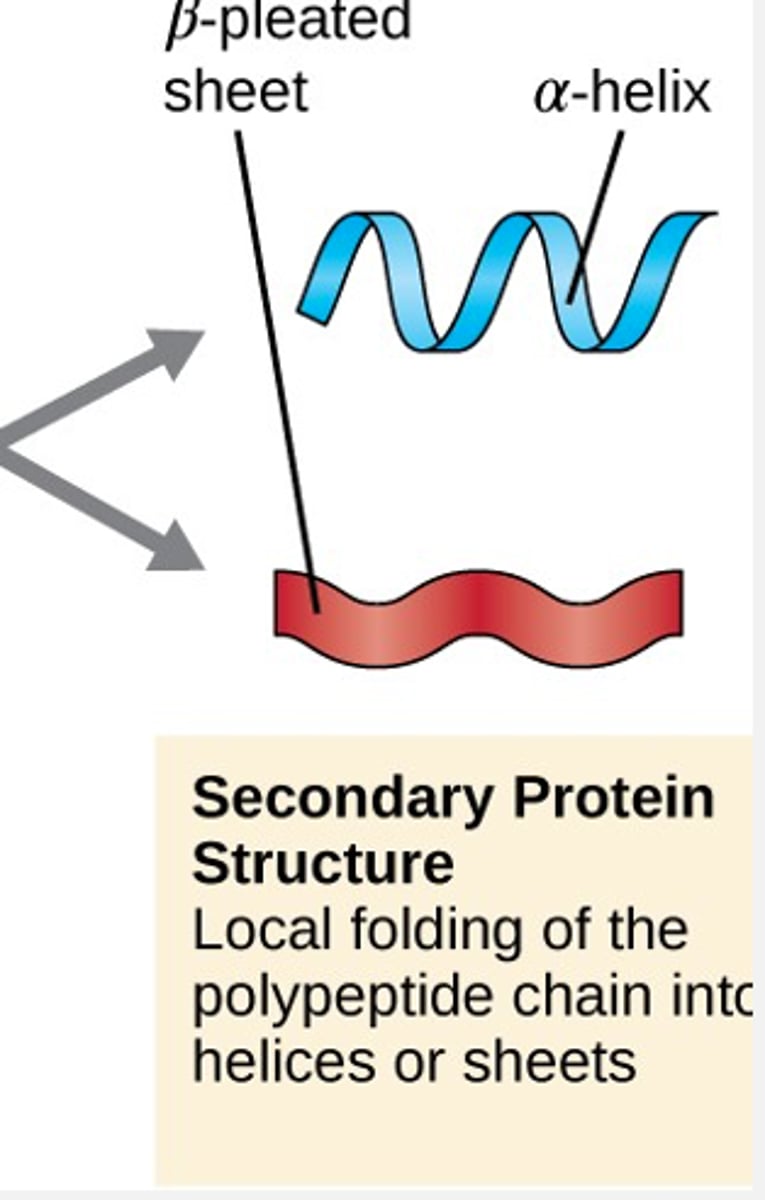
secondary protein structure
local folding of the polypeptide chain into helices or sheets
weak electrical attractions between H+ and O-
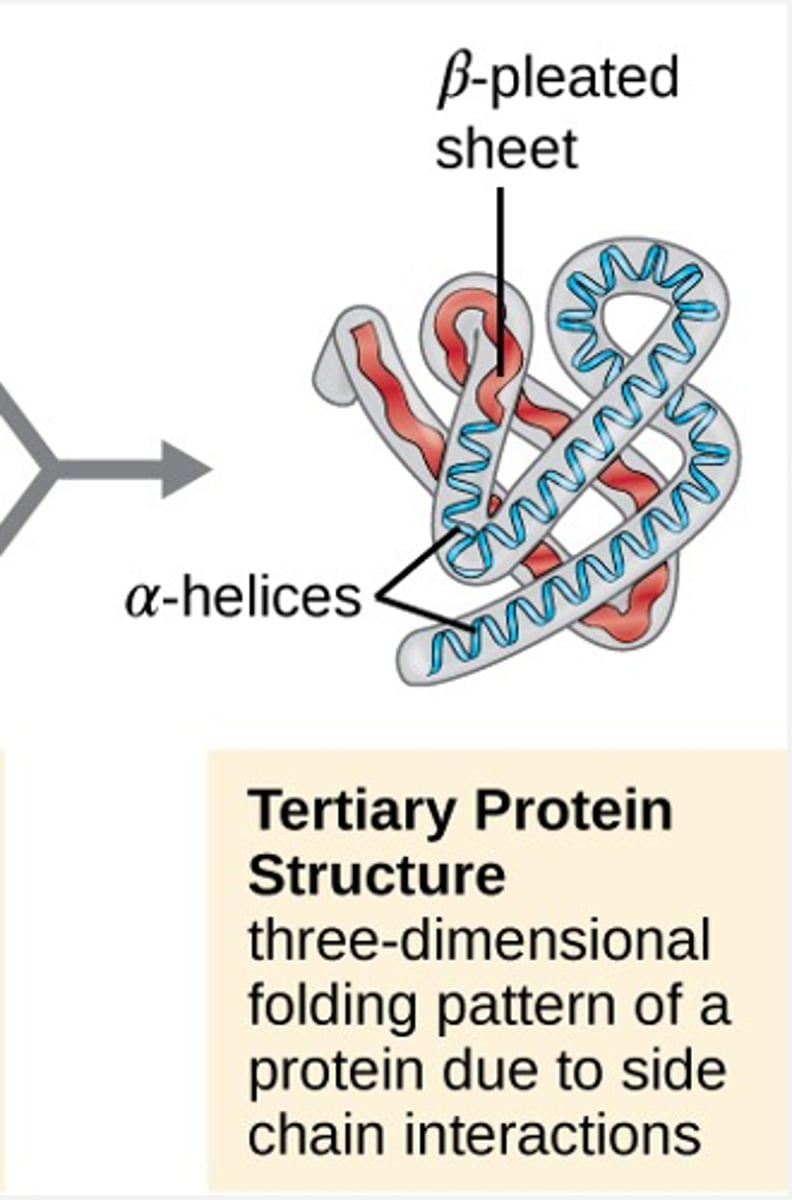
tertiary protein structure
3D folding or coiling pattern of a protein due to side chain interactions
quaternary protein structure
protein consisting of more than one amino acid chain
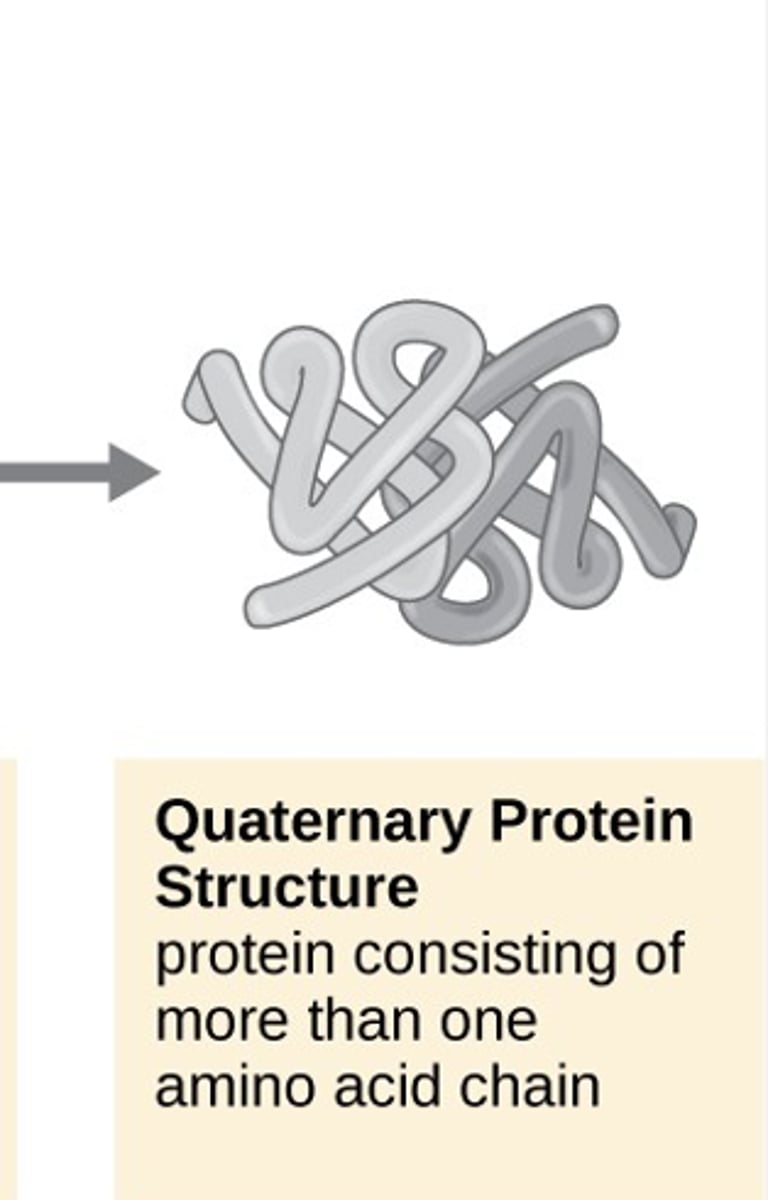
few proteins that reach quaternary structure
hemoglobin, DNA polymerase, ribosomes, antibodies, and ion channels
secondary protein structure can result in
alpha-helixes or beta-pleated sheets
Most proteins stop at the tertiary structure and are functional, but some work in groups of polypeptides called
subunits
hemoglobin always has
two alpha subunits and two beta subunits
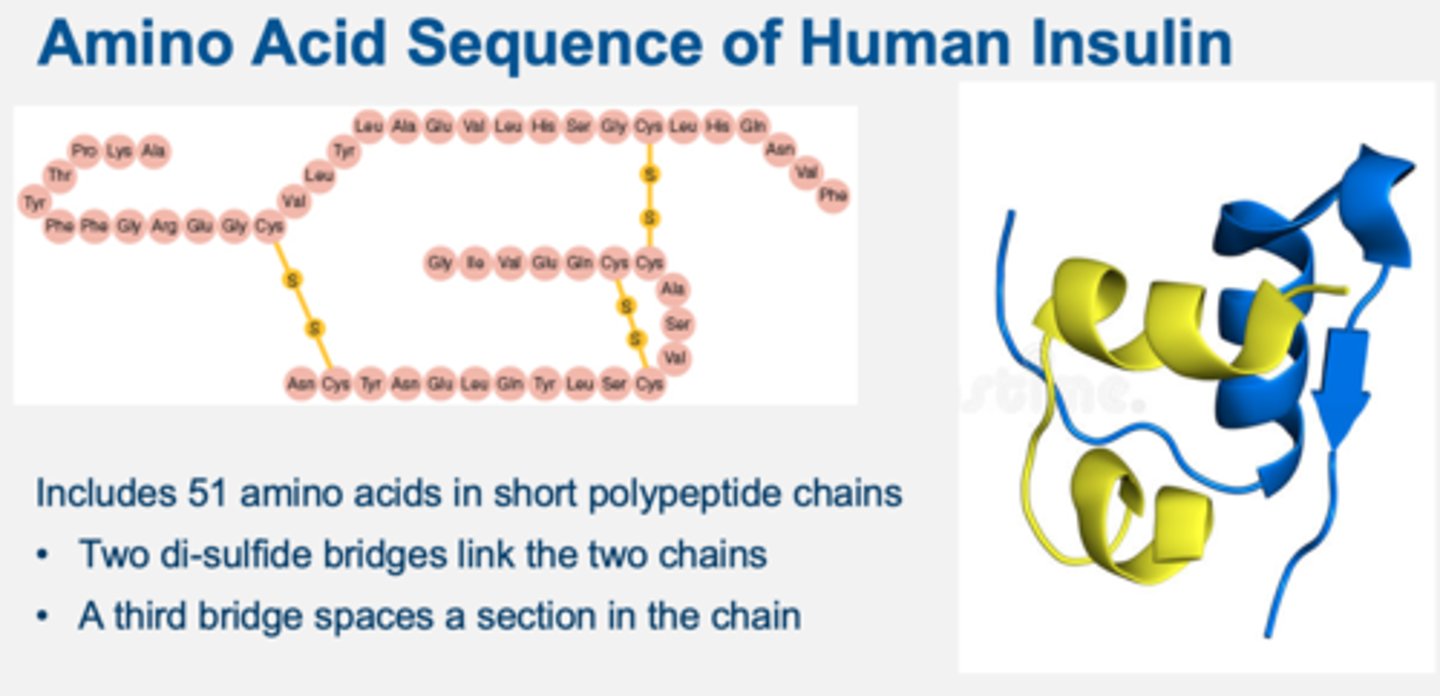
amino acid sequence of human insulin
include 51 amino acids in short polypeptide chain
two di-sulfide bridges link the two chains
a third bridge spaces a section in the chain
a protein can only do its job when its working in its
shape
proteins are often very
complex structures that include folds, loops, and curves
protein denaturation
when proteins are subject to heat, acid or other conditions that disturb their stability; protein uncoils, loses its shape, and loses its function
protein digestion occurs in
mouth, stomach, and small intestine
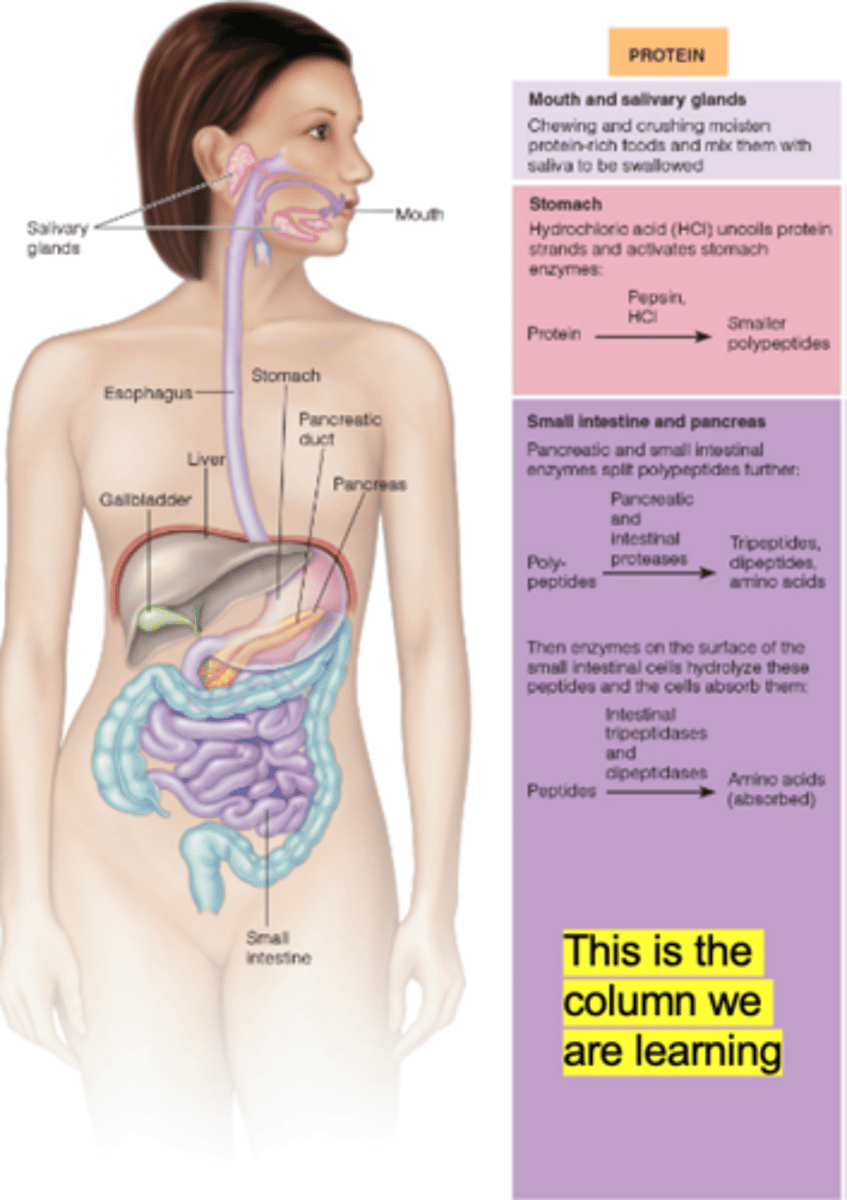
protein digestion in mouth
mechanical processing of protein due to chewing
protein digestion in stomach
hydrochloric acid denatures proteins
pepsinogen to pepsin
pepsin
Enzyme that breaks down proteins into smaller polypeptide fragments
protein digestion in small intestine
fate of pepsin
pancreas and intestinal wall enzymes
amino acids absorbed
What does hydrochloric acid do?
denatures proteins so digestive enzymes can attack the peptide bonds
converts the inactive form of the enzyme pepsinogen to its active form, pepsin
protein absorption is
straightforward
protein absorption transporters
there are about 7 transporters and 20 amino acids so they share transporters
once protein is absorbed
energy in small intestine cells
manufacture proteins in small intestine cells and mucus
rest go to liver
the liver turns amino acids into
glucose
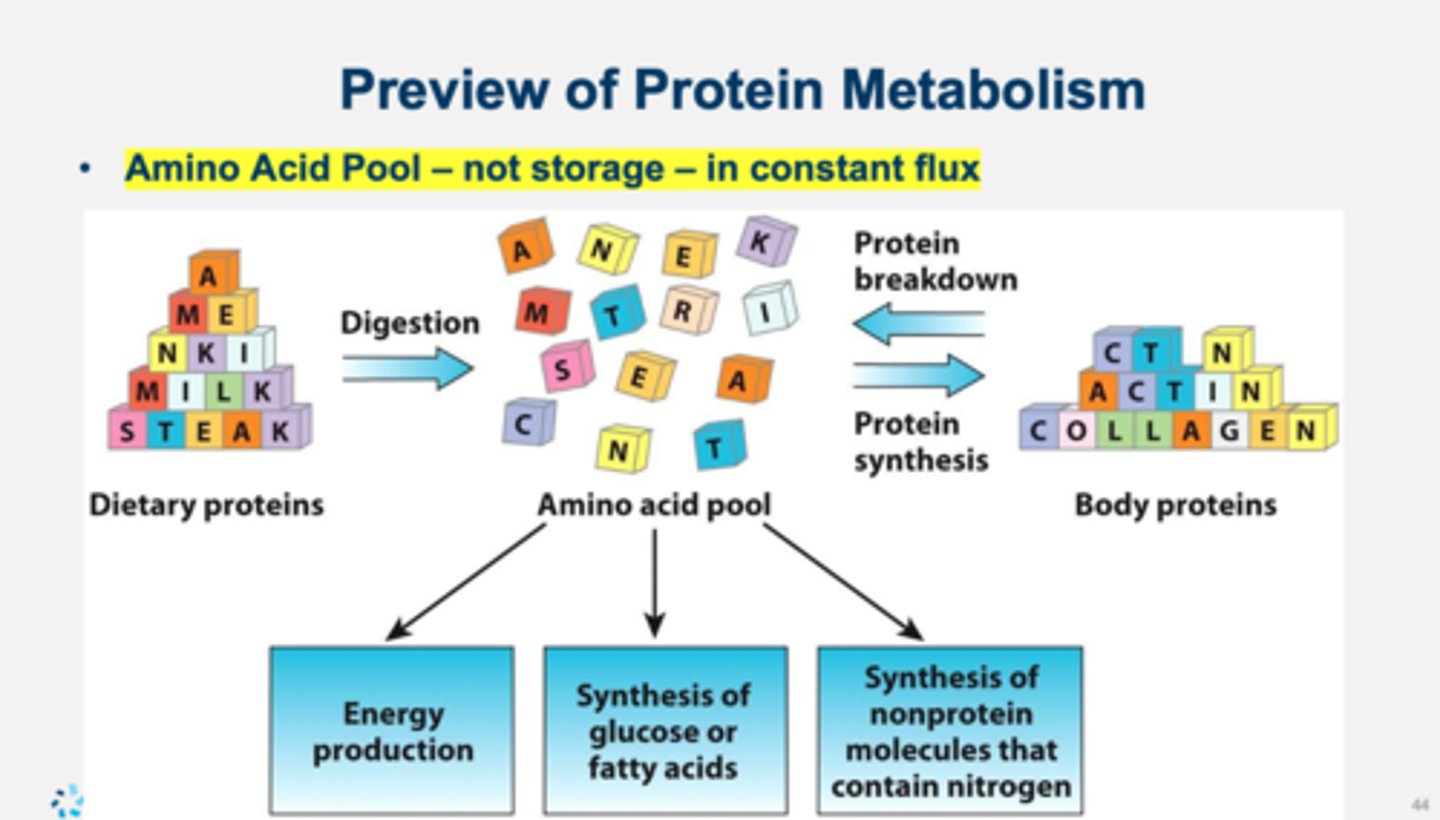
pre-digested proteins are not
useful
we do not store
amino acids (constant flux of pool)
once in the pool amino acids can be used to make more
proteins, ATP, glucose, fatty acids (for storage)
protein synthesis
the formation of proteins by using information contained in DNA and carried by mRNA
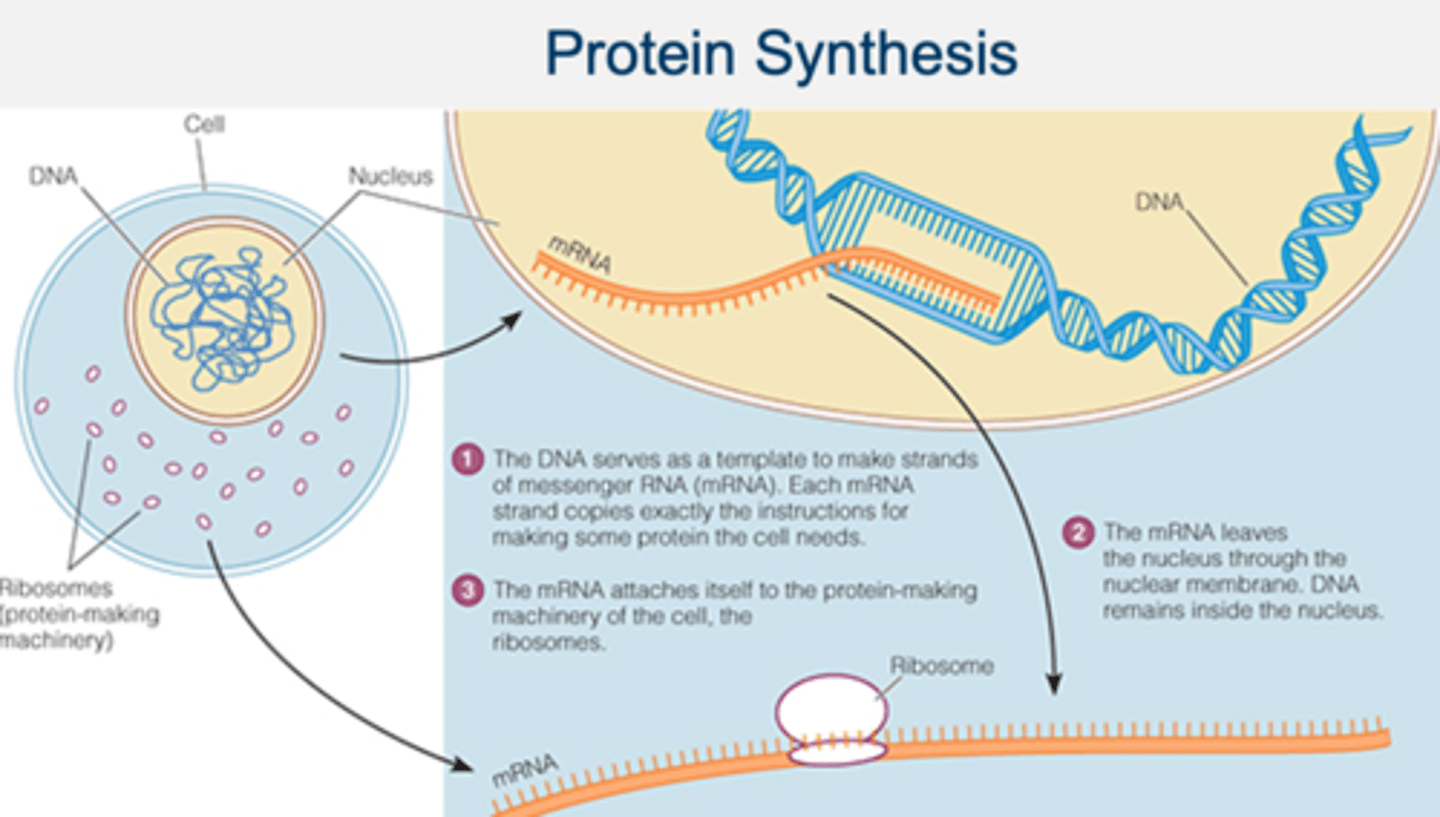
protein synthesis cont.
the process of synthesizing mRNA
transcription
protein-making factory of the cell
ribosome
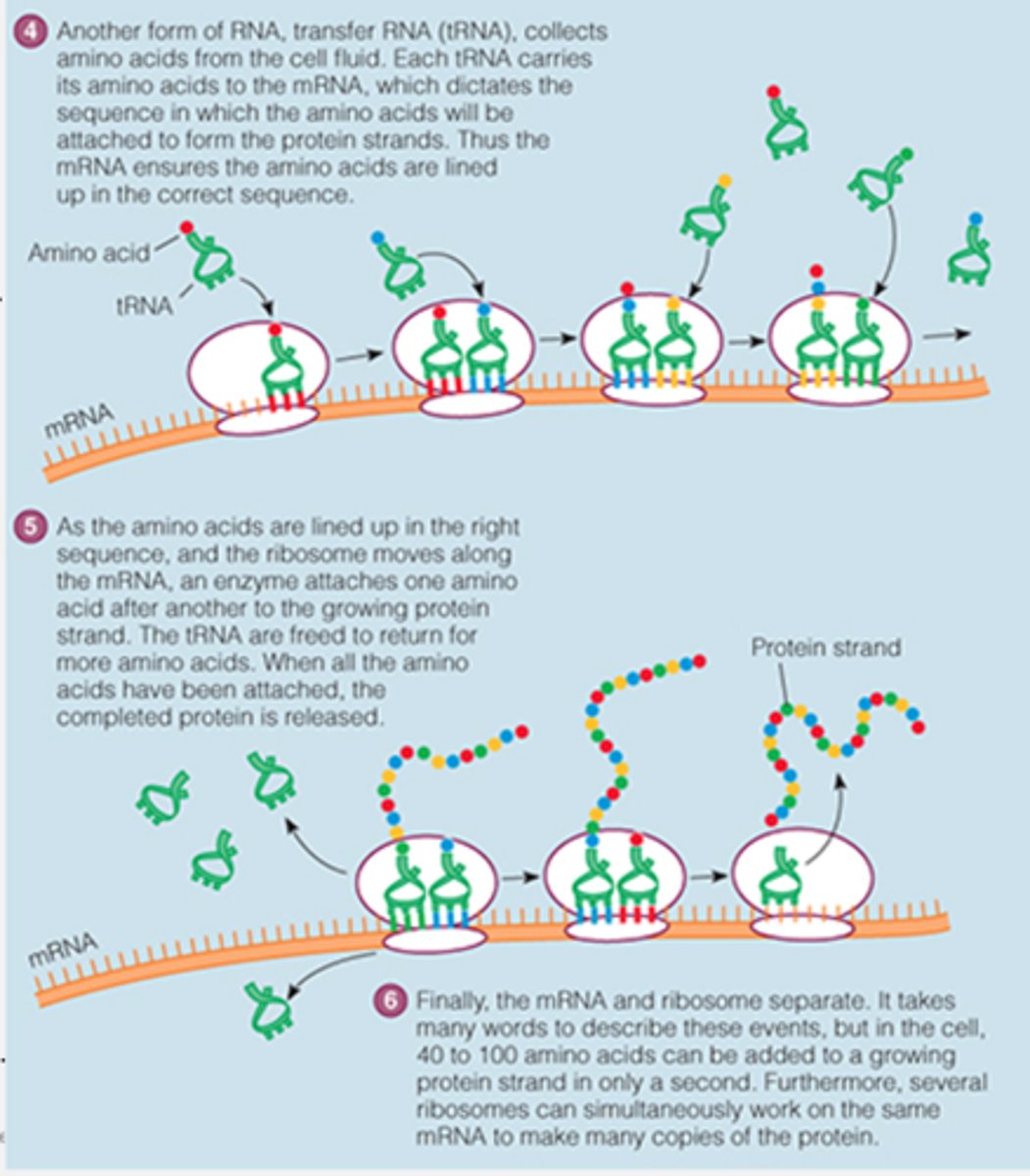
Another type of RNA carries amino acids to the mRNA
tRNA
the overall process of making a protein
gene expression
Process by which mRNA is decoded and a protein is produced at the ribosome
translation
an error in translation, therefore amino acid sequence, alters the final protein
true
the amino acid sequence determines the shape or structure, and this shape determines the
function
sequencing error: sickle cell vs red blood cell
hemoglobin has four polypeptide chains - in sickle-cell two of these chains have a variation
sickle cell anemia
Normally, red blood cells are disc-shaped, but in the genetic disorder sickle-cell anemia, red blood cells are sickle- or crescent-shaped. This difference in shape occurs because valine replaces glutamic acid in the amino acid sequences of two of hemoglobin's polypeptide chains. As a result of this difference in hemoglobin's shape, the capacity to carry oxygen is diminished.
gene expression and protein sysnthesis
capability of body cells
cell only makes proteins it needs
dietary influence on gene expression
nutrients affects your genes
genes affect your nutrients
role of protein
building blocks for most body structures
replacement or dead or damaged cells
enzymes
hormones
regulators of fluid balance
acid-base regulator
transporter
storage
antibodies
source of glucose and energy
collagen matrix
Filled with mineral crystals for bones or teeth
enzyme
a catalyst that speeds up chemical reactions in living cells
protein hormones
messenger molecules
transported in blood to target tissues
oxytocin and prolactin
support lactation
insulin and glucagon
hormones secreted by the pancreas to control levels of blood glucose
thyroxine
regulates metabolism
calcitonin and parathyroid hormone
regulate blood calcium
angiotension, renin, and antidiuretic
regulate fluid and electrolyte balance
protein as a regulator of fluid balance
fluid belongs in cell, between cells and in blood vessels
protein attracts water
edema
puffy swelling of tissue from the accumulation of fluid
protein as an acid-base regulator
body fluid needs right pH
protein acts as a buffer - both attracts hydrogen ions and releases hydrogen ions
blood stays at a pH of
7.35-7.45
protein as an antibody
defend against disease
immunity-memory
protein as a source of glucose and energy
gluconeogenesis
ATP 10%
in times of starvation and insufficient carb intake you use a lot of protein for energy
excess protein creates
fat
protein metabolism
nitrogen balance
the amount of nitrogen consumed compared with the amount excreted in a given time period
nitrogen balance in most healthy adults
0
disease/issues/condition could be indicated by
positive or negative nitrogen balance
when nitrogen intake and output are equal, a person is in nitrogen
equilibrium
infants, children, and pregnant women are in state of
positive nitrogen balance
if protein is being lost and nitrogen excretion is greater than intake, a person is in a state of
negative nitrogen balance
True or false?
Similar to fat and carbohydrates, protein is also easily stored in the body.
False
Proteins are not stored in the body. Recall from the amino acid pool that amino acids and proteins are in a constant state of flux or turnover.
using amino acids to make proteins
if an essential amino acid is missing?
break down lean tissue
skip making protein
using amino acids to make proteins
if a non-essential amino acid is missing?
make it
transanimate
deaminating amino acids produces
urea
deaminating amino acids
to use for energy must remove N
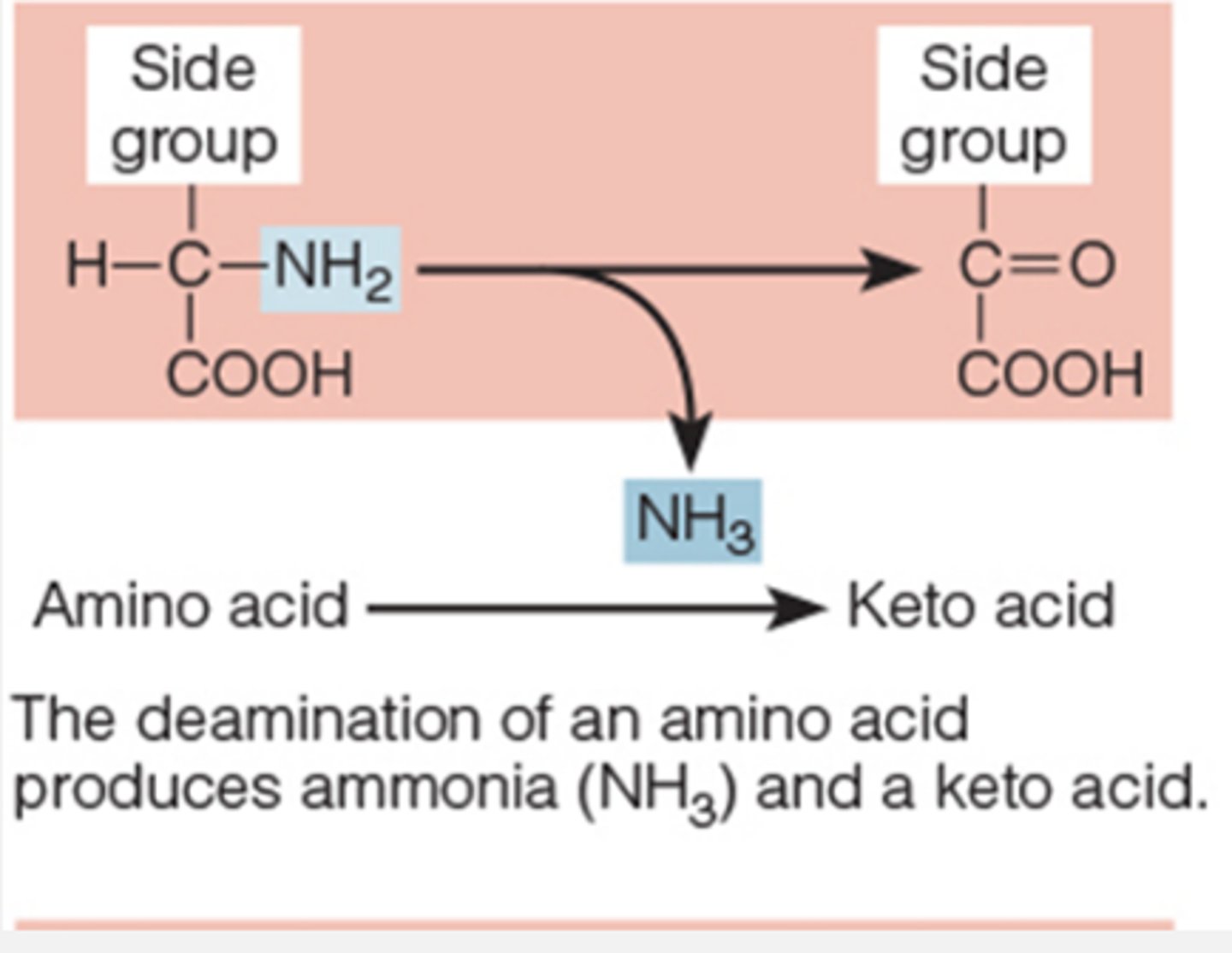
urea is the principle vehicle for excreting
unused nitrogen
urea
A chemical that comes from the breakdown of proteins
urea increases with
protein intake
urea requires water to
dilute and excrete urea
in order to use amino acids for energy
nitrogen must be removed
Without extra water, a person on a high-protein diet risks
dehydration (because the body uses its water to rid itself of urea. This explains some of the water loss that accompanies high-protein diets.)
protein quality determined by
digestibility and amino acid composition
digestibility of protein
▪Animal proteins high (90 - 99%)
▪Legumes close (~90%)
▪Other plants lower (70 - 90%)
amino acid composition of protein
have all essential amino acids?
protein synthesis stops if an essential amino acid is missing
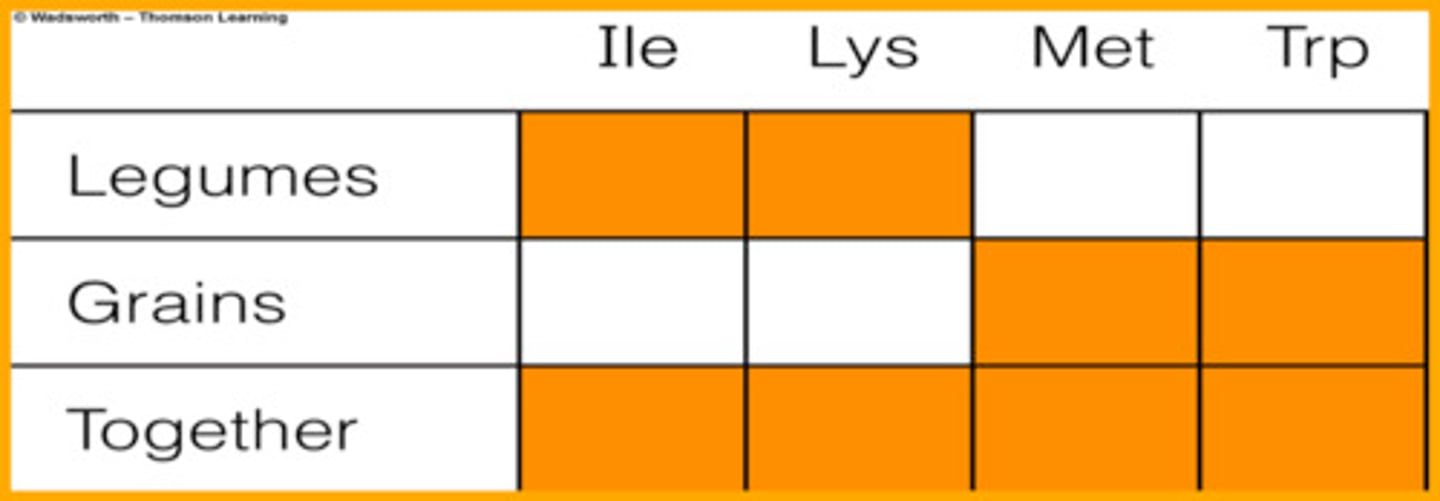
complementary protiens
proteins which can be paired in order to form a complete protein; categories
include grains, legumes, nuts and seeds
high quality (complete) complementary proteins
animal foods, soy (all contain essential amino acids)
The egg is "nature's most nearly perfect food"
gold standard
low quality (incomplete) complementary proteins
other plant foods (low or lacking some amino acids)
complementary proteins
two or more dietary proteins whose amino acid assortments complement each other in such a way that the essential amino acids missing from one are supplied by the other
egg protein
Egg protein has a 'chemical score' (essential amino acid level in a protein food divided by the level found in an 'ideal' protein food) of 100, a 'biological value' (a measure of how efficiently dietary protein is turned into body tissue) of 94, and the highest 'protein efficiency ratio' (PER: ratio of grams of weight gain to grams of protein ingested in young rats) of any dietary protein
we do not store
amino acids
we have an amino acid
pool in constant flux
health effects of protein deficiency
consequences, protein-energy malnutrition, marasmus and kwashiorkor
Marasmic and Kwashiorkor
combination of chronic energy deficit and chronic or acute protein deficiency
high animal-protein intake can cause
cancer
cancer
protein not the contributing factor
some protein foods are carcinogens