Collision Theory VMA
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Successful collision
A collision with sufficient energy and in the correct orientation
Increase rate of reaction
Can be caused due to increase in temp
increase in concentration/pressure (gas)
increase in surface area
increased temperature
particles gain energy = moves quicker = more frequent + faster collisions
increased concentration
more particles of reactant colliding between molecules = more frequent collisions
increased pressure
particles more compressed together = more frequent collisions
Increase in surface area
particles around solution have more area to work on = more frequent collisions
Activation energy
minimum amount of energy needed by particles to react
Catalyst
substance speeding up reaction
w/o being changed/used-up in reaction
The Collision Theory states that 3 conditions must be met for a collision to occur. What are the conditions?
1. Atoms must collide
2. Atoms must be in the correct orientation
3. Atoms must hit each other with enough energy.
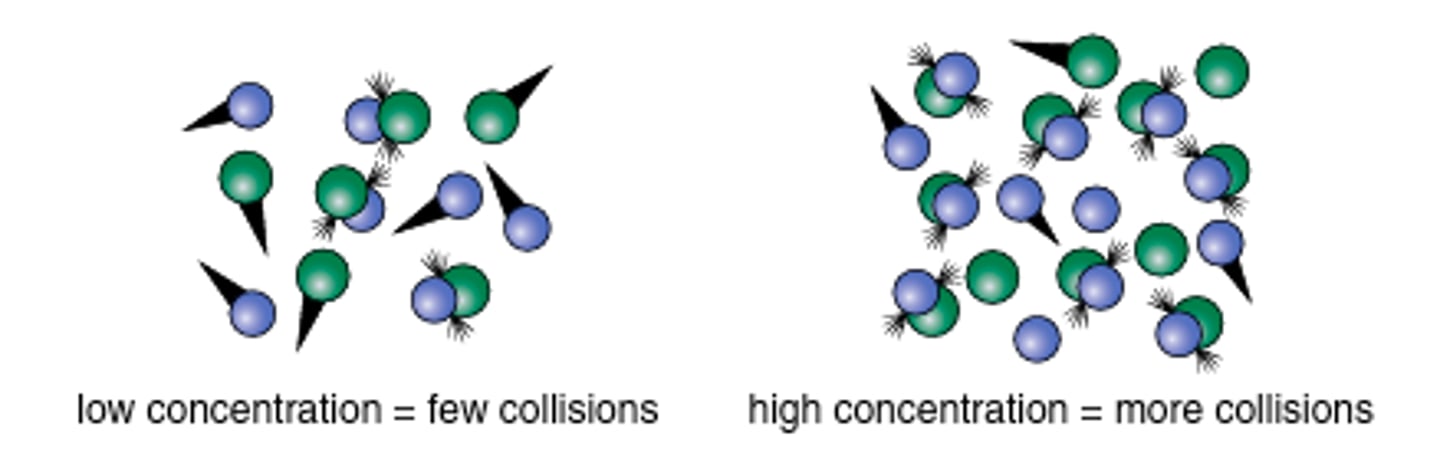
High concentration vs. Low concentration
The higher the concentration (amount) of reactants the more likely it is that there will be a collision
Energy profile diagram
A diagram that shows the energy changes during the course of a reaction.
rate of reaction
the change in concentration/mass/colour/ of a reactant or product per unit time
Types of reactions
synthesis, decomposition, single replacement, double replacement, combustion, neutralisation