Structural Materials
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
What is Carbonation?
Carbonation is where CO2 reacts with the chemicals in the cement mix, leading to a drop in the pH of the concrete, which leads to the corrosion of the steel reinforcement.
High water-cement ratio leads to more capillary pores (voids) which allows CO2 to diffuse through the concrete easier, which causes deeper carbonation.
Longer curing means the concrete has longer to gain strength and complete its hydration, which leads to a decrease in voids and therefore it is harder for CO2 to diffuse through the concrete causing less penetration.
(CO2 + H2O→ carbonic acid) and why the rate peaks at 50–70% relative humidity (too dry = no water for acid; too wet = pores blocked to gas diffusion).
What is Sulphate Attack?
The principle reaction in sulphate attack is the reaction of soluble sulphates with monosulphate to form ettringite. There’s an initial increase in strength and density due to pore filling.
Sulphates (found in soil, groundwater, or waste materials, like old plaster) gets into the mix and reacts with the cement content. This chemical reaction creates new, needle-like crystals called ettringite. The crystals formed take up a lot more space than the original components and causes immense pressure inside the concrete, which causes it to crack, swell and eventually crumble.
Sulphates react with aluminate phases to form expansive ettringite, causing cracking. SRPC resists this by having a very low C3A content.
What is Permeability?
Permeability is how easily water can go through the pore network of the cement paste of concrete. So basically high permeability, which comes with an increase in the water-cement ratio, means more water can travel through the pores (voids) of the concrete making it more susceptible to chemical attack and weakening durability.
High permeability means that there are more voids in the concrete which means that aggressive ions can more easily diffuse through the concrete (such as CO2 and chlorides).
Low permeability is favourable as it means there are less pores for aggressive ions to diffuse through.
What are the Factors that Affect Permeability?
Water-Cement Ration - A high water-cement ratio means more voids are filled with water, which will eventually evaporate leaving capillary pores which decreases the strength of the concrete (Increased permeability).
Hydration and Age - As hydration continues over-time , hydration products ( like C-S-H gel) progressively fill the capillary pores, causing permeability to reduce as the pore structure becomes more refined.
Cement Replacement Materials - The use of CRMs (like PFA or GGBS) reduces permeability. These materials change the morphology of the binder into a foil-like structure, which creates a finely distributed, poorly interconnected network of pores that slows down the diffusion of corrosive elements like chlorides.
High Performance Concrete - HPC is engineered to have very low permeability by using a very low water-cement ratio and fine particles (like silica fume) to eliminate the porous interfacial transition zone and fill remaining voids.
What is Porosity?
Porosity is the volume of voids (empty spaces in a material not filled by solids) within a material.
Pore Refinement - As hydration continues, solid products fill the capillary voids, making the pore network finer and less connected, which significantly reduces permeability and enhances durability.
Effects of CRMs - The addition of CRMs like slag or fly ash changes the morphology of the binding phase to a foil-like structure. This creates a poorly interconnected porosity, which is specifically noted for reducing chloride diffusion rates.
Types of Porosity in Cement;
Capillary Pores - These are the voids remaining from originally water-filled spaces that were not replaced by hydration products.
Gel Pores - These are much smaller voids found between particles of C-S-H. Unlike capillary pores, gel pores have no impact on strength or durability.
Entrapped Air Voids - These are larger voids that remain if the fresh concrete is not properly vibrated or compacted.
What Factors Affect the Pore Structure?
Water-Cement Ratio - Mixing water occupies space that is not initially filled by solids. A higher water-cement ratio creates more originally water-filled voids. Higher water-cement ratio leads to high permeability. Any void that is filled by water cannot be filled by a solid hydration and remains as a capillary pores.
Type of Cement - CRMs change the morphology of the outer product C-S-H from fibrillar structure to a foil-like structure. This foil-like morphology fills space with a finely distributed, poorly-interconnected porosity. This significantly reduces the connectivity of capillary pores, which lowers permeability and chloride diffusion rates.
Curing Time - If concrete is poorly cured (e.g. air cured), hydration may stop prematurely due to a lack of water, leaving a significantly more porous structure compared to moist-cured concrete. As hydration continues, solid products (like C-S-H gel) progressively fill the capillary voids. This reduces the volume of capillary pores while increasing the volume of much smaller gel pores.
Compaction - Adequate vibration or power compaction is required to remove air bubbles. If the mix reaches its compaction limit (often due to a water-cement ratio that is too low for the chosen method), large air voids will remain, drastically increasing the overall porosity.
What are Air Entraining Agents?
Enhances resistance to environmental weathering, especially freeze-thaw cycles.
They allow for microscopic disconnected air bubbles into the hardened cement paste. These bubbles provide expansion chambers for water when it freezes, preventing the internal pressure from cracking the concrete.
This isnt the same as entrapped air due to bad compaction. The difference lies in their size, origin, and im[act on the material’s performance. Entrapped air is typically large pores that are irregular in shape and randomly distributed. Voids slightly reduce strength but greatly improves weathering resistance.
What are Superplasticisers?
Reduce the amount of water needed in a cement mix.
Allow for high workability while using a very low water-cement ratio (e.g. 0.27). Lowering the water content is essential for reducing porosity and increasing strength.
While expensive they can be reduced by including cement replacement materials like PFA and GGBS, which naturally improve workability because they react slow or not at all in the first few hours.
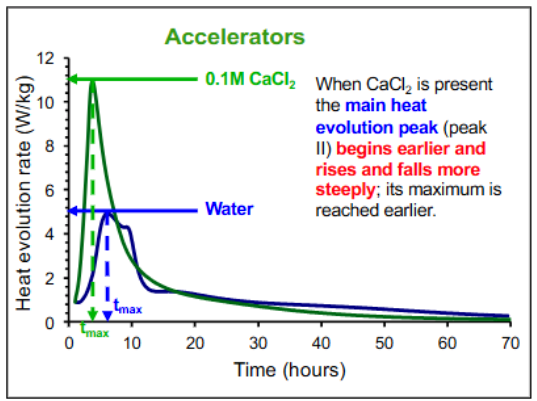
What are Accelerators?
Added to concrete to increase the rate of hydration, thereby speeding up setting and strength development.
Used in cold weather, to speed up the hydration reaction.
Calcium Chloride (CaCl2) is the most effective accelerator. However, because chloride ions promote the corrosion of steel reinforcement, Calcium Chloride is no longer used in reinforced concrete.
Accelerators promote the growth of the primary binding phase in concrete, Calcium Silicate Hydrate (C-S-H).
What are Retarders?
Used to delay the chemical process of hydration, slowing down the setting or hardening of the mix.
These are essential for hot weather, for delayed placing.
Many organic materials act as retarders, particularly sugars.
Hydration occurs when cement grains come into contact with water, triggering chemical reactions that aid strength develepment. Retarders coat these cement grains to prevent water from reacting with them.
How is Standard Portland Cement made?
A mixture of limestone and clay is ground into raw feed.
The raw feed enters a preheater where it is heated and the clay undergoes dehydration (at 450°C).
Calcination occurs, this is where the limestone is heated and separated into lime and carbon dioxide.
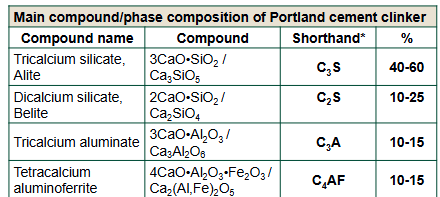
The materials enter a long, rotating kiln where the lime reacts with the silica and alumina to form the four main clinker phases Alite (C3S), Belite (C2S), Aluminate (C3A), and Ferrite (C4AF).
The resulting clinker is rapidly cooled to 20°C and then grounded with gypsum to produce the final cement.
The cement is then packaged and distributed.
What are the Four Main Portland Cement Clinker Phases?
Alite (Tricalcium Silicate, C3S)
50 - 70% of Portland Cement Clinker.
Reacts rapidly during acceleration period to provide early strength.
High, rapid release heat during first few days.
Belite (Dicalcium Silicate, C2S)
15 - 30% of Portland Cement Clinker.
Reacts very slowly contributing to long-term strength after one week.
Heat evolution is much slower than Alite.
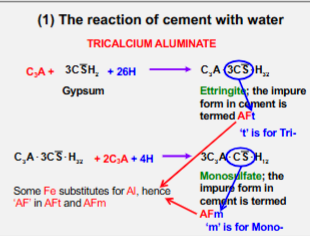
Aluminate (C3A)
5 - 10% of Portland Cement Clinker.
Reacts instantly with water.
Generates the highest and most intense heat out of all the phases.
Ferrite (C4AF)
5 - 15% Portland Cement Clinker.
Controls clinker formation temperature’s and gives concrete it’s concrete it’s grey colour.
Evolves a moderate amount of heat at a highly controlled slow rate.
NOT A CLINKER BUT STILL NEED TO KNOW!
Ettringe - C3A * 3CSˉ * H_32
Gypsum - CSˉH_2
Monosulphate - 3C_3A⋅CSˉ⋅H_12
What is Calcium Aluminate Cement (CAC)?
Produced by fusing limestone and bauxite at 1450-1600 °C. While more energy intensive, it provides specialized properties like rapid strength gain and chemical resistance. Primary cementing phase is monocalcium aluminate (CA), which typically makes up 60-70% of the material by mass.
CAC sets in a similar time but develops strength more rapidly than CEM I. The day 1 strength is actually higher than the 28 day strength for the CEM I.
It has excellent resistance to chemical attack especially against sulphates.
Suitable for use at low temperatures.
What are the Main Causes of CO2 Emissions?
There will more than likely be a question in the exam asking you to calculate CO2 emissions (Look at past exam)!!
Calcination - During cement production limestone is heated to roughly 900 °C to produce lime (CaO), releasing CO2 as an unavoidable by product (CaC03 + heat → CaO + CO2).
Fuel Combustion - Large amounts of fossil fuels are burnt to reach the high kiln temperatures (up to 1400-1450°C) required for clinker formation.
About 4 billion tonnes of cement is produced each year. Each tonne of cement releases about 850 kg of CO2. These emissions are intrinsic to the manufacturing process. Cement is manufactured by blending limestone (85%) and clay (15%) and then heating it to ~1350oC before cooling it rapidly and grinding it. This gives 3 sources of CO2 emissions: decarbonation of limestone (~500kg), energy required to heat the mix of limestone and clay to ~1350oC (~300 kg) and grinding and processing materials on site (~50kg).
What are Functional Units?
Engineers use functional units like eCO2 per unit of structural performance (e.g., the CO2 cost to support a specific bending moment). The functional unit accounts for emissions during mining, transport and processing but not operation.
Comparing materials by "kg CO2 per kg of material" is invalid. They must be compared based on the structural job they do (e.g., eCO2 per MN of load capacity).
Timber is often the lowest carbon for small, domestic structures, while concrete is often the most efficient for very large-scale infrastructure
Calculating eCO2: Calculate the footprint of a blend (e.g., 0.8×clinker eCO2+0.2×PFA eCO2).
What are the Strategies for Reducing Carbon Emissions?
Cement Replacement Materials (CRMs) - Replacing clinker with industrial waste like PFA or GGBS can reduce the environmental impact by up to 22%.
Alternative Binders- New binder systems aim to eliminate or reduce the calcination of limestone.
Geopolymers - Calcium-free systems that produce no calcium, and therefore no CO2.
Magnesia Cements - These utilize magnesium carbonate, which decomposes at lower temperatures and can recombine (absorb) CO2 during hardening.
Celitement - A hydrothermal process that reduces production energy by 50%.
Lifespan and Reuse - Extending the life of existing buildings or reusing them is the most effective long-term strategy for emission reduction.
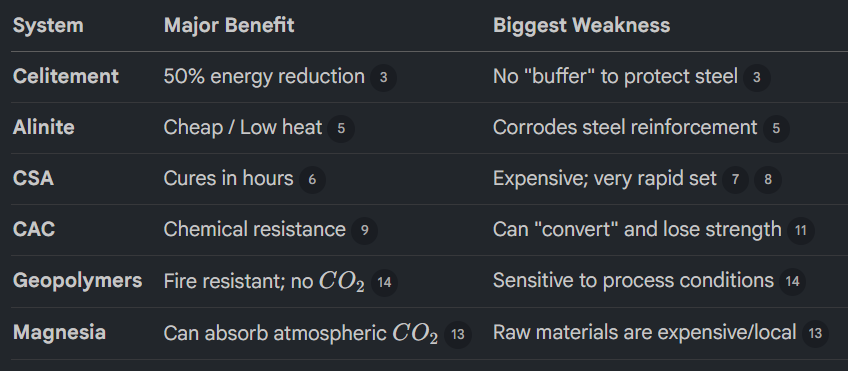
What are Alternate Cement Systems?
Celitement - Uses a two-stage hydrothermal process to create a binder that requires 50% less energy to produce standard cement. It is strong but doesn’t contain calcium hydroxide (CH), which protects the steel reinforcement from rusting.
Alinite Cement - Uses chlorides as a flux to lower the kiln temperature to 1000°C. While it is cheaper and uses waste materials, the chlorides within the cement will corrode steel reinforcement.
Calcium Sulphoaluminate - These cements reach full strength in hours rather than days. They are expensive and can be hard to work with because they set so quickly.
Calcium Aluminate Cement (CAC) - Known for extreme chemical resistance and fast 1-day strength. However, if kept in warm, moist conditions, it can undergo conversion, where the internal structure shifts and loses half its solid volume, potentially leading to a collapse if the water-cement ratio is too high.
C02 Recombination - Because magnesium carbonate decomposes at a much lower temperature (~450°C) than limestone (~700°C), it uses less energy to make.
Absorption - Most importantly, these cements can absorb atmospheric CO2 as they harden, potentially making them carbon neutral or even carbon negative.
Geopolymers - They are marketed as no calcium, no CO2 binders. Instead of heating limeston, they take industrial waste (like fly ash or slag) and activate it with a strong alkaline chemical. They have excellent fire resistance and rapid strength gain, however they are very sensitive to how they are made and require handling acid-like chemicals.
What is the Difference Between Hardened Concrete and High Strength Concrete?
Hardened Concrete - This is a general term describing the state of the material once it has transitioned from its fluid fresh state to a solid that has gained structural strength. All concrete used in structures is hardened concrete, regardless of its specific strength class.
Microstructural;
Normal Concrete (<60 MPa) - The strength is dominated by its weakest link, the Interfacial Transition Zone (ITZ) a 20-50 micro meter thick layer of cement paste surrounding the aggregate particles. This zone is characterized by higher porosity and a higher concentration of calcium hydroxide (CH). The ITZ is the weakest link due to its higher porosity from a higher water-cement ratio.
High-Strength Concrete (HSC) - HSC is designed to have no ITZ. By using very low water-cement ratios and extremely fine particles like silica fume, the voids in this zone is filled so effectively that the paste becomes as strong as the aggregate.
Fracture and Failure Mechanism;
Facture Path - In normal concrete because the ITZ is weak, cracks typically travel around the aggregate. In HSC, the paste and aggregates strength are comparable, cracks travel through the aggregate itself.
How do you Achieve High Strength Concrete?
Low water-cement ratio - less water so that the cement particles are closer together.
Superplasticisers - Maintained workability at low water contents.
Silica Fume - Due to its extreme fineness, SF is almost essential for strengths above 100 MPa, it fills the tiny gaps between cement grains and strengthens the ITZ.
Aggregates - Crushed stone with a rough surface texture is preferred over smooth gravel to ensure a superior mechanical bond. Additionally, the maximum aggregate size is typically reduced as the target strength increases.
Curing - Proper curing is more critical for HSC. Because the water-cement ratio is low, hydration will stop completely if all internal water is consumed, even if no moisture escapes the structure.
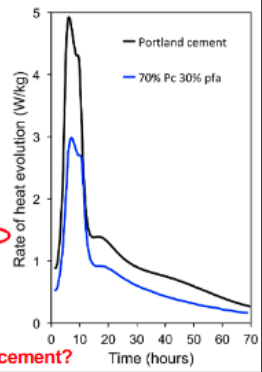
What is Calorimetry?
Calorimetry is used in the study of cementitious materials to measure the heat of hydration. Because cement hydration reactions are exothermic (they release heat), measuring the rate and total amount of heat evolved provides direct insight into the progress of the chemical reactions.
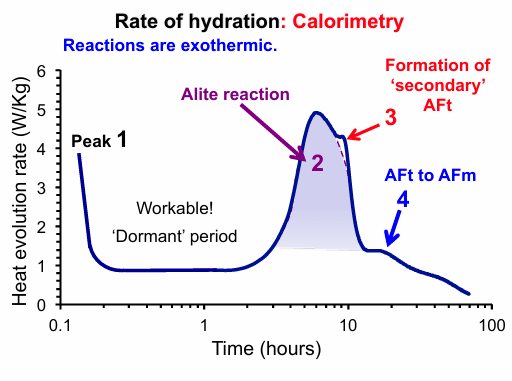
What do the Peaks in the Rate of Hydration Graph Represent?
Peak 1 - Occurs immediately upon contact of cement with water as the most reactive phases begin to hydrate.
The Dormant Period - A period of very low chemical activity following the initial peak. This stage is crucial for engineers as it is when the concrete remains workable and can be placed.
Peak 2 - This is the main evolution peak, primarily associated with the reaction of Alite (C3S). The maximum rate of heat evolution during this peak is typically coincides with the setting of the paste.
Peak 3 and 4 - These smaller peaks are associated with the transition of aluminate phases, such as the formation of secondary AFt (ettringite) or the conversion of AFt to AFm (monosulphate).
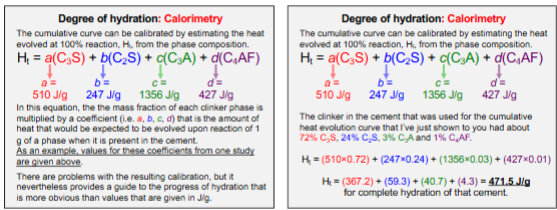
Degree of Hydration: Calorimetry
What is Ettringite?
Ettringite (AFt) is a crystalline hydration product that forms early in the chemical reaction between cement and water.
What are the Impacts of Admixtures and CRMs on Calorimetry?
Accelerators - Chemical admixtures like calcium chloride (CaCl2) cause the main heat evolution peak to start earlier and rise and fall more steeply, reaching its maximum sooner.
Retarders - Organic retarders (like sugars) poison the growth of hydration products, delaying the peaks and lowering the maximum rate of heat evolution.
Cement Replacement Materials - The use of materials like PFA and GGBS typically reduce the total heat evolution and the rate of heat flow, which is beneficial for preventing thermal cracking in large structures.
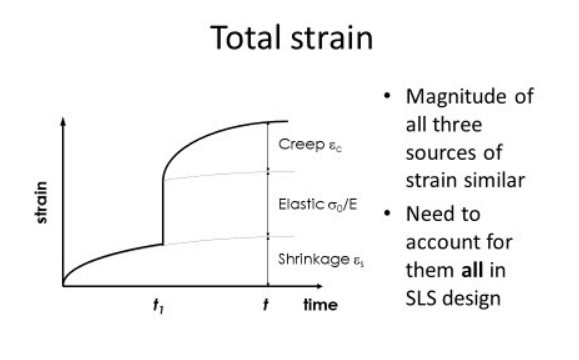
What are the 3 Sources of Strain in Concrete?
Elastic Strain - The immediate deformation that occurs as soon as a load is applied to the concrete structure.
In normal concrete, as stress increases into the non-linear region, microcracks begin to grow within the Interfacial Transition Zone (ITZ) and break out, leading to strain that cannot be recovered when the load is removed.
Drying Shrinkage Strain - The long-term deformation of the material driven by the loss of water from the internal microstructure as the concrete dries.
Aggregates act to restrain shrinkage; therefore, concrete with higher aggregate content generally exhibits less shrinkage than pure cement paste. If this shrinkage is restrained by the rest of the structure, it often leads to cracking.
Shrinkage is increased by a higher water-cement ratio and smaller structural sections (which dry faster).
Creep Strain - The time dependent increase in strain that occurs while the concrete us under a constant, sustained load.
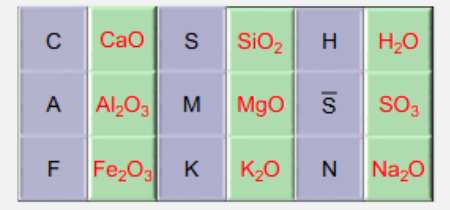
What are the Chemist Short-Hands?
See diagram.
Characteristic Strength
f_ck = f_cm - 1.64*standard deviation. → Characteristic Strength is the strength of which 95% of the samples are expected to exceed.
f_cm = mean strength (function of the design mix)
f_ck = characteristic strength (function of quality control)
Good Quality Control: Results in a smaller standard deviation (4 MPa).
Average Quality Control: Results in larger standard deviation (8 MPa). This is the minimum standard deviation that you plug into the formula and check the mixes against.
C50/60
The first number is the cylinder strength.
The second is the cube strength.
Cylinder strength is lower. It is more accurate.
Longer samples reduce test machine interference.
Testing plates cause crushing stress.
Short cubes trap this stress.
Longer cylinders escape it.
Compaction of Concrete
See Diagram
Removes entrapped air voids from fresh concrete, for increased density. If concrete not fully compact, air voids form within the mix, reducing strength.
Generally, reducing the water-cement ratio increases concrete strength, however if you reduce it too much it becomes unworkable and dramatically reduces in strength (there is not enough cement paste to fully surround the aggregate).
How does the C-S-H binding phase change when Portland cement is blended with Cement Replacement Materials (CRMs).
Chemical Change: The Ca/Si ratio reduces and aluminium is incorporated into the chains, forming C-A-S-H.
Nano-structural Change: Silicate chain length increases (MCL increases).
Morphological Change: The structure shifts from fibrillar (needle-like) to foil-like.
Durability: This foil-like structure refines the pores, reduces interconnectivity, and lowers permeability, making it much harder for chlorides or sulphates to penetrate
The BRE Mix Design Method
Target Mean Strength: First, calculate the target strength by adding 1.64σ to the required characteristic strength.
w/c Ratio: Use BRE Table 2 (reference strength at 0.5 w/c) and Figure 4 (strength vs. w/c curves) to find the design w/c ratio.
Water & Cement: Use BRE Table 3 to find water content based on required slump (workability) and aggregate size, then divide by the w/c ratio to find the cement content.
Aggregates: Subtract water and cement weights from the total wet density, then split the remainder between fine and coarse aggregate using BRE Figure 6.
Inner vs Outer Product C-S-H
The principle binding (glue) phase in concrete Calcium Silicate Hydrate (C-S-H), is categorized into inner and outer product based on where it forms around the cement grains. The location of the product determines the concrete’s strength, permeability, and durability.
Inner Product C-S-H
Forms inside the original boundaries of the anhydrous cement grains. It grows to fill the space between the grain and the hydrated shell, eventually slowing the reaction.
Fine and homogenous (consistent in texture) in appearance.
Outer Product C-S-H
Grows outwards from the grain boundaries into the spaces originally occupied by mixing water. It typically grows early during the dormant and setting periods.
When blended with CRMs, like GGBS or PFA the outer-product morphology changes from fibrillar to foil-like.
Both (Inner and Outer Product)
In neat Portland cement, both inner and outer product maintain a similar average calcium-to-silica ratio of approximately 1.7 to 1.8.
Microscopy Resolution and Techniques? (Probably won’t come up)
Resolution Scales:
Human Eye: 100 μm – 1 mm.
Light Microscopy: 1 μm – 100 μm.
Scanning Electron Microscopy (SEM): 10 nm – 100 μm; ideal for AFt needles (~250 nm).
Transmission Electron Microscopy (TEM/HRTEM): 1 nm down to <1 Å; required to see silicate chains.
SEM Signals and Imaging:
Secondary Electrons (SE): Provide topographical info; typically used on fracture surfaces.
Backscattered Electrons (BSE): Provide atomic number info; used on flat polished surfaces.
BSE Visuals: White (Steel/Unhydrated Cement), Light Grey (CH), Medium Grey (C-S-H/Aggregate), Black (Pores).
Morphology Identification:
C-S-H: Inner Product is fine/homogeneous; Outer Product is fibrillar (pure cement) or foil-like (with CRMs).
AFt (Ettringite): Short rods (early) or long slender needles (later).
AFm (Mono-sulfate): Hexagonal plates.
CH (Portlandite): Large hexagonal prisms.
“Global cement production is responsible for 6-8% of global CO2 emissions”. Discuss this statement with regards to the cement manufacturing process and global cement production?
About 4 billion tonnes of cement is produced each year.
Each tonne of cement releases 850Kg of CO2.
There are 3 main sources of CO2;
Decarbonation of Limestone into Lime and CO2.
The energy of fuel required to heat the limestone and clay mix to 1350 degrees.
Grinding and processing of the materials to form the cement powder.
GGBS
Environmental Benefits
It is a waster product and would go into a landfill otherwise.
Partially replaces cement content, less cement = less CO2 emissions.
Heat Evolution
Peak heat evolution will be lower when using GGBS. This makes the material particularly good for large concrete pours.
Strength Development
CEM IIIA develops strength more slowly than CEM I, so early-age strengths are reduced. By 28 days the performance is similar, but the pozzolanic reaction continue for longer, such that by 56 days and above compressive strength for CEM III/A concrete exceeds that of an equivalent CEM I mix.
Resistance to Chloride ingress
The partial replacement of cement with CRMs change the morphology from fibrillar to foil like. This reduces the interconnectivity of the pores/voids, which helps to prevent the diffusion of chlorides through the material.
Prolonged hydration leads to a reduction in capillary porosity and so reduced permeability. This restricts the ingress of chlorides.
PFA
PFA is a pozzolan so it will react with portlandite to produce additional C-S-H. This means the permeability will be reduced, but there will be negligible portlandite present in the hardened cement paste at later ages.
Environmental Benefits
Heat Evolution
Strength Development
Resistance to Chloride ingress
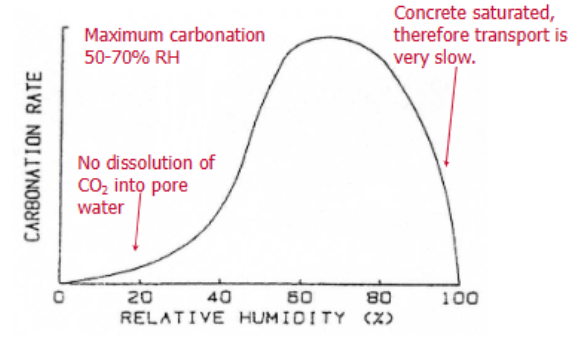
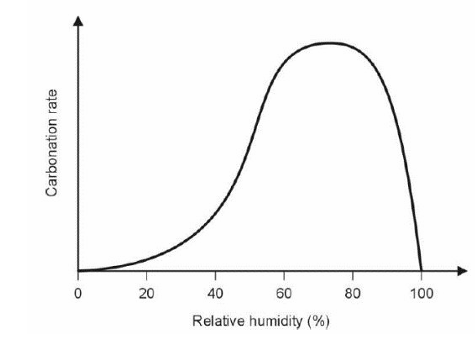
Show, with the aid of a diagram, how the carbonation rate of concrete varies with relative humidity. Explain the relationship?
Carbonation only occurs in the presence of water.
At low Relative Humidity, the rate of carbonation is low because there is little water for the formation of the carbonic acid.
The rate rises with increasing humidity because more carbonic acid can be formed.
At very high relative humidity (above 80%), the rate drops dramatically because the concrete pores become saturated with water, which blocks the diffusion of CO2 gas into the material.
What does C-S-H do?
Extra C-S-H is produced when CRMs react with portlandite (CH) during the pozzolanic reaction.
Pore Filling - Additional C-S-H fills the internal voids and capillary pores that were originally occupied by mixing water.
Reduced Permeability - By filling the pores, the connectivity is disrupted, which significantly reduces the permeability of the hardened cement paste.
When CRMs are used, the morphology of the outer product C-S-H is shifted from fibrillar to foil-like, which is effective at blocking interconnected pores.
Sulphate Resisting Portland Cement
Why is SRPC not being used as much?
SRPC has a low C3A (aluminate) content and chlorides are bound by C3A. This reduces the formation of ettringite, i.e. the expansive phase.
The low C3A contents, means the clinker must be prepared at much higher temperatures than standard cement to achieve the necessary chemical reactions. Higher kiln temperatures means more fossil fuels are burned to reach the necessary temperature.
What is the standard emission range per Kg for cement?
0.7 to 0.9 kg CO2 per kg of cement.
cement mass / concrete mass

Why is curing necessary?
The primary reason for curing is to facilitate prolonged hydration. Continued hydration creates more C-S-H gel, which fills the internal capillary pores. This allows for stronger concrete.
Proper curing reduces the connectivity of the pore network, significantly lowering the materials permeability and therefore increasing durability and strength.
Curing is even more critical when using CRMs;
CRMs hydrate slower than Portland cement.
The CRM must be allowed enough time to react with Portlandite to produce additional C-S-H. If curing is cut short, the CRM may never fully react, leaving the concrete with a higher porosity and lower durability.
What are the different exposure classes?
Corrosion Induced by Carbonation (XC)
XC1: Dry or permanently wet.
XC2: Wet, rarely dry. Long-term water contact.
XC3: Moderate humidity.
XC4: Cyclic wet and dry. Inside buildings with moderate air humidity.
Corrosion Induced by Chlorides (XD and XS)
XD (Chlorides other than sea water);
XD1: Moderate Humidity. Airborne chlorides.
XD2: Wet, rarely dry. Swimming pools.
XD3: Cyclic wet and dry. Parts of bridges exposed to spray containing chlorides.
XS (Chlorides from sea water);
XS1: Surfaces exposed to airborne salt but not in direct contact with sea water.
XS3: Tidal, splash and spray zones.
Trends and Requirements:
Aggressiveness: XS classes (sea waters) are generally considered more aggressive than XD classes because of the additional Prescence of magnesium ions and sulphates in sea water.
Wetting and Drying: Classes ending in "3" or "4" (cyclic wet/dry) are more aggressive than those ending in "1" or "2" (permanently wet or dry) because the cycling causes harmful salts or acids to concentrate in the pores.
Stringency: As the risk increases (e.g., from XC1 to XC4 or XD1 to XD3), the requirements become more demanding, necessitating higher characteristic strengths (fck), lower w/c ratios, and greater concrete cover.

What are the factors affecting mean strength of concrete?
Water-Cement Ratio
Age / Curing of the Concrete
Choice of Aggregate
What are the three design variables for concrete mix design?
The three core variable for producing a design mix;
Strength - This basically sets the water-cement ratio. Designers must calculate a target mean strength (fcm) that is higher than the characteristic strength (fck) to account for statistical variability.
Durability - This is determined by the environmental exposure class. It places a maximum limit on the water-cement ratio and dictates a minimum cement content.
Workability - This describes how easily the fresh concrete can be transported, placed, and compacted. It sets the water content required for the mix.
Why, if the process is easier to control, are wet kilns being phased out of modern cement plants?
In a wet kiln, the materials are fed into the system as a slurry. A significant amount of energy is wasted simply evaporating this water before the actual chemical reactions can begin.
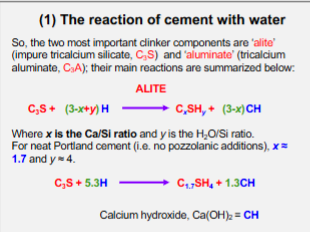
Alite Chemical Reaction
Tricalcium Aluminate
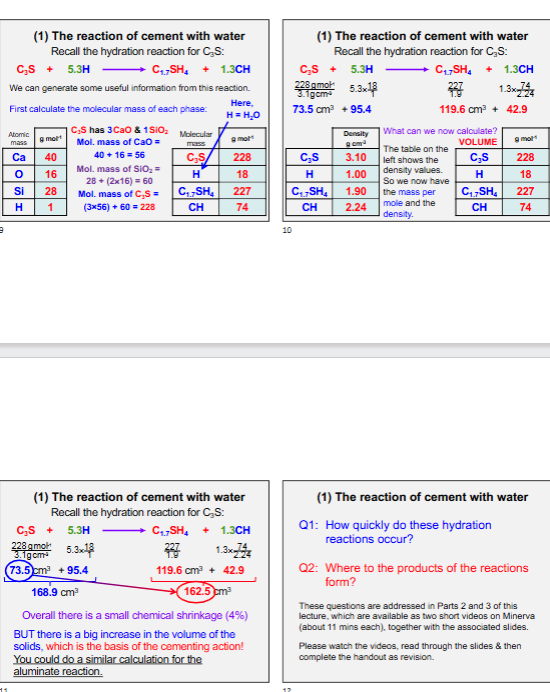
Reaction of Cement with Water
Alite
Responsible for initial and early strength development of concrete.
Alite reacts quickly with water to produce C-S-H, which is the main binding phase that holds the concrete matrix together.
Most abundant compound in Portland Cement Clinker (40-70% of its mass).
The hydration of Alite is highly exothermic. It is responsible for the second and most significant peak in calorimetry.
Alongside the strength-giving C-S-H gel, the hydration of alite also produces calcium hydroxide (CH). This phase provides the alkalinity necessary to protect the steel reinforcement.

Pozzolanic Reactions
A pozzolanic reaction is a chemical reaction that occurs when a CRM react with the products of Portland Cement hydration to form additional binding phases.
Pretty much all CRMs are considered pozzolanic, however GGBS consumes portlandite (CH) so it is technically considered a latent hydraulic material.
CRMs have glassy or poorly crystalline structures, the highly alkaline nature of Portland cement attacks these structures, breaking the bonds between the silicon/aluminium and oxygen.
In blended cements, aluminium from the CRM substitutes for silicon in the silicate chains (forming C-A-S-H).
The morphology shifts from fibrillar to foil-like.
Portland Cement Notations.
Rapid Hardening Portland Cement (RHPC)
Low Heat Portland Cement (LHPC) - CRMs like GGBS and PFA are frequently used to achieve these low-heat characteristics. These have significantly longer curing times.
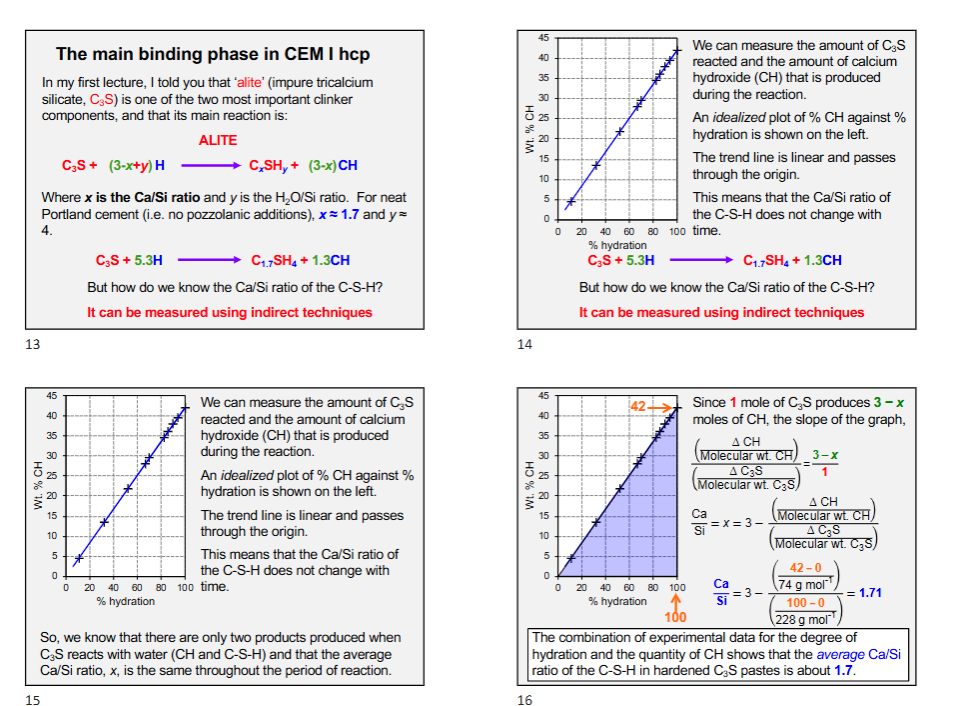
Average Calcium-Silicate Ratio (Ca/Si)
Ca/Si ratio can be measured directly by X-Ray analysis in an electron microscope.
Nanostructure of C-S-H?
Hydration begins with the formation of monomers, which quickly form dimers (chains of two tetrahedra). Continued hydration leads to pentamers (five tetrahedra) and octamers (eight tetrahedra) by inserting a single tetrahedra into the bridging positions.
The 2 crystalline phases that are used as models for C-S-H present in hardened cement (tobermorite and jennite).
The higher the Ca/Si ratio, the more additional structure (chain length) is required.
The distribution of chain lengths follows the sequence 3n−1 (where n is an integer).
The Ca/Si ratio typically decreases as the percentage of CRM increases.
Linear Silicate Chains - These chains are attached to the central Ca-O layer in a kinked pattern.
Tetrahedra Types - Within these chains, some silicate tetrahedra are linked directly to the central layer (paired tetrahedra) while others are not directly linked (bridging tetrahedra).
Use schematic diagrams to explain and illustrate how the nanostructure of the main binding phase changes with time.
The MCL data in the table show that the average lengths of the silicate chains in the C-S H increase with age. Dimeric groups form initially and the average length increases from 2 to 4 about 3 after a month or so (at about 85% reaction) and then increases very slowly to about 5 over a period of 30 years. [2 marks] The dimers are linked by monomers to form pentamers, octamers, etc. i.e. a 3n-1 sequence, where n is integer for individual structural units. A very rough sketch of the following should be made:
![<p>The MCL data in the table show that the average lengths of the silicate chains in the C-S H increase with age. Dimeric groups form initially and the average length increases from 2 to 4 about 3 after a month or so (at about 85% reaction) and then increases very slowly to about 5 over a period of 30 years. [2 marks] The dimers are linked by monomers to form pentamers, octamers, etc. i.e. a 3n-1 sequence, where n is integer for individual structural units. A very rough sketch of the following should be made:</p>](https://assets.knowt.com/user-attachments/36002082-65fc-4ead-a769-4b694ee9516f.png)
How do Accelerators or Retarder affect the growth of C-S-H?
Retarders
They poison the formation of the Ca-O polyhedra. By interfering with these polyhedral, the retarder inhibits the growth of the C-S-H binder phase.
Accelerators
Accelerators work by promoting the growth of C-S-H rather than changing the fundamental hydration features. The accelerator influences the rate at which calcium complexes are transferred from the pore solution to the growing particles of C-S-H.