Biology Midterm 2-12 Paletzki
1/75
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
Many of water’s unique properties are due to its
polarity
Polar molecule
A molecule with an uneven distribution of charge, electrons are shared between the Oxygen and Hydrogen atoms.
How does the polarity of water contribute to its unique properties
Hydrogen bonds enable water to be attracted to itself and other polar molecules
Cohesion
water molecules attracted to themselves
adhesion
water molecules are attracted to other polar substances
capillary action
water can creep up thin tubes
Water resists..
temperature change
water expands…
when it freezes
solution
when one substance is dissolved in another
solute
substance being dissolved in the liquid
solvent
substance doing the dissolving
acid
a substance that forms higher concentrations of H+ ions in a solution than water does (therefor lower concentrations of OH-)
base
a substance that forms lower concentrations of H+ ions in a solution than water does (therefore higher concentrations of OH-)
H+
hydrogen ion
OH-
hydroxide ion
pH scale
the concentration of H+ ions measured
properties of acids
taste sour, neutralize bases, corrode metal,
properties of bases
slippery, neutralize acids, also known as alkaline
macromolecules
very large molecules composed of carbon and other atoms
living organisms depend on __ to survive
macromolecules
there are many types of organic compounds because
carbon can bond easily with itself and other atoms
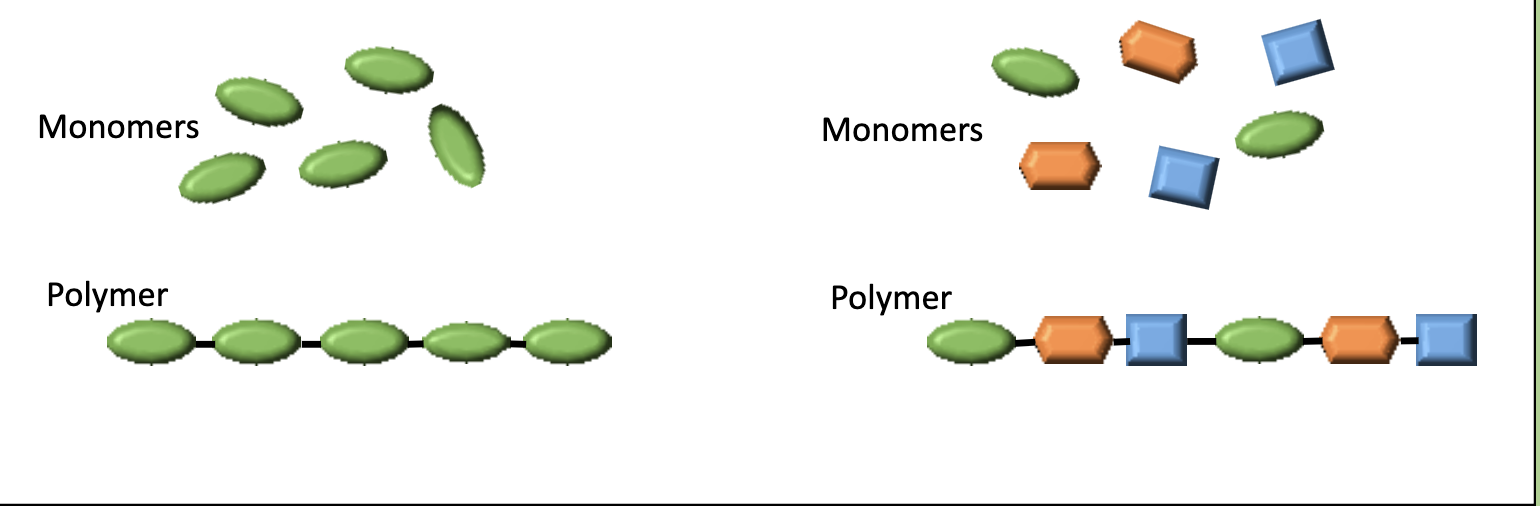
macromolecules are polymers…
made of smaller units called monomers (mono=one, poly=many)
polymers can…
be made of only one type of monomer or several different types
4 major groups of macromolecules
carbohydrates, lipids, proteins, and nucleic acids
carbohydrates
commonly known as sugars, monomers: simple sugars like glucose, fructose, and galactose, polymers: starch, glycogen, and cellulose
carbohydrates function
provide structural support and quick energy, plants use a complex sugar (cellulose) to build leaves and stems, sugar can be stored in complex carbohydrates to be broken down later for energy
lipids
fats, steroids, oils & waxes, insoluble in water because their molecules are hydrophobic
lipids are comprised of
1 glycerol molecule and 1,2, or 3 molecules of fatty acids
function of lipids
provide long term energy storage, form membranes within cells, used as messengers between cells
nucleic acids
made of nucleotide monomers, each nucleotide contains a sugar, a phosphate group, and a nitrogen base
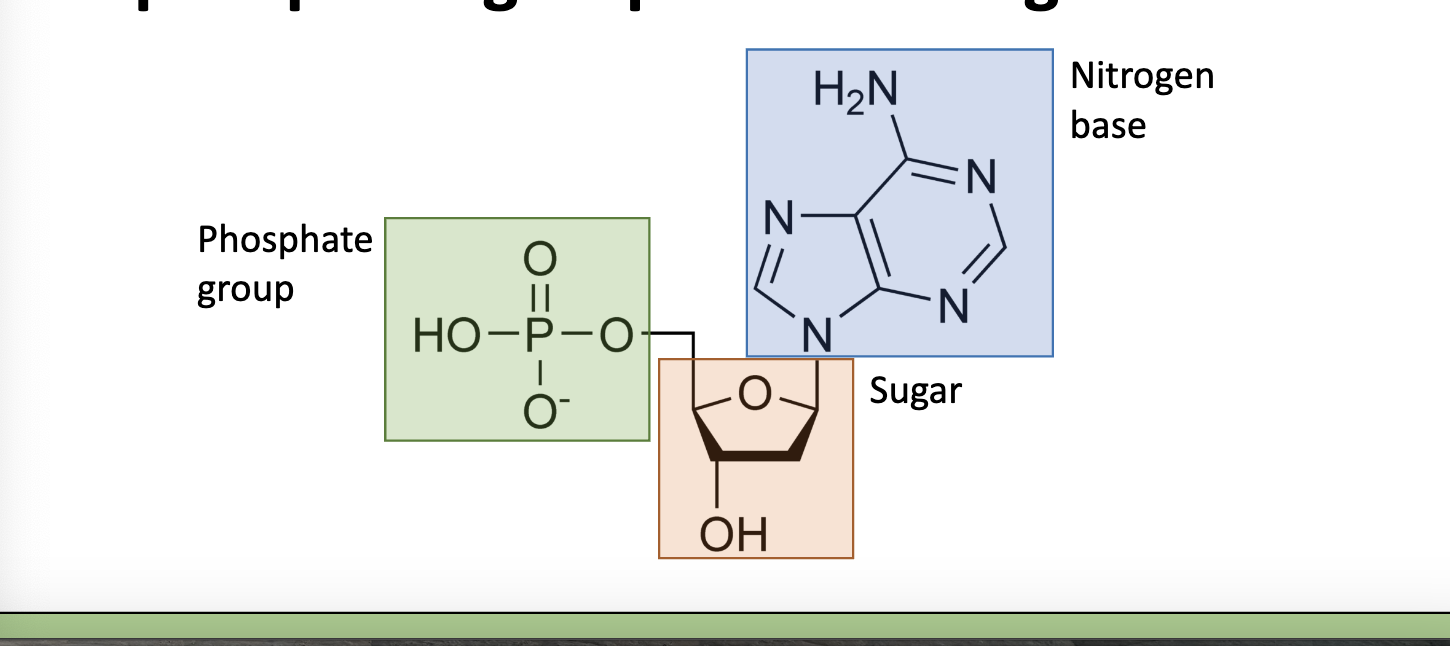
function of nucleic acids
stores genetic information in cells in the form of a code
proteins
contain not only carbon, hydrogen, and oxygen, but also nitrogen atoms, proteins are composed of long chains of amino acids
function of proteins
regulate cell processes, form bones and muscles, and transport materials
the 4 macromolecules are…
polymers made of simple units called monomers and each contains a different proportion of elements
what happens during a chemical reaction
the molecules in living organisms aren’t stagnant. they are constantly being formed and changed through chemical reactions. chemical reactions always involve breaking or forming bonds between atoms.
two types of chemical reactions
energy-releasing reactions & energy-absorbing reactions
energy releasing reactions
can occur spontaneously
energy absorbing reactions
require activation energy to get them started
enzymes
proteins that act as catalysts, speeding up the rate of a chemical reaction by reducing the amount of energy needed to start it - a special type of protein that changes one molecule (substrate) into another (product) with less activation energy than if the reaction had occurred on its own
substrate
the substance on which an enzyme acts
2 major types of reactions caused by enzymes
synthesis & decomposition
synthesis
when molecules are joined together to form a larger product
decomposition
when molecules are broken apart to form smaller products
examples of reactions using enzymes
lactase - breaks down lactose (milk sugars) during digestion, helicase - unwinds DNA strands for copying during cell division
tonicity
the ability of a solution to cause a cell to gain or lose water
isotonic solution
the amount of solute is the same inside the cel as it is outside - water flows equally in and out of the cell
hypertonic solution
a higher amount of solute than the cell does - water will flow out of the cell towards the environment
hypotonic solution (think hypo- hippo)
the amount of solute particles is lower than inside the cell - water will flow into the cell
active transport
movement from low to high - DOES require energy, like rolling a ball up a hill
protein pumps
allow substances to move from low concentration to high concentration (opposite of the normal concentration gradient)
passive transport
movement from high to low - does NOT require energy, like rolling a ball down a hill
bulk transport
moving large molecules in and out of the cell
endocytosis
using vesicles to move very large substances into the cell
exocytosis
using vesicles to move very large substances out of the cell
autotrophs
make their own food from photosynthesis
heterotrophs
need to consume other organisms for food
where does photosynthesis occur
chloroplasts
inside chloroplasts
are stacks of sac-like thylakoids, space surrounding stacks is called stroma
when light energy is absorbed..
by chlorophyll, it adds energy to electrons. these high-energy electrons are transported between the light-dependent reactions and the light-independent reactions on a transport molecule: NADP+
Autotrophs require 4 resources for photosynthesis
sunlight, carbon dioxide (CO2), water (H2O), chlorophyll
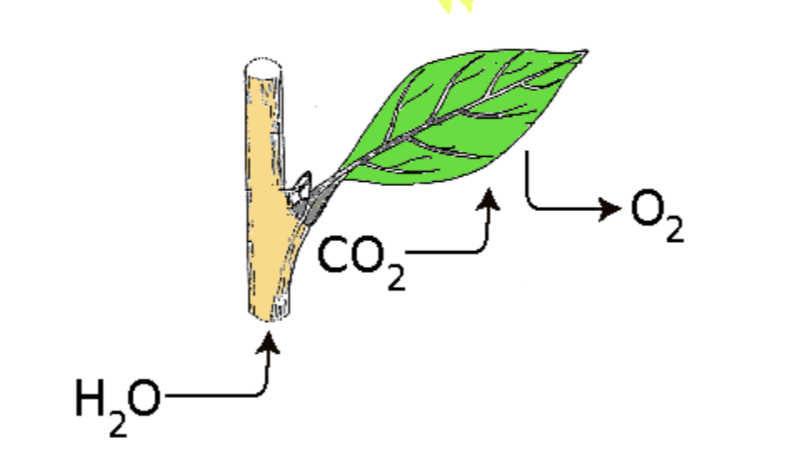
The full equation for photosynthesis
6CO2 (carbon dioxide) + 6H2O (water) + light —chlorophyll→ C6H12O6 (sugar/glucose) + 6O2 (oxygen)
cellular respiration
the process by which all organisms break down glucose to gradually produce energy in the form of ATP
The inputs of photosynthesis are..
the outputs of respiration and vice versa - scientists say that photosynthesis is the “opposite” of cellular respiration
major steps of cellular respiration
glycolysis, the krebs cycle, the electron transport chain
where does cellular respiration occur?
glycolysis occurs in the cytoplasm and the krebs cycle and ETC occur in the mitochondria
how much ATP does cellular respiration yield if oxygen is available to the cell
about 36 (aerobic respiration)
how much ATP does cellular respiration yield if oxygen is NOT available to the cell
2 ATP (anaerobic respiration)
do i want to kill myself thanks to mr paletzki
yes
cellular respiration occurs in…
ALL organisms to produce energy from glucose
what are stem cells
unspecialized cell that can divide indefinitely and become many different types of cells
differentiation
the process of forming specialized cells
multicellular organisms begin..
as one cell that divides into a small clump of cells called an embryo
totipotent
cells that can differentiate into any cell in the body
as the organism continues to develop…
the cells specialize and have a more limited potential to differentiate
how are stem cells used
stem cells can be used to replace damaged tissues or repair organs destroyed by disease (known as stem cell therapy or regenerative medicine)
what role does dna play in differentiation
each specialized cell uses the sections of DNA that create proteins it needs to function