Unit 9
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Conformations
Different arrangements of atoms are a result of rotating about carbon-carbon single bonds (no change in atom connectivities)
Newman Projection
A representation of one conformation that looks directly down a bond to show the spatial arrangement of bonds on two adjacent atoms
Dihedral Angle
The angle created by the outer two bonds over three consecutive bonds
Temperature Dependence
The rotation of sigma bonds is temperature dependent
Increasing Size Effect on Rotation of Sigma Bonds
Increases the energy barriers between the low and high energy conformations
Bulky groups hinder rotation
Decreasing Temperature Effect on the Rotation of Sigma Bonds
Makes rotation about sigma bonds more difficult
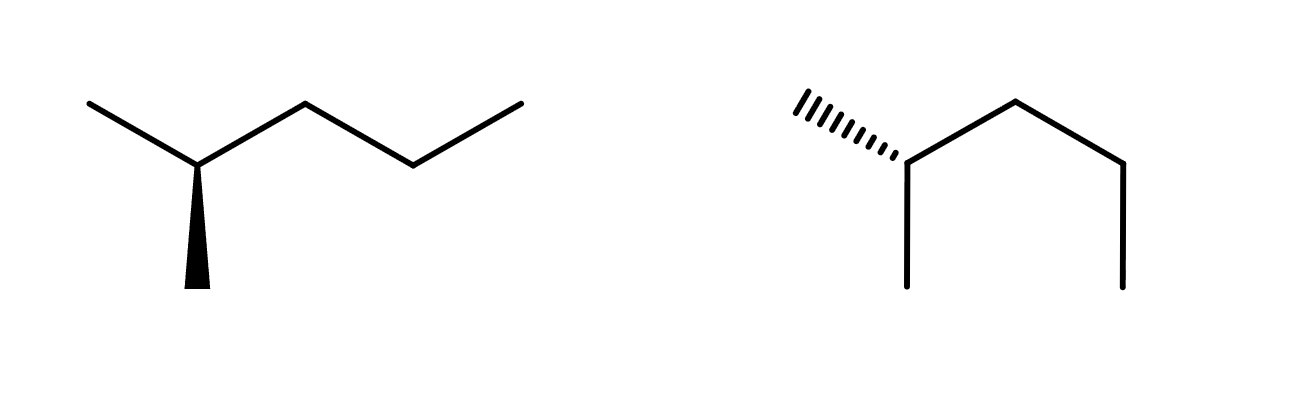
Which term best describes the relationship between the pair of structures shown below?
Identical, but in different conformation
Cycloalkanes
Hydrocarbons that contain one or more rings
With the exception of cyclopropane, the carbons in these rings are not coplanar
Angle Strain
Strain due to deviations from ideal bond angles
Cis Substituents
Are on the same side
Trans Substituents
Are on opposite sides
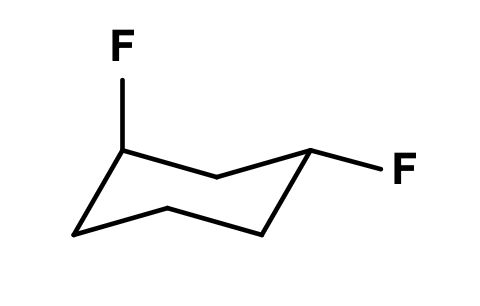
Identify the whether the two fluorine atoms are cis or trans to each other:
trans
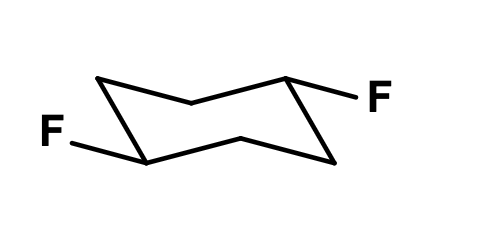
Identify the whether the two fluorine atoms are cis or trans to each other:
trans
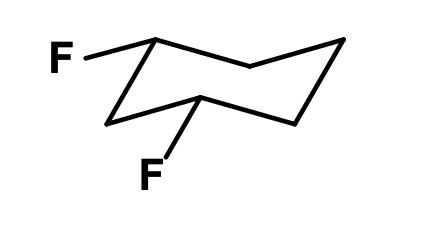
Identify the whether the two fluorine atoms are cis or trans to each other:
cis
Substituted Cyclohexanes
There is no energy difference between the two chair conformations of the unsubstituted cyclohexane
For mono substituted cyclohexanes, there is an energy difference between the two chair conformations due to steric interactions
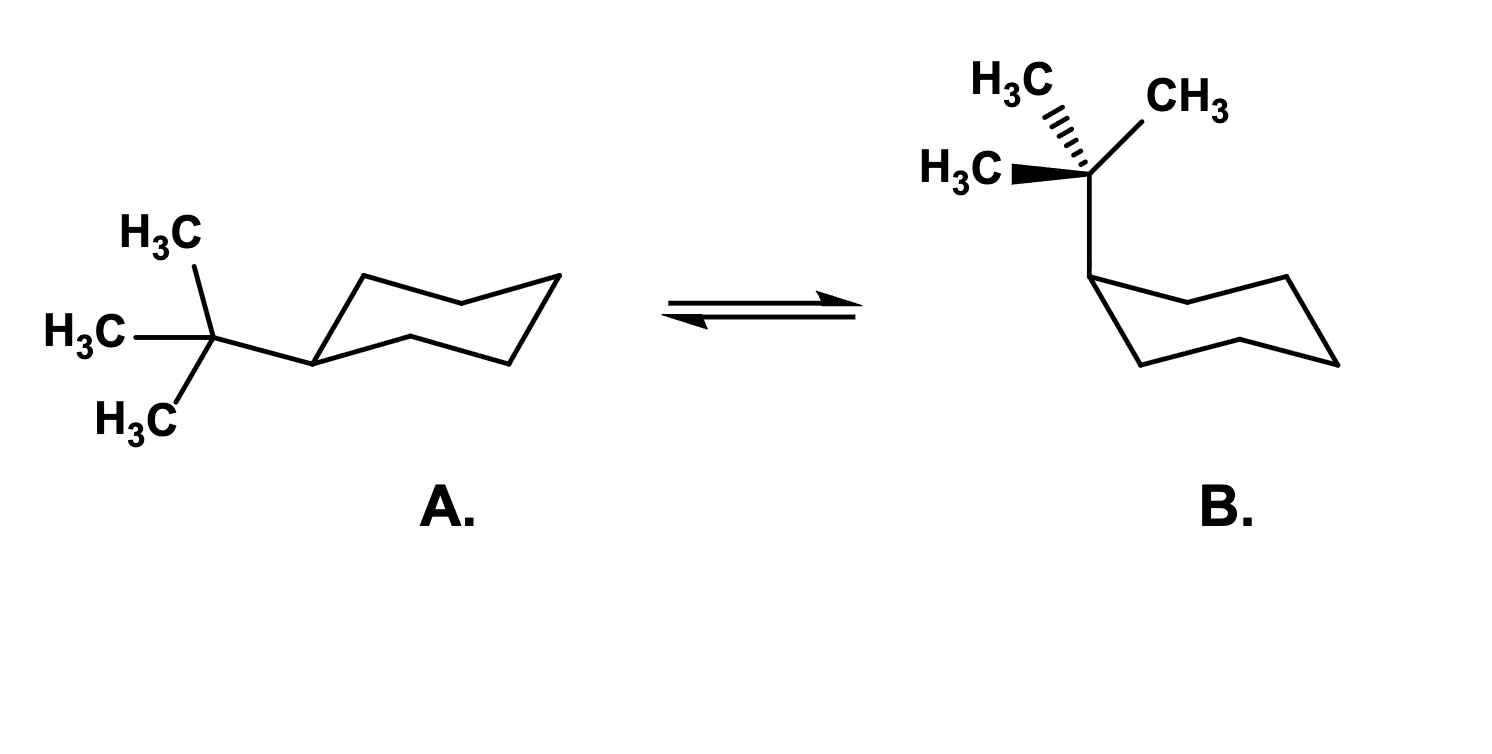
Which of the two chair conformations is favored at equilibrium?
A
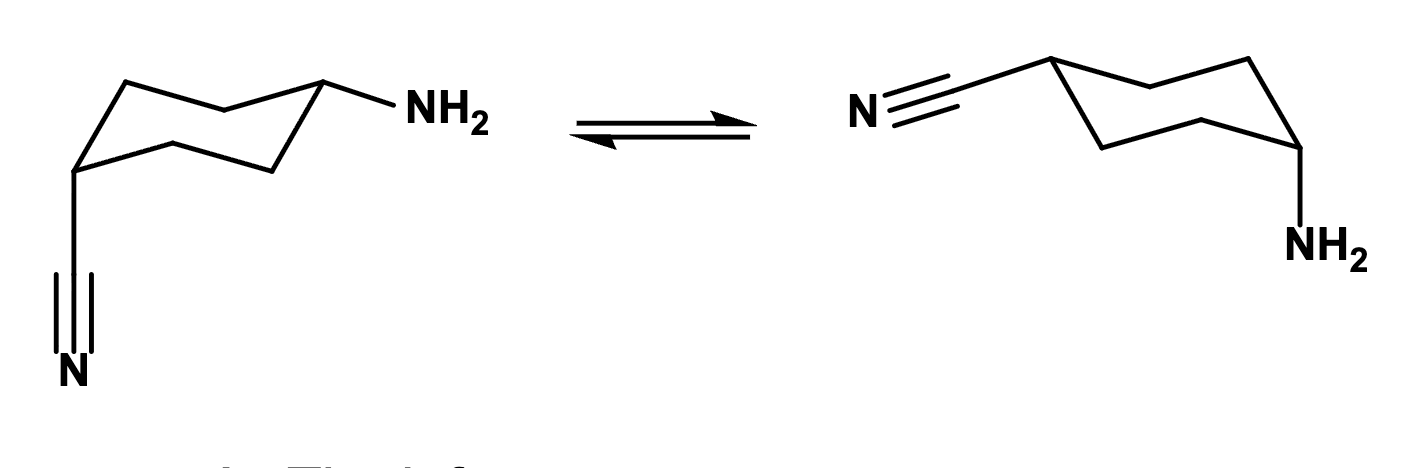
Which of the following conformations is favored at equilibrium?
The left
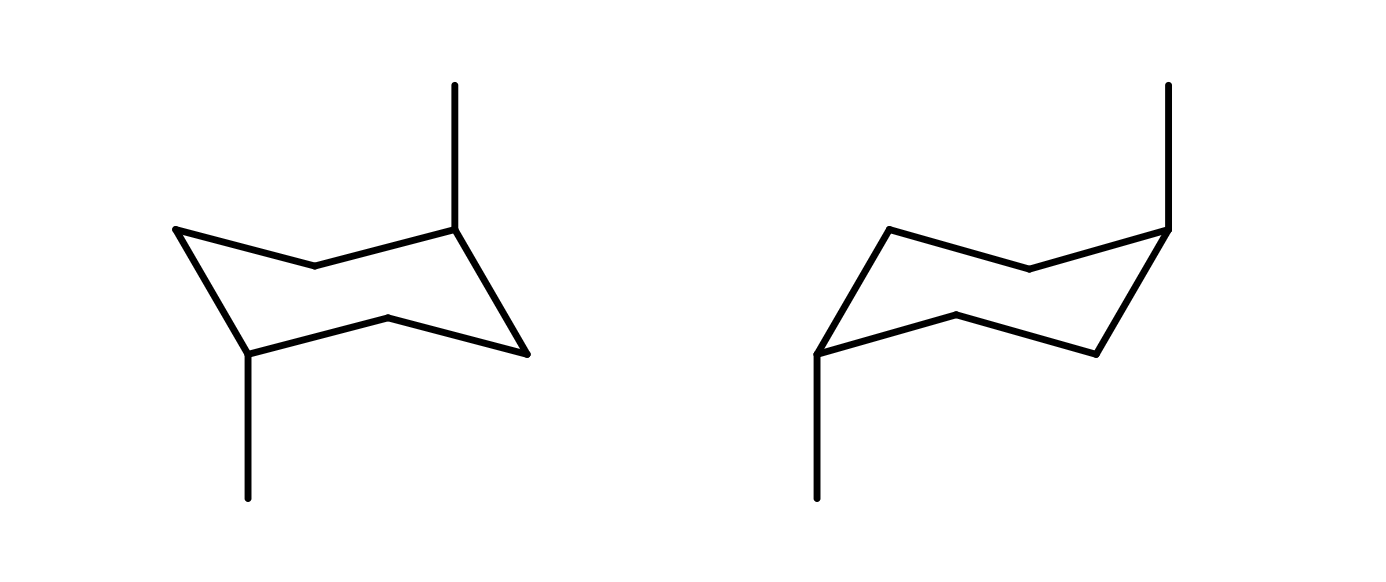
How are these two structures related?
They are the same compound