5 Matter and Thermal
1/17
Earn XP
Description and Tags
CIE Physics IGCSE
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Explain, in terms of momentum, how molecules exert a pressure on the walls of a container. [4]
M1: Gas molecules move randomly and collide with the walls of the container.
M2: During each collision the molecule undergoes a change in momentum (change in p).
M3: Force is the rate of change of momentum.
F = change in momentum / timeM4: Pressure is force acting per unit area.
P = F / A
Explain the cooling effect of evaporation in terms of molecules. [4]
M1: The fastest (highest energy) molecules escape from the surface of the liquid.
M2: These molecules have enough energy to overcome forces of attraction.
M3: The average kinetic energy of the remaining molecules decreases.
M4: Temperature is proportional to average kinetic energy, so the temperature falls.
Explain the purpose of gaps in steel bridge sections and the consequence of omitting them. [3]
M1: Steel expands when heated (thermal expansion).
M2: Gaps allow the sections to expand without pressing against each other.
M3: Without gaps, large forces would build up and the bridge could buckle or deform.
Explain why the temperature of melting ice remains constant at 0 degrees Celsius even though thermal energy is absorbed. [3]
M1: Thermal energy is used to break or weaken intermolecular bonds in the ice lattice.
M2: This increases the potential energy of the molecules.
M3: The average kinetic energy does not increase, so the temperature remains constant.
Describe the two mechanisms of thermal conduction in a metal. [3]
M1: Lattice vibration – atoms at the hot end vibrate more and pass energy to neighbouring atoms.
M2: Free electrons gain kinetic energy at the hot end.
M3: These electrons move through the metal and carry energy to colder regions
Explain the random zig-zag motion of smoke particles (Brownian motion). [4]
M1: Smoke particles are bombarded by much smaller air molecules.
M2: Air molecules move very fast in random directions.
M3: Collisions are uneven or unbalanced at any moment.
M4: This produces a resultant force causing random motion of the smoke particle.
Explain two design changes that make a liquid-in-glass thermometer more sensitive. [3]
M1: Use a larger bulb so more liquid expands.
M2: Use a narrow capillary tube so a small volume expansion gives a large movement.
M3: Use a liquid with high expansivity.
Explain how a convection current forms in a beaker of water heated from below. [3]
M1: Water at the bottom is heated and expands.
M2: Its density decreases so the warm water rises.
M3: Cooler, denser water sinks to replace it, forming a convection current.
Using kinetic theory, explain why the pressure of a gas increases when heated at constant volume. [3]
M1: Gas molecules gain kinetic energy and move faster.
M2: They collide with the container walls more frequently.
M3: Collisions are more forceful, increasing pressure.
Compare the cooling rate of a matte black object and a polished silver object in a vacuum. [3]
M1: The matte black object cools faster.
M2: Matte black surfaces are good emitters of infrared radiation.
M3: Polished silver is a poor emitter and good reflector, so it loses heat more slowly.
Explain, in terms of forces and motion of the air particles, the cause of the motion of the air particles. (4)
M1 air molecules bombard smoke particles
M2 air molecules are small compared to smoke particles
M3 air molecules are very fast moving
M4 air molecules move in random directions
M5 collisions exert unbalanced forces on smoke particles
State and explain the TWO ways that thermal energy is conducted in a metal. (3)
M1: delocalised / free / mobile electrons
M2: electrons carry energy, moving through the metal
M3: lattice vibrations transfer energy to neighbouring particles
Describe how energy from the Sun reaches the Earth and warms the road. (3)
M1: radiation
M2: light / infrared / electromagnetic (radiation)
M3: travel through space / vacuum
M4: absorbed by road
Describe how warming affects the molecules of the air. (2)
M1: they / molecules speed up OR gain kinetic energy
M2: they / molecules move further apart
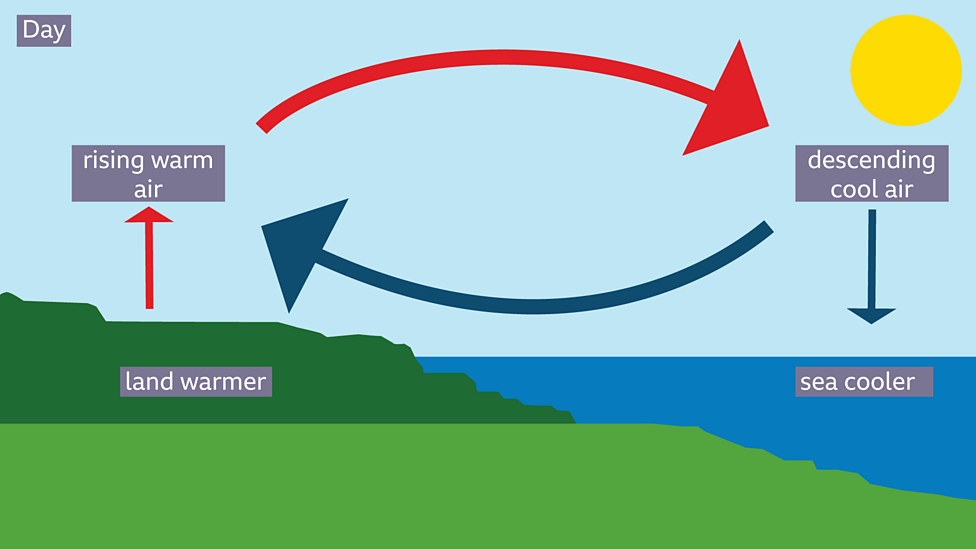
Explain how convection produces a sea breeze when the land is warmer than the sea. (3)
M1: density (of air above road) decreases OR density (of hot air) decreases
M2: air (above land / road) rises OR air (that is hot) rises
M3: air (above road) replaced by cool
air / air from above sea
Dish A is in sunlight with NO wind.
Dish B is in sunlight with wind.
Dish C is in a dark room.
Explain, in terms of particles, why the three dishes have different volumes of water after some time. (4)
M1: energy from the Sun transfers to / is absorbed by (water) molecules, (so KE of (water) molecules increases)
M2: molecules with high(er) energy / KE / fast(er) moving molecules escape (from the surface)
M3: wind removes molecules when they have left the surface (so they do not re-enter the liquid)
M4 (any one from):
1. wind increases the rate of evaporation
2. (absorption of) energy from the Sun increases the rate of evaporation
least / less water evaporates / lower rate of evaporation from dish C
most / more water evaporates / higher rate of evaporation from dish B
Explain, in terms of molecules, why a liquid expands when heated. (2)
M1: molecules / they speed up or gain kinetic energy
M2: molecules move further apart or push others away
Explain, in terms of molecules, why a liquid expands more than a solid when heated. (2)
M1: forces between liquid molecules weak(er than in solids)
M2: less energy / work done to separate molecules or greater separation for same work done / same increase in energy