5.2_Bacterial Metabolism
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
Anabolism
Two Major Components of Metabolism:
Biosynthetic Reactions
Energy-requiring metabolic reactions that build complex molecules (e.g., proteins, nucleic acids) from simpler compounds.
Synthesis of complex molecules from simpler ones
Requires energy input
Examples:
Carbon fixation (CO₂ → glucose or acetyl-CoA)
Amino acid synthesis
Nucleotide synthesis
Purpose:
Cell growth
Repair
Biomolecule production
Anaerobic Respiration
Respiration that uses terminal electron acceptors other than oxygen (e.g., nitrate, sulfate, fumarate).
Electron acceptors: o Nitrate o Sulfate o Fumarate
Lower ATP yield than aerobic respiration
Assimilatory Reduction
The reduction of inorganic compounds (e.g., nitrate or sulfate) for incorporation into cellular biomass rather than for energy production.
Autotroph
An organism that uses carbon dioxide (CO₂) as its primary carbon source.
use CO₂ as their sole or primary carbon source, reducing it into organic compounds via carbon fixation pathways.
They require: • An energy source • A source of reducing power
Carbon source: CO₂
Examples: ▪ Cyanobacteria ▪ Nitrifying bacteria ▪ Sulfur-oxidizing bacteria
Biogeochemical Cycle
The circulation of chemical elements (e.g., carbon, nitrogen, sulfur) between biological, geological, and chemical components of Earth.
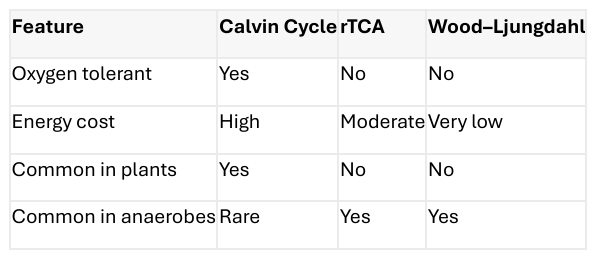
Calvin–Benson–Bassham (Calvin) Cycle
A carbon fixation pathway that incorporates CO₂ into organic carbon using ATP and NADPH; common in plants and cyanobacteria.
The most widespread carbon fixation pathway
Used by: o Cyanobacteria o Algae o Plants o Some chemoautotrophic bacteria
Operates under aerobic conditions
Requires: o ATP o NADPH
Produces triose phosphates, which feed into biosynthesis
Key Enzyme: RuBisCO
Carbon Fixation
The conversion of inorganic carbon (CO₂) into organic compounds by living organisms.
is the process by which inorganic carbon (CO₂) is converted into organic molecules that can be used to build cellular components such as carbohydrates, lipids, proteins, and nucleic acids.
Catabolism
Two Major Components of Metabolism:
Energy-Yielding Reactions
Metabolic reactions that break down complex molecules to release energy.
supports anabolism.
Breakdown of chemical compounds
Releases energy (often stored as ATP, NADH, FADH₂)
Examples:
Glucose oxidation
Sulfur oxidation
Hydrogen oxidation
Ammonia oxidation
Purpose:
Provide energy for cellular work
Generate precursor molecules for biosynthesis
Chemolithoautotroph
An organism that obtains energy from the oxidation of inorganic compounds and carbon from CO₂.
are invisible primary producers and are as important as the photoautotrophs.
Energy from oxidation of inorganic chemicals
Carbon source: CO₂
Examples: • Nitrosomonas (ammonia oxidizer) • Thiobacillus (sulfur oxidizer)
Chemoorganotroph
An organism that obtains energy and electrons from organic compounds.
Denitrification
An anaerobic process in which nitrate (NO₃⁻) is reduced to gaseous nitrogen (N₂ or N₂O).
Dissimilatory Sulfate Reduction
An anaerobic respiratory process in which sulfate (SO₄²⁻) is reduced to hydrogen sulfide (H₂S) for energy conservation.
Electron Acceptor
A molecule that receives electrons during a redox reaction (e.g., O₂, NO₃⁻, SO₄²⁻).
Electron Donor
A molecule that supplies electrons during a redox reaction (e.g., organic compounds, H₂, H₂S).
Fermentation
An anaerobic metabolic process where organic molecules act as both electron donors and electron acceptors, yielding limited ATP.
No external electron acceptor
Organic molecule serves as final electron acceptor
Produces acids, alcohols, gases
Examples: • Lactic acid fermentation • Alcoholic fermentation • Mixed-acid fermentation
Glycolysis
A central metabolic pathway that converts glucose into pyruvate, producing ATP and NADH.
Glucose → pyruvate
Produces: o ATP o NADH • Occurs in nearly all heterotrophic microbes
Heterotroph
An organism that relies on organic compounds as its carbon source.
Carbon source: organic compounds
use pre-formed organic compounds as their carbon source.
Examples: ▪ Escherichia coli ▪ Fungi ▪ Most pathogens ▪ Decomposer microbes in soil and water
Heterocyst
A specialized, thick-walled cell in some cyanobacteria that provides an oxygen-free environment for nitrogen fixation.
Mixotroph
An organism capable of switching between autotrophic and heterotrophic metabolism depending on environmental conditions.
Nitrogenase
A multi-enzyme complex that catalyzes the reduction of atmospheric nitrogen (N₂) to ammonia (NH₃).
Nitrogen Fixation
The biological conversion of atmospheric nitrogen gas (N₂) into ammonia (NH₃).
Nitrification
A two-step aerobic process in which ammonia is oxidized to nitrite and then to nitrate by chemolithoautotrophic bacteria.
Photoautotroph
An organism that uses light as an energy source and CO₂ as a carbon source.
Energy from light
Carbon source: CO₂
Examples: ▪ Cyanobacteria ▪ Purple sulfur bacteria
Carbon fixation pathway: o Calvin–Benson–Bassham cycle (most common)
Redox Reaction
A chemical reaction involving the transfer of electrons between molecules (reduction and oxidation).
Reverse (Reductive) TCA Cycle
A carbon fixation pathway in which CO₂ is reduced to organic molecules by running the TCA cycle in reverse.
Essentially the TCA cycle run in reverse
Found in: o Green sulfur bacteria o Some ε-proteobacteria o Deep-sea or microaerophilic microbes
Fixes CO₂ to form: o Acetyl-CoA o Pyruvate
More energy-efficient than the Calvin cycle
Requires specific enzymes to bypass irreversible TCA steps
Typically found in: o Anaerobic o Low-oxygen environments
Reflects ancient metabolic origins
This pathway is related to early Earth conditions (low oxygen).
RuBisCO
Ribulose-1,5-bisphosphate carboxylase/oxygenase,
the key enzyme that catalyzes CO₂ fixation in the Calvin cycle.
Inefficient and prone to oxygen interference
Key Enzyme of Calvin–Benson–Bassham (CBB) Cycle
is a critical enzyme in photosynthesis that catalyzes the first major step of carbon fixation—converting atmospheric into organic energy-rich molecules.
It is considered the most abundant protein on Earth, found in plants, algae, and many bacteria.
Sulfur Cycle
The movement and transformation of sulfur between inorganic and organic forms mediated largely by microorganisms.
Sulfur Oxidation
The oxidation of reduced sulfur compounds (e.g., H₂S, S⁰) to sulfate (SO₄²⁻), often coupled to energy generation.
Tricarboxylic Acid (TCA) Cycle
A central metabolic pathway that oxidizes acetyl-CoA to CO₂ while generating reducing equivalents for respiration.
Oxidizes acetyl-CoA → CO₂
Produces: o NADH o FADH₂ o Precursors for amino acid biosynthesis
Central hub of metabolism
In many microbes, the ____ is amphibolic (both catabolic and anabolic).
Wood–Ljungdahl Pathway (Acetyl-CoA Pathway)
An anaerobic carbon fixation pathway that reduces CO₂ directly to acetyl-CoA, used by acetogens and methanogens.
One of the simplest and most ancient carbon fixation pathways
Used by: o Acetogenic bacteria o Methanogenic archaea
Two branches: 1. Methyl branch (CO₂ → methyl group) 2. Carbonyl branch (CO₂ → CO)
Combines methyl and carbonyl groups to form acetyl-CoA
Important in: o Anaerobic sediments o Animal digestive systems
Thought to resemble some of the earliest metabolic pathways on Earth
Microbial metabolism
refers to the complete set of chemical reactions that occur within microbial cells that allow them to:
Obtain energy
Build cellular components
Maintain cell structure and function
Grow and reproduce
Interact with and modify their environment
underpins life on Earth and drives major element cycles (C, N, S, P).
extraordinary metabolic diversity
Unlike plants and animals, microbes display ______, allowing them to survive in environments ranging from oxygen-rich soils to deep-sea hydrothermal vents, acid mine drainage, and animal guts.
energy input
Anabolism Requires ____
nitrate (nitrifiers)
Examples of Unusual Microbial Metabolisms:
Oxidation of ammonia →
hydrogen sulfide
Examples of Unusual Microbial Metabolisms:
Reduction of sulfate →
NH₃
Examples of Unusual Microbial Metabolisms:
Fixation of nitrogen gas (N₂ →
Chemotrophs
Energy from chemical compounds
Organotrophs
electrons from organic compounds
Lithotrophs
electrons from inorganic compounds (e.g., H₂, H₂S, NH₃)
Carbon cycle
Carbon fixation
Respiration
Decomposition
Nitrogen cycle
Nitrogen fixation
Nitrification
Denitrification
Sulfur cycle
Sulfate reduction
Sulfur oxidation
CO₂
is abundant but chemically stable and biologically unusable without fixation
base of food webs
Fixed carbon forms the ____
ATP & NADPH
Calvin–Benson–Bassham (CBB) Cycle requires:
triose phosphates
Calvin–Benson–Bassham (CBB) Cycle produces ___, which feed into biosynthesis
Carboxylation
Three Main Phases of Calvin–Benson–Bassham (CBB) Cycle:
___-CO₂ fixation
Reduction
Three Main Phases of Calvin–Benson–Bassham (CBB) Cycle:
___-conversion to G3P
Regeneration of RuBP
Three Main Phases of Calvin–Benson–Bassham (CBB) Cycle:
*third phase*
Acetyl-CoA & Pyruvate
Reverse (Reductive) Tricarboxylic Acid (rTCA) Cycle Fixes CO₂ to form:
Methyl branch (CO₂ → methyl group)
Carbonyl branch (CO₂ → CO)
Two branches of Wood–Ljungdahl Pathway (Acetyl-CoA Pathway):
acetyl-CoA
Wood–Ljungdahl Pathway (Acetyl-CoA Pathway) Combines methyl and carbonyl groups to form ___
methyl and carbonyl groups
Wood–Ljungdahl Pathway (Acetyl-CoA Pathway) Combines ______to form acetyl-CoA
3-Hydroxypropionate Cycle
Found in some green non-sulfur bacteria
Fixes CO₂ into organic acids
3-Hydroxypropionate/4-Hydroxybutyrate Cycle
Found mainly in archaea
Adapted to:
High temperature
Extreme environments
Sugars (glucose, lactose) • Organic acids (acetate, lactate) • Amino acids • Lipids • Complex polymers (cellulose, lignin)
Examples of organic carbon sources:
central metabolism
Heterotrophic metabolism centers on a small number of core metabolic pathways, often called ____
ATP & NADH
Glycolysis (Embden–Meyerhof–Parnas Pathway) Produces:
Entner–Doudoroff pathway
common in Gram-negative bacteria
is an alternative metabolic route to glycolysis used by many prokaryotes to catabolize glucose into pyruvate.
It yields less energy than standard glycolysis (net 1 ATP per glucose), but requires fewer enzymes, making it efficient for specific bacteria like Pseudomonas.
NADH, FADH₂, and Precursors for amino acid biosynthesis
Tricarboxylic Acid (TCA) Cycle produces:
Aerobic respiration
Final electron acceptor: O₂
High ATP yield
Nitrate, Sulfate, and Fumarate
Electron acceptors for Anaerobic respiration:
acids, alcohols, gases
Fermentation produces:
Decompose organic matter
Recycle carbon back to CO₂
Form the base of microbial food webs
Critical in soil, gut, and aquatic ecosystems
Ecological Role of Heterotrophs
Ammonia (NH₃) • Nitrite (NO₂⁻) • Hydrogen sulfide (H₂S) • Elemental sulfur • Ferrous iron (Fe²⁺)
Common inorganic energy sources:
energy-intensive
Autotrophs face several constraints:
CO₂ fixation is ____
oxygen-sensitive
Autotrophs face several constraints:
Enzymes may be ___
Decomposers, consumers
Ecological role of Heterotrophs
Primary producers
Ecological role of Autotrophs
Soils
Linking Carbon Metabolism to Ecosystems:
Autotrophs introduce new organic carbon
Heterotrophs decompose plant and microbial biomass
Oceans
Linking Carbon Metabolism to Ecosystems:
Cyanobacteria and chemoautotrophs dominate primary production
Extreme environments
Linking Carbon Metabolism to Ecosystems:
Autotrophic microbes often form the base of food chains