Nuclear physics
1/59
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
What does Einstein’s mass-energy equation mean?
Mass and energy are equivelent
What are the two interpretations of Einstein’s mass-energy equation?
mass is a form of energy
energy has mass
Define rest mass
The mass of an object, such as a particle, when it is stationary
Why is the rest mass of an electron different to an electron in a particle accelerator?
The electron in the particle accelerator is moving so has more energy than an electron at rest
Since ∆E = ∆mc², the mass of the electron in the particle accelerator must be greater than the rest mass of an electron
Explain why energy is released in alpha decay
The total mass of the alpha particle and daughter nucleus are less than the parent nucleus. As such total mass decreases.
Since the mass has changed, some energy must be released during the decay for mass-energy to be conserved
Energy is released (not absorbed) as the mass decreases
Explain why energy is released in beta decay
The total mass of the electron or positron and daughter nucleus are less than the parent nucleus. As such total mass decreases.
Since the total mass has changed, some energy must be released during the decay for mass-energy to be conserved
Energy is released as the mass decreases
What is the value of an atomic mass unit?
1 u = 1.661 × 10–27 kg
How would you calculate the binding energy from the mass defect?
Binding energy = mass defect × c²
Explain why the binding energy is different for different nuclei.
Different nuclei have different numbers of nucleons so have different bingding energies
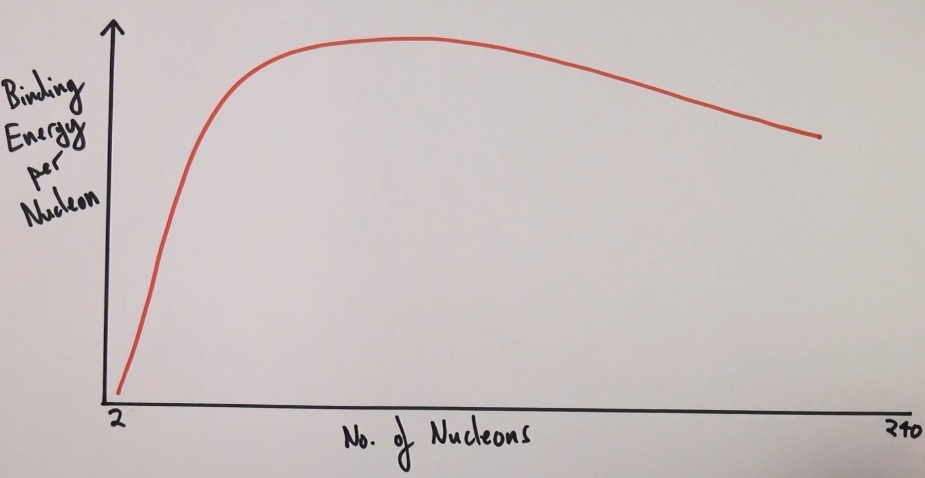
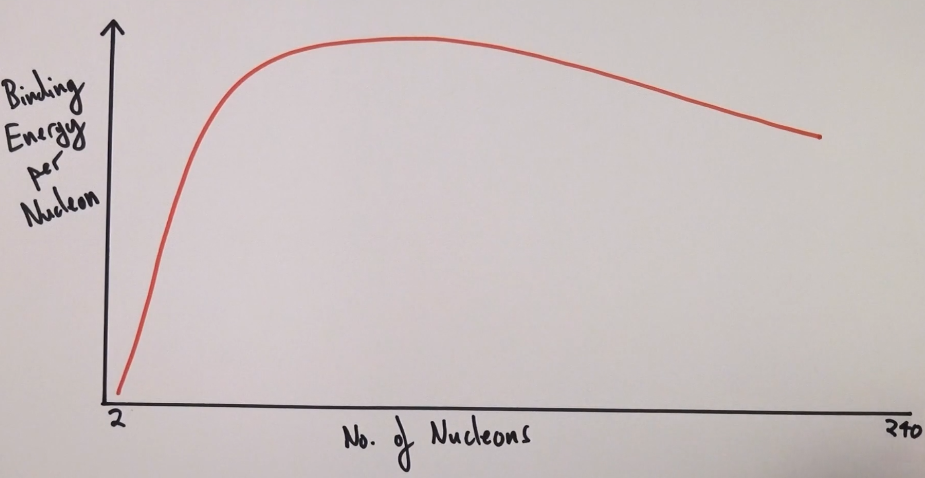
Draw a sketch of binding energy per nucleon against nucleon number.
Where on a sketch of binding energy per nucleon against nucleon number does fission take place?
To the right of iron
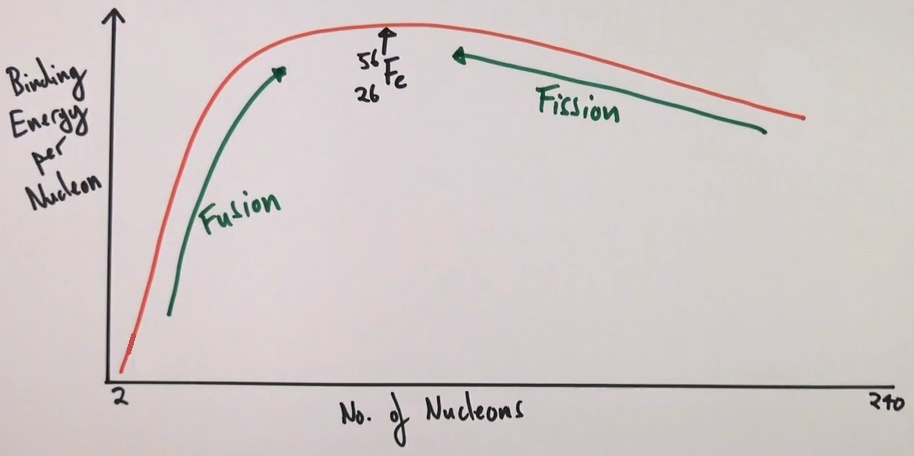
Where on a sketch of binding energy per nucleon against nucleon number does fusion take place?
To the left of iron
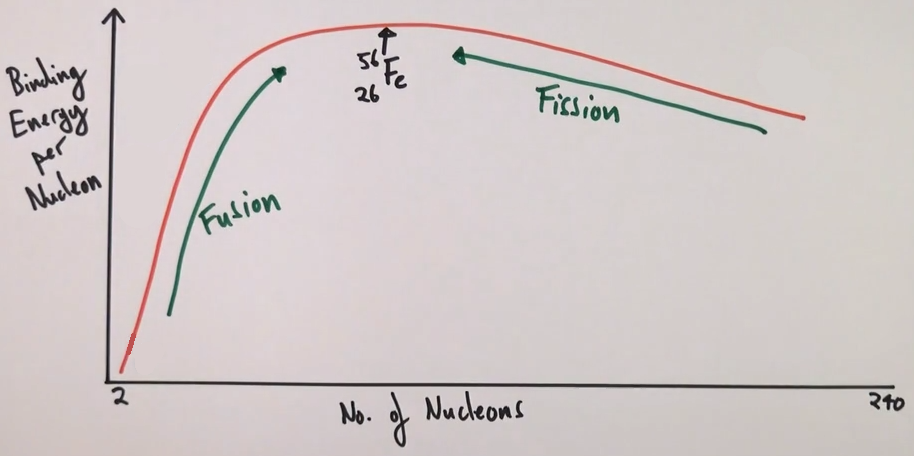
Which is the most stable nuclei?
iron-56
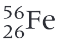
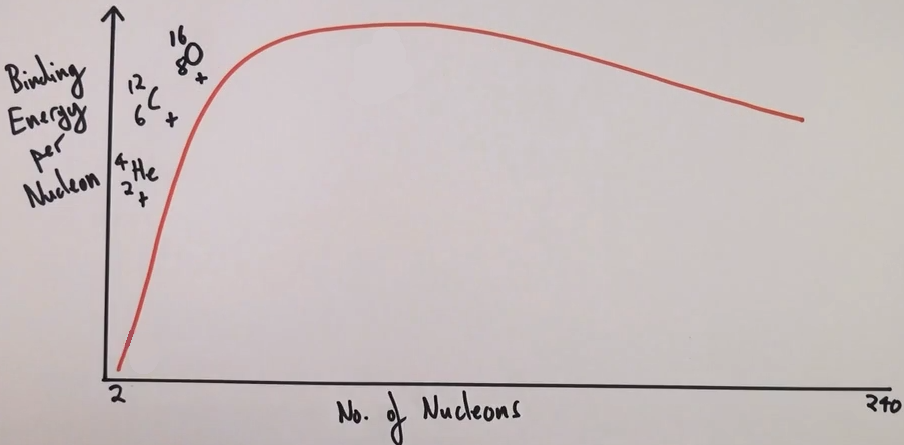
Which three elements don’t fit the curve?
Helium, oxygen and carbon
What is the composition of mined Uranium?
99.3% uranium-238
0.7% uranium-235
Why are thermal neutrons needed in a fission reactor?
The fuel in fission reactors can split spontaneously without neutrons, but this is unlikely.
Thermal neutrons are needed to induce fission: the neutrons are absorbed by the fuel, making it unstable and causing it to decay.
Define Fission
The splitting of a nucleus into two smaller nuclei
What is induced fission?
The splitting of a nucleus as a neutron is absorbed
What is fusion?
When two smaller nuclei join together to form one larger nucleus
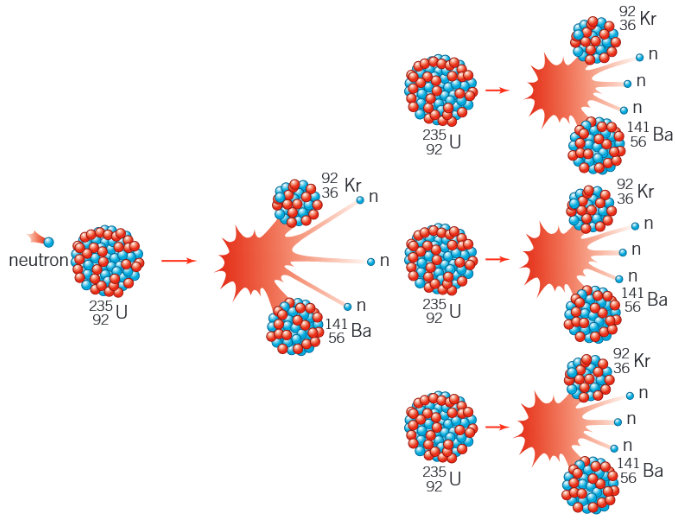
What is the decay equation for the induced fission of uranium-235 by a thermal neutron?

Draw a diagram to show an uncontrolled fission reaction
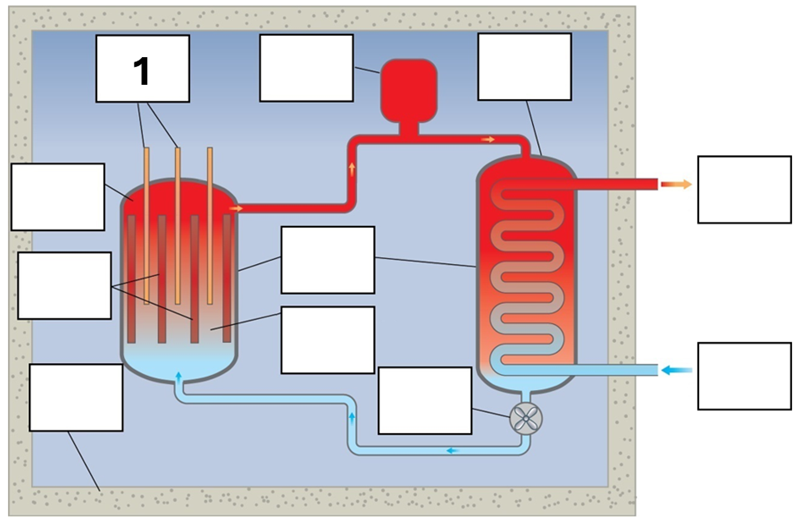
What is component 1 in this fission reactor?
Control rods – boron/cadmium
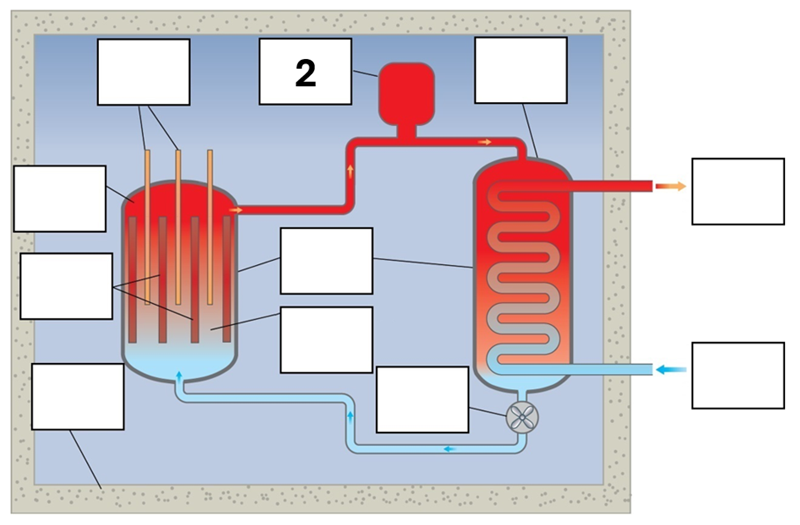
What is component 2 in this fission reactor?
Pressuriser
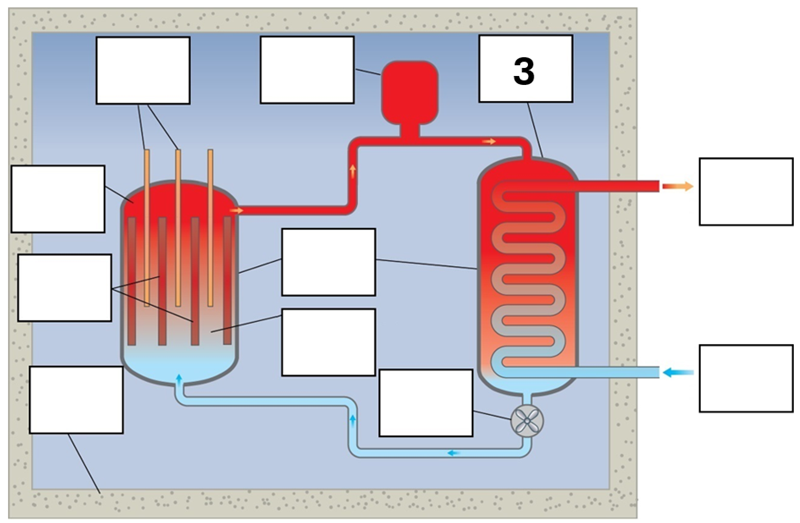
What is component 3 in this fission reactor?
Heat exchanger
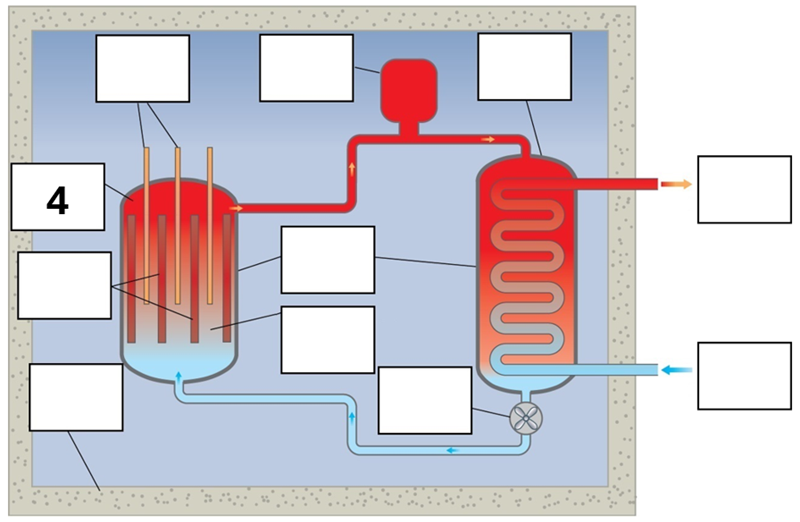
What is component 4 in this fission reactor?
Reactor core
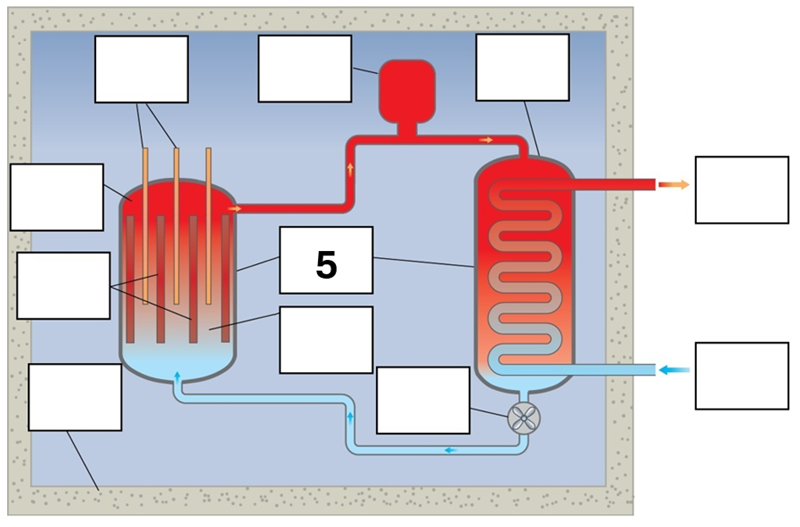
What is component 5 in this fission reactor?
Vessel – steel
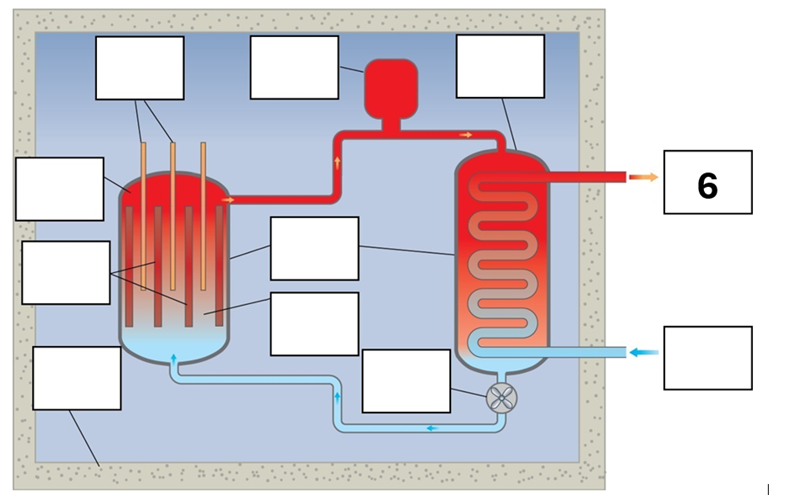
What is component 6 in this fission reactor?
Steam to turbines
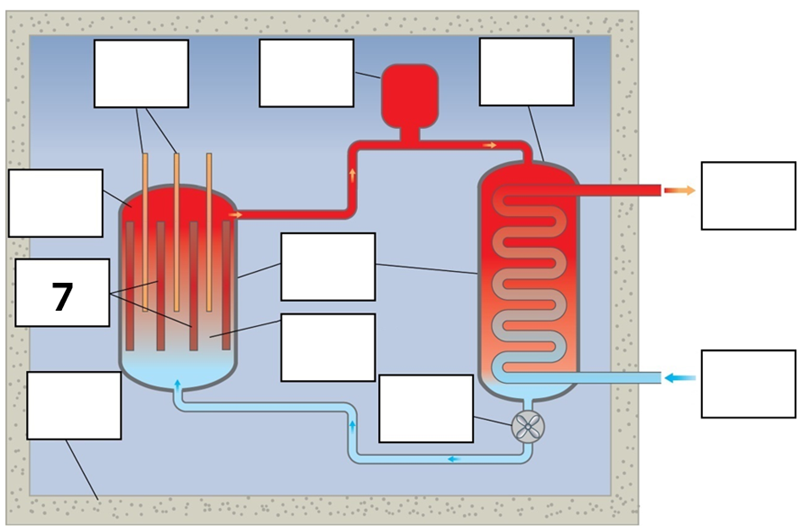
What is component 7 in this fission reactor?
Fuel rods – Enriched uranium, that is, extra U235 added
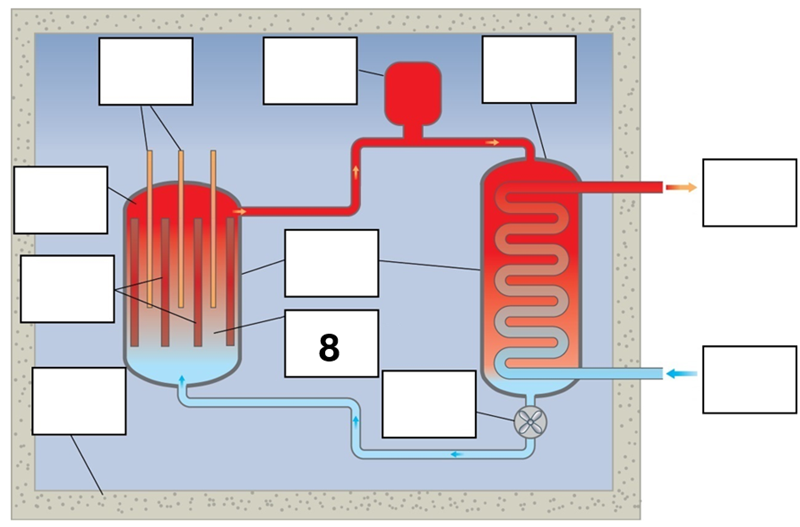
What is component 8 in this fission reactor?
Moderator (graphite/water) and coolant (water)
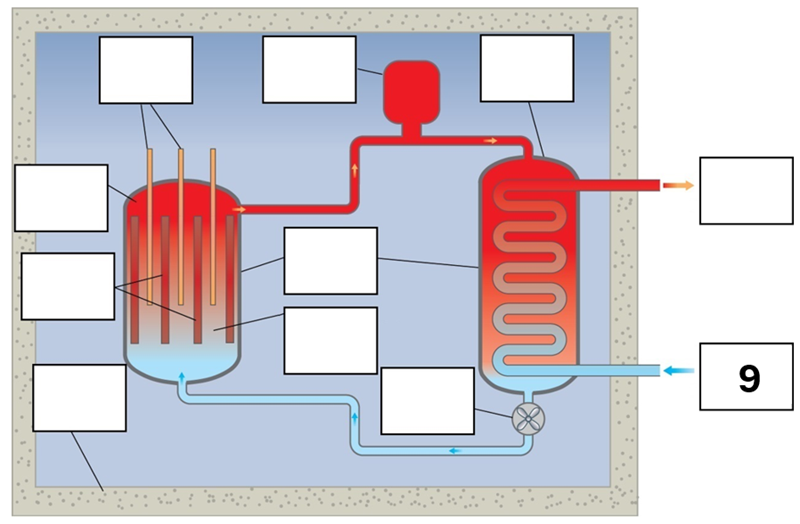
What is component 9 in this fission reactor?
Water from turbines
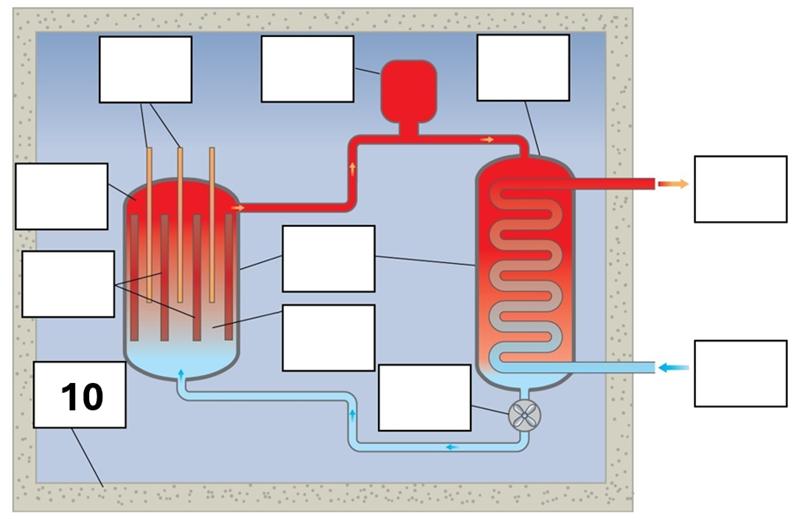
What is component 10 in this fission reactor?
Shielding – reinforced concrete
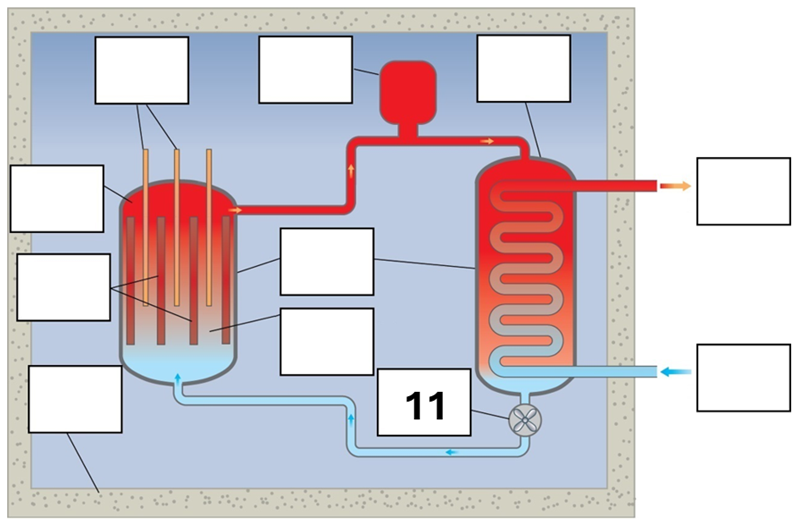
What is component 11 in this fission reactor?
Pump
What is the purpose of the coolant in a fission reactor?
Takes heat from the reactor (produced from the fission reactions) and tranfers it to the heat exchanger
What is the purpose of the moderator in a fission reactor?
The fast neutrons produced in fission reactions elasticaly collide with moderator atoms which have small mass.
Energy is transferred from neutrons to atoms.
This slows down the fast neutrons to produce thermal neutrons
Why is a moderator needed in a fission reactor?
Induced fission of uranium-235 produces fast neutrons.
The chance of these fast neutrons being absorbed by uranium-235 nuclei is quite small, whereas thermal neutrons have a greater chance of producing induced fission.
As such a moderator is needed to slow down the fast neutrons and produce thermal neutrons
What is the purpose of the control rods in a fission reactor?
Absorbs neutrons to control the rate of the reaction and maintain it at a steady rate where 1 neutron per fission goes on to produce further fission
What is the purpose of the fuel rods in a fission reactor?
To undergo fission and release energy when hit with neutrons
What is its purpose of the shielding in a fission reactor?
Reduce intensity of gamma rays.
Absorb some neutrons. (No effect on neutrinos since do not interact with matter.)
What properties do the shielding in a fission reactor have?
Often thick and high density to absorb gamma rays
Withstands high temperatures
Why is enriched uranium used in the fuel rods?
So that the fuel rods contain enough Uranium-235 to sustain continous fission reactions
What is a chain reaction?
When each fission reaction produces more neutrons which go on to induce further fission reactions
This leads to an exponential increase in the rate of fission reactions
What are the benefits of fission reactors?
They don’t emmit CO2
A small quantity of fuel produces large amounts of energy
What are the environmental impacts of fission reactors?
They produce hazardous and radioactive waste materials such as plutonium 239.
These materials can have half lives which are tens of thousands of years
These materials cannot enter our water or food suplies so must be buried underground for centuries
Why is it difficult to get fusion reactors to work?
Extremely high temperatures are needed for fusion to occur. It is very hard to maintain such temperatures for long enough to sustain fusion.
It is very hard to confine the extremely hot fuel in a reactor
What is deuterium?
An isotope of hydrogen which contains one proton and one neutron in its nucleus

What is the nuclear equation for the fusion of two protons?
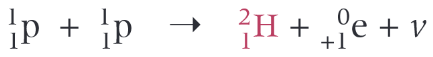
What is the equation for the fusion of a proton and deuterium nucleus?
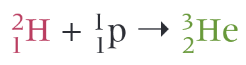
What is the equation for the fusion of two Helium-3 nuclei?

Show the equations for proton-proton chain.
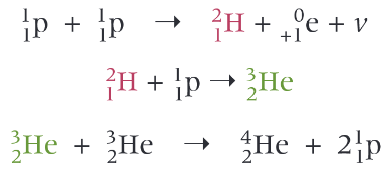
What is the nuclear symbol for a electron?
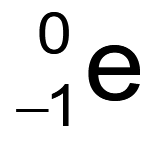
What is the nuclear symbol for a positron?
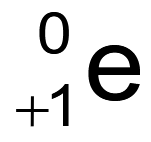
What is the binding energy of a nucleus?
The minimum energy required to completely separate a nucleus into its constituent protons and neutrons
Define binding energy per nucleon
The binding energy divided by the number of protons and neutrons in the nucleus
Define the mass defect
The difference between the mass of the separate nucleons and the mass of the nucleus
Define annihilation
The complete destruction of a particle and its antiparticle when they meet, releasing energy in the form of identical photons
What happens when an electron and positron meet?
They annihilate each other
Their mass transforms into energy in the form of 2 identical photons
An electron/ positron pair annihilate. Derive the minimum energy of each photon produced in terms of me (the rest mass of an electron/positron)
Minimum energy as we assume they have 0 KE
Δm = 2me
ΔE = Δmc² = 2mec²
mimimum energy of the 2 gamma photons = 2mec²
minimum energy of each gamma photon = mec²
What is pair production?
The replacement of a single photon with a particle and corresponding antiparticle of the same total energy
What are the similarities between fission and fusion reactions?
Energy is produced in both reactions
The total binding energy of products is greater
What are the differences between fission and fusion reactions?
More energy produced per reaction in fission
In fusion, nuclei repel each other
Fusion requires high temperatures, fission does not
Fission reactions are triggered by neutrons, fusion is not
Chain reaction is possible in fission but not fusion