chem 104 ray quiz 5
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
for electrolytic cell, mass of the anode will increase during reaction
f
Pt may be used as an inert electrode for the anode without changing the products
f
The concentration of anode ions will increase during the course of this reaction
t
A power source of 1 V will be sufficient to allow this cell to work as electrolytic (0.34 and -0.76)
f, must be 1.1
Zinc metal will be oxidized (Cu=0.34, Zn=-0.76)
f, zinc is cathode
The arrhenius factor, A, must be exp determined
t
The higher the temp of a system, the less energy in collisions
f
Adding a catalyst lowers the activation energy of the system, allowing more collisions to have the min energy needed
t
An increase in the number of collisions in a reaction indicates a the solution has been diluted (at constant temperature)
f
Activation energy will always be positive
t
(A→products) if you plot [A] vs time for a zero-order process the slope of the resulting line will be negative
t
(A→products) With the proper integrated rate law you would be able to determine how long it would take for the reaction to reach equilibrium
t
(A→products) If you plot [A] vs time and find a straight line relationship, the initial concentration of “A” can be found by taking the natural log (ln) of the y-intercept
f
(A→products) if this process is first order the concentration will decrease linearly over time
f
(A→products) the units of the rate constant, k, will depend on the order of the reaction
t
electrons are pushed to the cathode by the power source
t
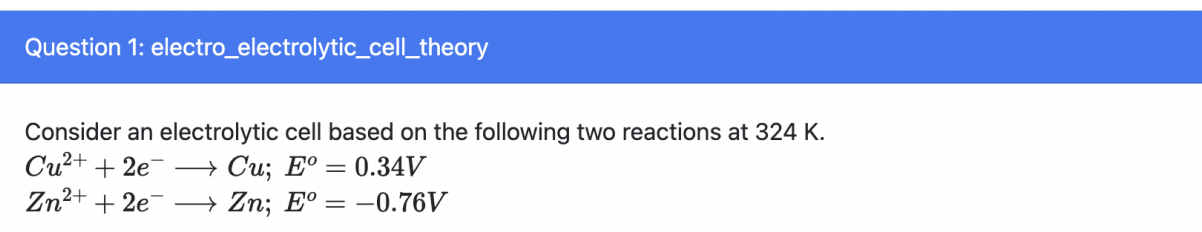
The concentration of Cu2+ ions will increase
t
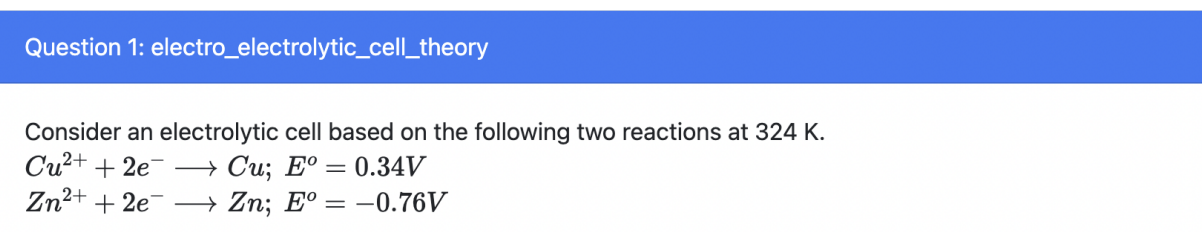
The concentration of Zn2+ ions will decrease
t
Pt may be used as an inert electrode for the anode without changing the products of the reaction
f
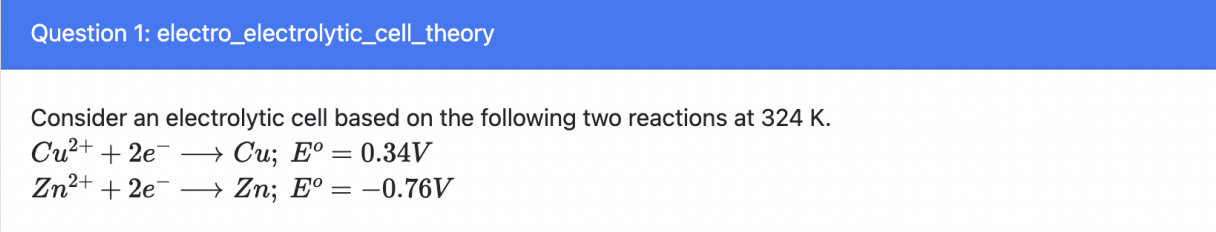
The reaction will spontaneously form Cu2+ ions during the reaction
f, electrolytic can’t be spontaneous
most collisions do not lead to a chemical reaction
t
activation energy is the max energy needed for a collision to be successful
f
activation energy will always be positive
t
heating a reaction increases reaction rate by decreasing the energy in the collisions
f
the arrhenius factor, A, must be experimentally determined
t
the plot of a second-order integrated rate law would have time on the Y and 1/[A] on the X
f, x and y are flipped
the first-order integrated rate law will directly allow us to determine the rate of a first-order reaction
t
with the proper integrated rate law you would be able to determine how long it would take for the reaction to reach equillibrium
t
the units of the rate constant, k, will depend on the order of the reaction
t
adding water/diluting all species will increase amount of work
t
increasing reactant concentration will increase amount of work
t

adding solid NaOH will increase work
t, reacts with h3o and removes products
(electrolytic cell) electrons will be pushed to the cathode portion of the cell by the power source
t

the mass of the copper metal will increase during the course of the reaction
f

Pt may be used as an inert electrode for the anode w/o changing products of the reaction
f

the concentration of zn2+ ions will decrease during the course of the reaction
t

Pt may be used as an inert electrode for the cathode w/o changing the products of the reaction
t
the higher the temperature of a system, the less energy in collisions
f
adding a catalyst lowers the activation energy of the system, allowing more collisions to have the minimum energy needed
t
the arrhenius factor, A, must be experimentally determined
t
activation energy will always be positive
t
most collisions do not lead to a chemical reaction
t
If you plot [A] vs time and find a straight line relationship, the initial concentration of “A” can be found by taking the natural log (ln) of the y-intercept
f, initial concentration is just [A]0
(A → products) If this process is first order the concentration will decrease linearly over time
f
The plot of a second-order integrated rate law would have the time on the y axis and 1/[A] on the x axis
f
The plot of [A] vs time for a zero-order process will give a straight line with a positive slope
f, negative
If you plot [A] vs time for a second-order process the result will be a curved line
t

decrease concentration of Fe3+
t

increase mass of Sn
f

decrease concentration of Sn2+
f

add equal amounts of water to each half cell
f

increase the concentration of Sn2+
t