High Yield MCAT Chem - Johan
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
rules for filling orbitals
Pauli exclusion principle- no two electrons in the same atom can have the same quantum numbers, same spin
aufbau principle- electrons fill in lowest energy orbitals first, two exceptions where s electrons are promoted: copper (and silver, gold) (d9 becomes s1d10) and chromium (and molybdenum) (d4 becomes s1d5)
hund's rule- electrons fill each orbital in a subshell before doubling up
when removing electrons, 4s electrons are removed before 3d to create ions
Fe = 4s2 3d6
Fe2+ = 3d6
isoelectronic
two atoms have same number of electrons
magnetism
paramagnetic- at least one unpaired electron, attracted to an external magnetic field
diamagnetic- all electrons are paired, repelled from an external magnetic field
shielding
valence electrons have lower effective nuclear charge because they are further away from nucleus and are repelled by core electrons
more protons means greater effective nuclear charge
more orbital rings means lower effective nuclear charge
more electrons (greater negative charge) means lower effective nuclear charge
electrostatic attraction
increases from lower left to upper right
1. down a group, valence electrons are farther away means weaker pull
2. across a period, more protons means stronger pull
3. becoming more negatively charged, more shielding means weaker pull
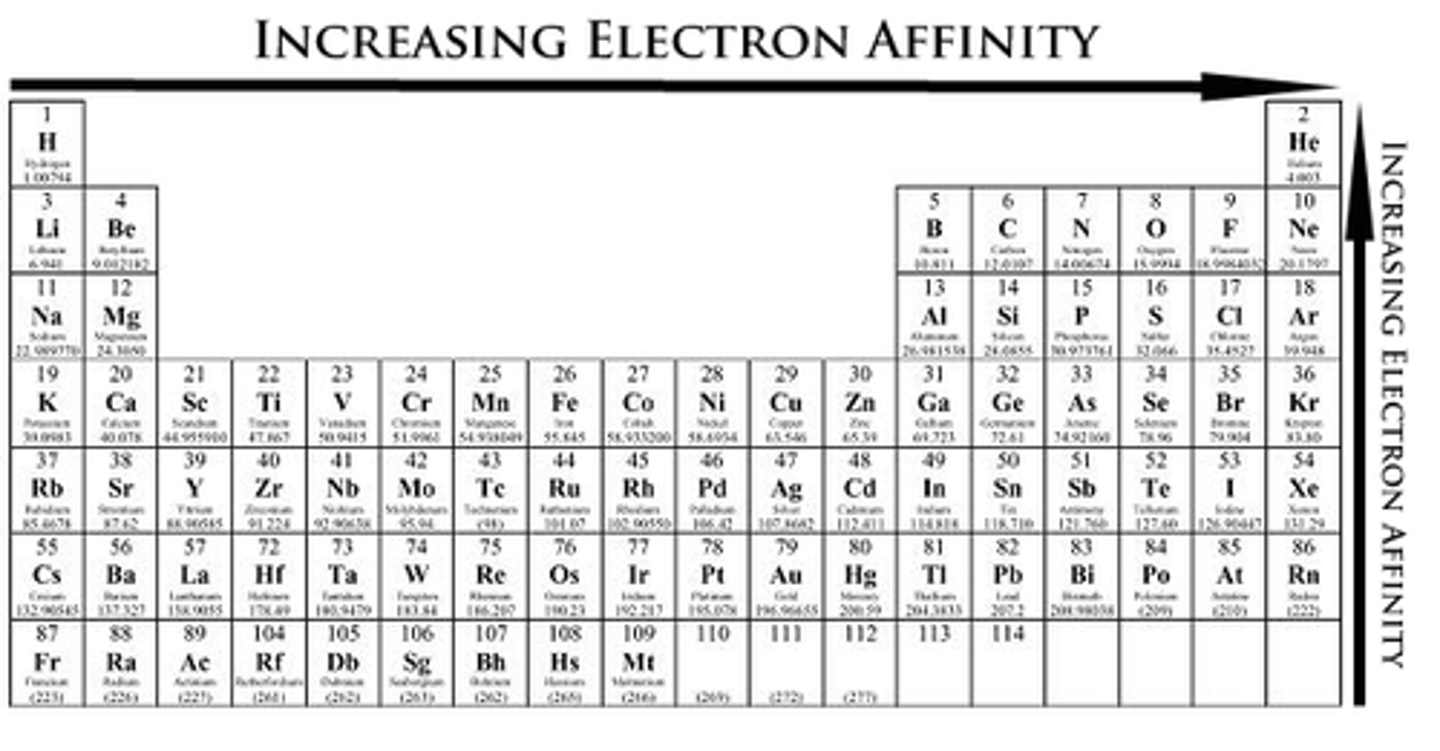
electron affinity
increases from lower left to upper right (value becomes more negative, but magnitude increases)
1. down a group, more rings means less energy released
2. across a period, more protons means more energy released
defined as energy change when adding an electron to valence shell in gas state
high electron affinity means high reduction potential
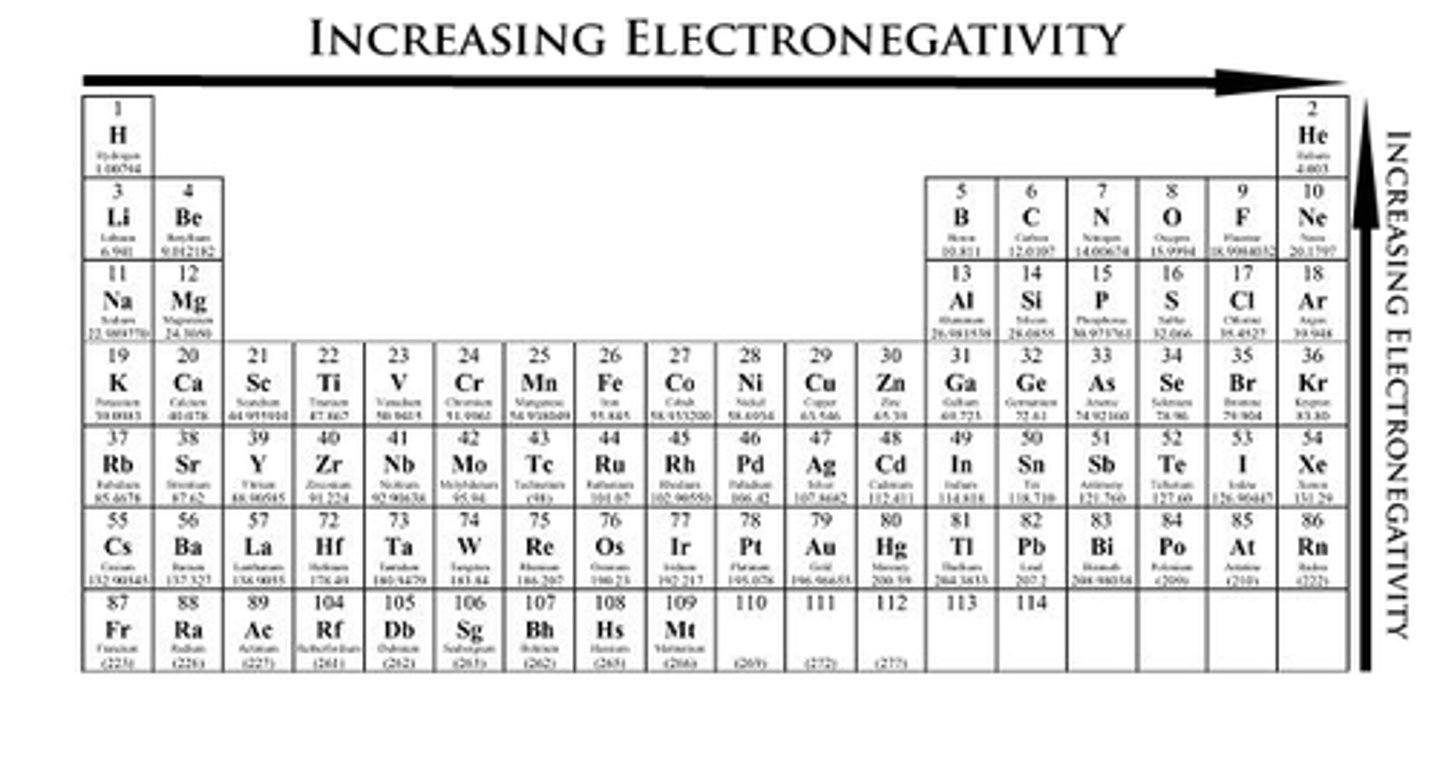
electronegativity
increases from bottom right to upper left
1. down a group, more rings means less pull
2. across a period, more protons means more pull
metals lose electrons in presence of non-metals
F > O > N > Cl > Br > I > S > C = H
acidity
increases from upper left to lower right (different!)
1. down a group, size explains: larger atom means conjugate base (cation) is more stable
2. across a period, electronegativity explains: more protons means conjugate base (cation) is more stable
Lewis structures and formal charge
1. place least electronegative atom is center, always C
2. start with all single bonds, deduct 2 electrons for each
3. start with most electronegative atoms and make octets
4. any remaining electrons add to central atom
5. form bonds to fill any atoms without octets
6. assign formal charge = valence electrons - sticks - dots
positive formal charge should be on less electronegative atoms
try to reduce set of formal charges
check that formal charges add up to overall charge
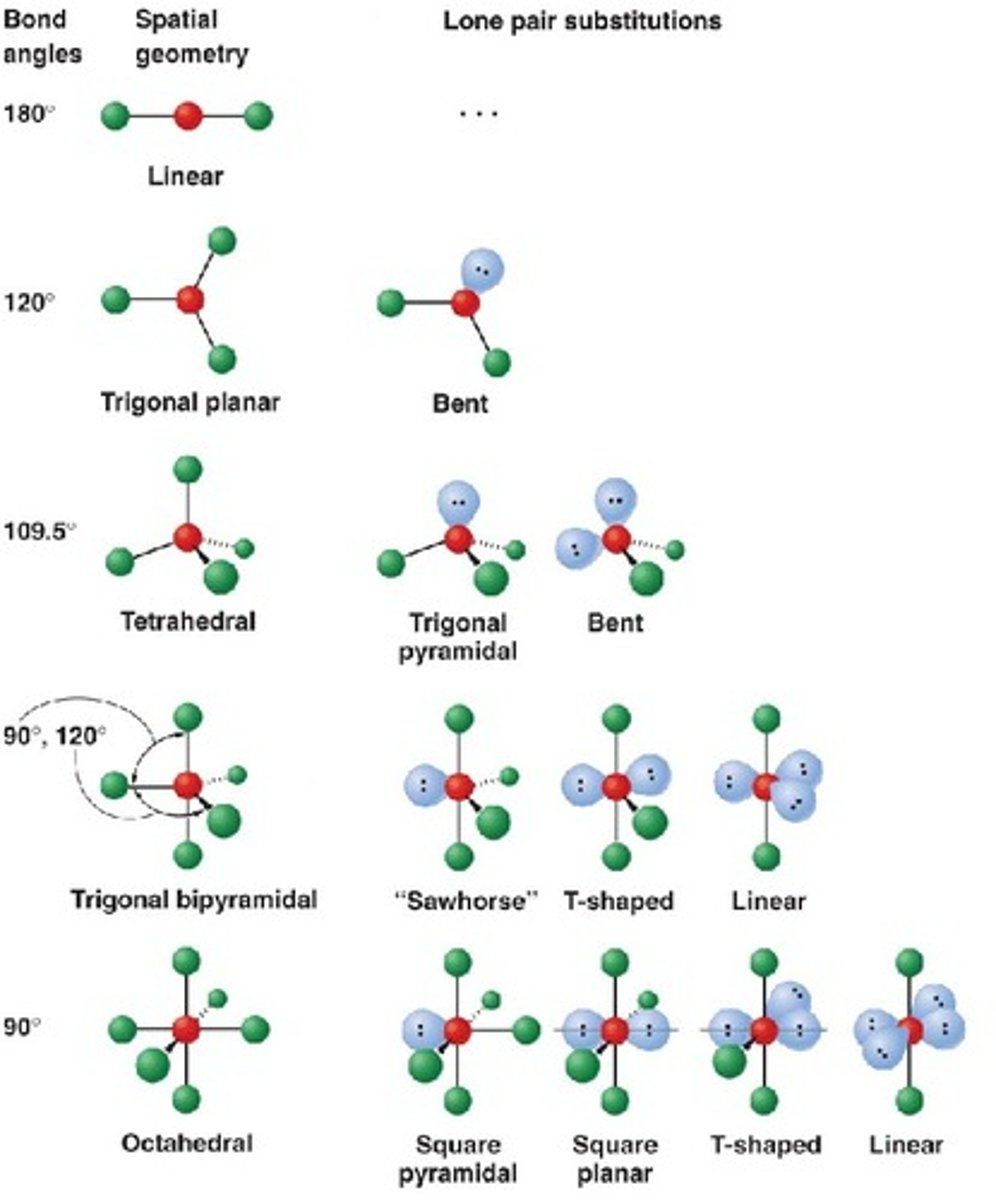
hybridization
sp for 2 groups, sp2 for 3 groups, sp3 for 4 groups
lone pairs determine molecular geometry: NH3 is trigonal pyramidal (sp3), H2O is bent (sp3), XeF4 is square planar (sp3d2), SF6 is octahedral (sp3d2)
octahedral is 6 bonds, 90 degrees (sp3d2)
sp3 to sp2 conversion to attain planarity and aromaticity (4n+2 electrons resonating in a ring)
amide N can convert from sp3 to sp2 to get resonance stabilized by carbonyl O
orbitals with more s character are more stable
bond length
depends on atomic radius (which increases to lower left)
depends on bond order, more electrons shared is a stronger bond and closer bond
breaking bonds
breaking a bond is endothermic (positive dH) but also exergonic! (negative dG)
ATP ->ADP + H2O requires heat but produces net energy
stronger bonds:
1. have more electrons shared
2. shorter distance between atoms
3. have higher dissociation energy
ATP phosphates
alpha- phosphate closest to sugar
beta- middle one
gamma- phosphate on tip
covalent bonds
metal and nonmetal share electrons
electronegativity differences creates dipoles at each bond
molecular dipole found by adding up all the bond dipoles
metallic bonds
sea of electrons delocalized between metal ions
usually S and D block elements
these compounds are conductors and malleable
coordinate covalent bond
formed between atoms with lone pairs and atoms that are electron deficient
usually between transition metals and organic compounds, where compounds donate electron pairs to coordinate with the metal
coordinate number- number of compounds coordinating with metal ions
Fe2+ can form 6 coordinate covalent bonds (that completely fill up its valence shells)
Fe2+ forms coordinate covalent bonds with hemoglobin
ionic bonds
formed between cations and anions
ions dissociate in aqueous solution, become conductor
as solids, these compounds are insulators and brittle
insulator/conductor
insulator- valence electrons tightly bound to atom
conductor- delocalized electrons, metallic bonds
intermolecular forces
pulling apart atoms is always endothermic!
4 types of IMFs:
1. ion-dipole forces- ions and polar molecule
2. dipole-dipole forces- two polar molecules, align along the molecular dipoles, H-bonding is special case
3. dipole-induced dipole forces- polar, nonpolar molecules
4. london dispersion forces- Van der Waals, temporary
5. Hydrogen bonds- align along the bond dipoles, require a donor and an acceptor, only N, O, and F can do hydrogen bonds
solvation shell
A cagelike network of solvent molecules that forms around a solute in a solution
decrease in entropy
active site
acidic and basic amino acids can undergo H-bonding and ionic interactions with the substrate
ATP has negatively charged phosphates that interact well with H, R, and K
entropy
disorder, always increasing in universe
what increases entropy (S):
1. increasing number of particles
2. increasing volume
3. increasing temperature
formation of a more organized compound or state would decrease entropy
enthalpy
breaking bonds is endothermic (dH > 0)
forming bonds is exothermic (dH < 0)
heat of formation (dHf) of elements in standard state is 0
heat of reaction (dH) = Hf products - Hf reactants
multiply by number of moles
positive dH means endothermic, heat is reactant
negative dH means exothermic, heat is product
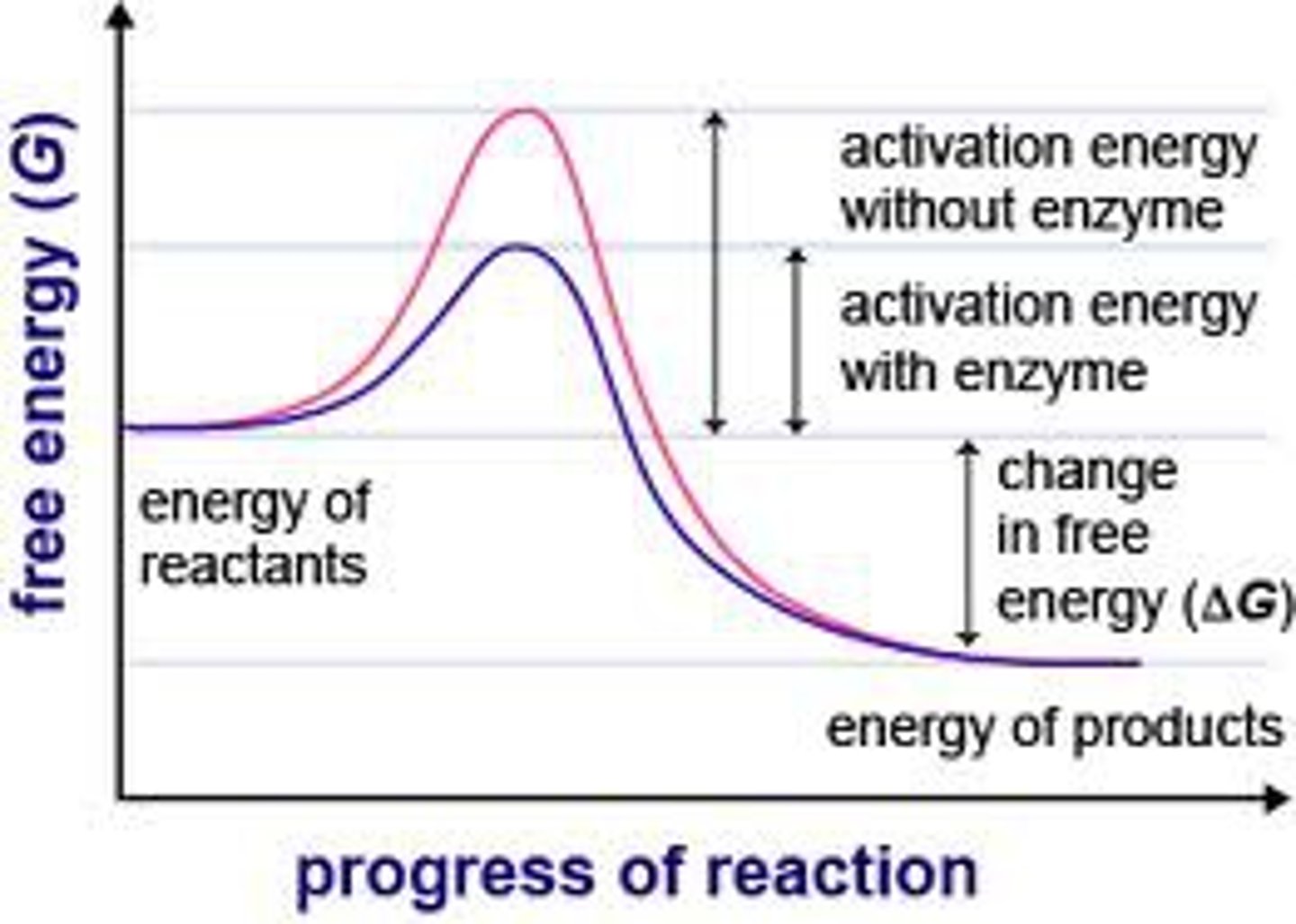
Gibbs free energy and spontaneous reactions
dG = dH - TdS
dGo = - RTlnK
dG = dGo + RTlnQ
free energy of formation (dGf) of standard state elements is 0
spontaneous process is exergonic (dG < 0)
nonspontaneous process is endergonic (dG > 0)
examples:
combustion (-dH, +dS) is spontaneous at all temperatures
freezing (-dH, -dS) is spontaneous at low temperatures
ATP -> ADP (+dH, +dS) is spontaneous at high temperatures
basic gas laws
Avogadro's law- n proportional to V, so 1 mol is 22.4 L at STP
Boyle's law- P inverse to V, PV = PV
Charles' law- T proportional to V, V/T = V/T
Gay-Lussac's law- T proportional to P, P/T = P/T
Dalton's law- partial pressure proportional to mole fraction, Ptotal = P1 + P2
PdV curve
x-axis is volume
y-axis is pressure
work = PdV = area under the curve
filling up more volume under constant pressure does more work
units of pressure
1 atm = 100 kPa = 760 torr = 760 mmHg
ideal gas law
PV = nRT
R = .08 Latm/Kmol
but you don't need this, just use 22.4 L/mol at 0 C and 1 atm
real gas law and non-ideality
P_theoretical > P_obs, since particles have IMFs with each other
V_theoretical < V_obs, since particles have size
(P_obs + an^2/V^2)(V_obs - nb) = nRT
1. a increases as IMF increase
2. b increases as particle size increases
non-ideal conditions are high pressures and low temperatures, which maximize IMFs:
1. V_obs is greater than predicted
2. P_obs is smaller than predicted
Faraday's constant
charge per 1 mol of electrons
F = 100,000 C/mol
Faraday's Law and electroplating
over time, electrons can be used to do work like electroplating
I = Q/t
Q = nF
Faraday's constant is charge per mole
calculate moles of metal plated given current and time:
1. determine charge: Q = It
2. determine moles of electrons: ne = Q/F
3. determine moles of metal: nmetal = ne/ionic charge (electrons per metal)
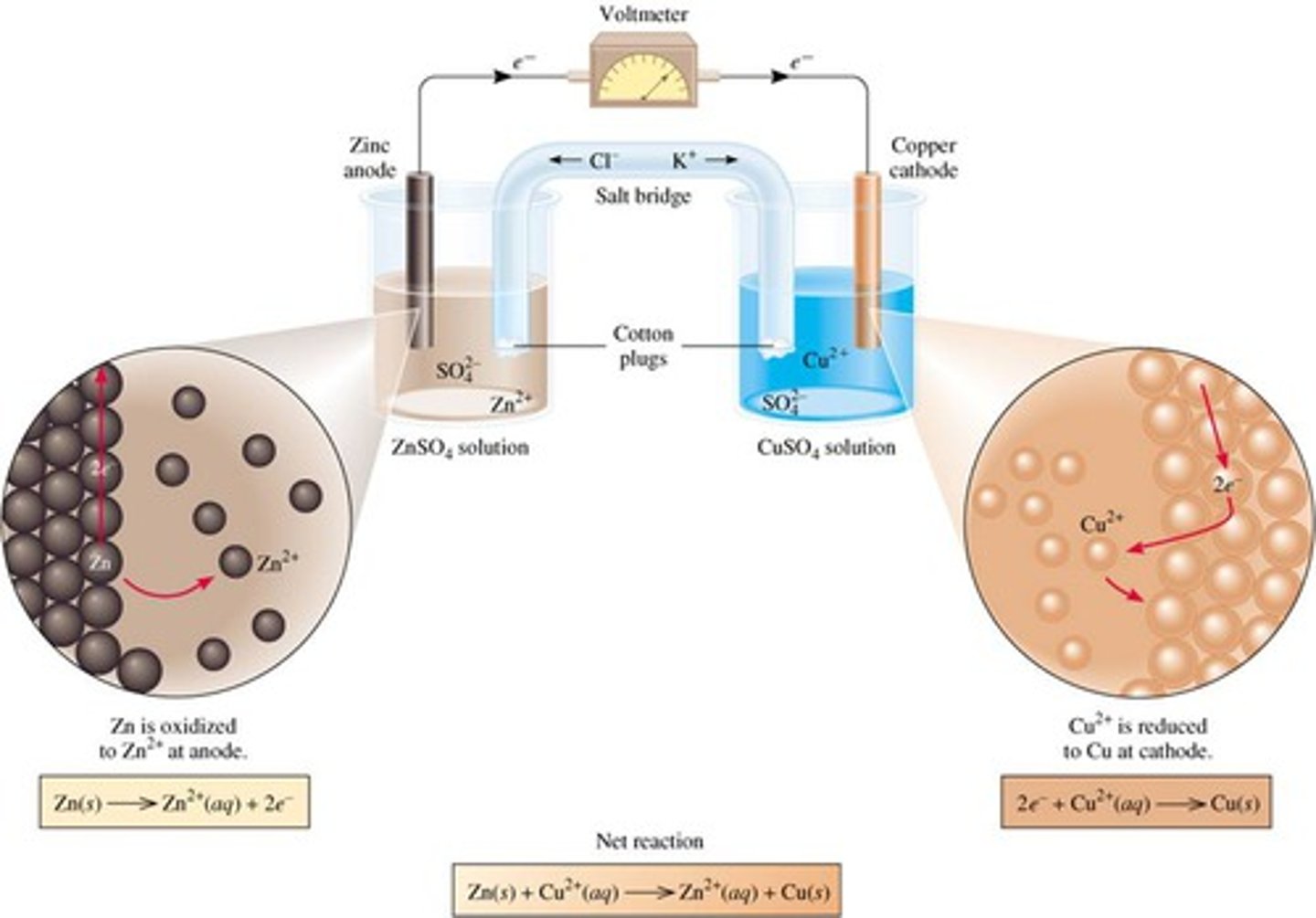
galvanic cell
cell must have two electrodes, an electrolyte bridge, and a wire with resistance (galvanic) or power source (electrolytic)
spontaneous, positive Ecell
oxidation at the anode (an ox), reduction at the cathode (red cat)
electron travel from anode (-) to cathode (+) through wire, discharging battery
cathode is plated, anode loses metal ions
Nerst equation
cell in equilibrium has an actual cell potential of 0 and a standard cell potential of not 0
E = E0 - (RT/nF)lnQ
E0 = (RT/nF)lnK
-as temp increases, Ecell decreases
-as battery approaches equilibrium, Ecell approaches 0
-E0 is constant
salt bridge
anion will always migrate towards anode, cation will always migrate toward cathode
in galvanic cell, salt bridge balances out movement of electrons from anode to cathode
in electrolytic cell, anion just is attracted to positively-charged anode
without a salt bridge, current immediately stops
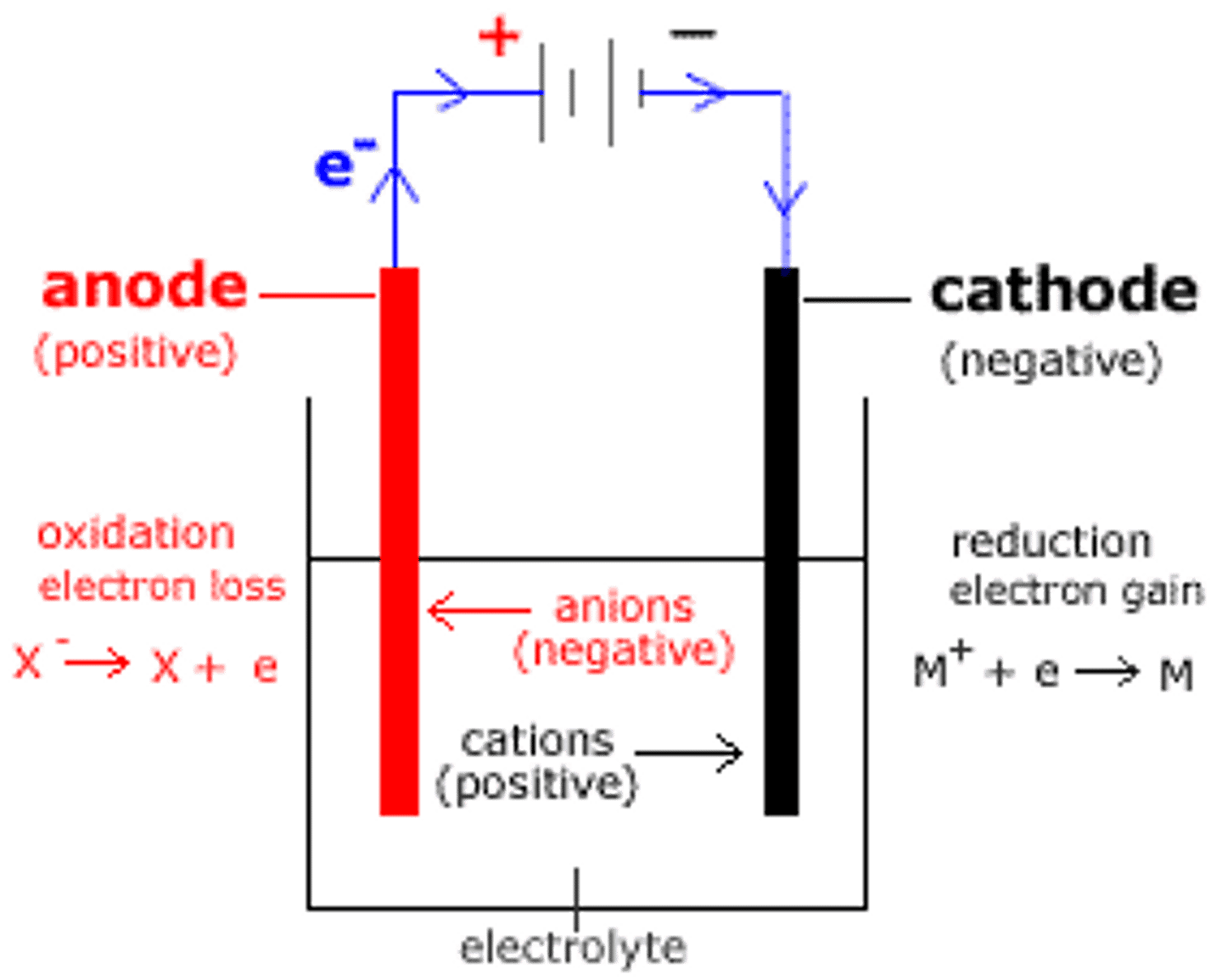
electrolytic cell
opposite of galvanic cell
recharging battery using external energy source reverses flow of electrons
electrons still going from anode to cathode, but are being forced to cathode is negative and anode is positive
increasing current will increase rate of electroplating
drives nonspontaneous reaction, negative Ecell
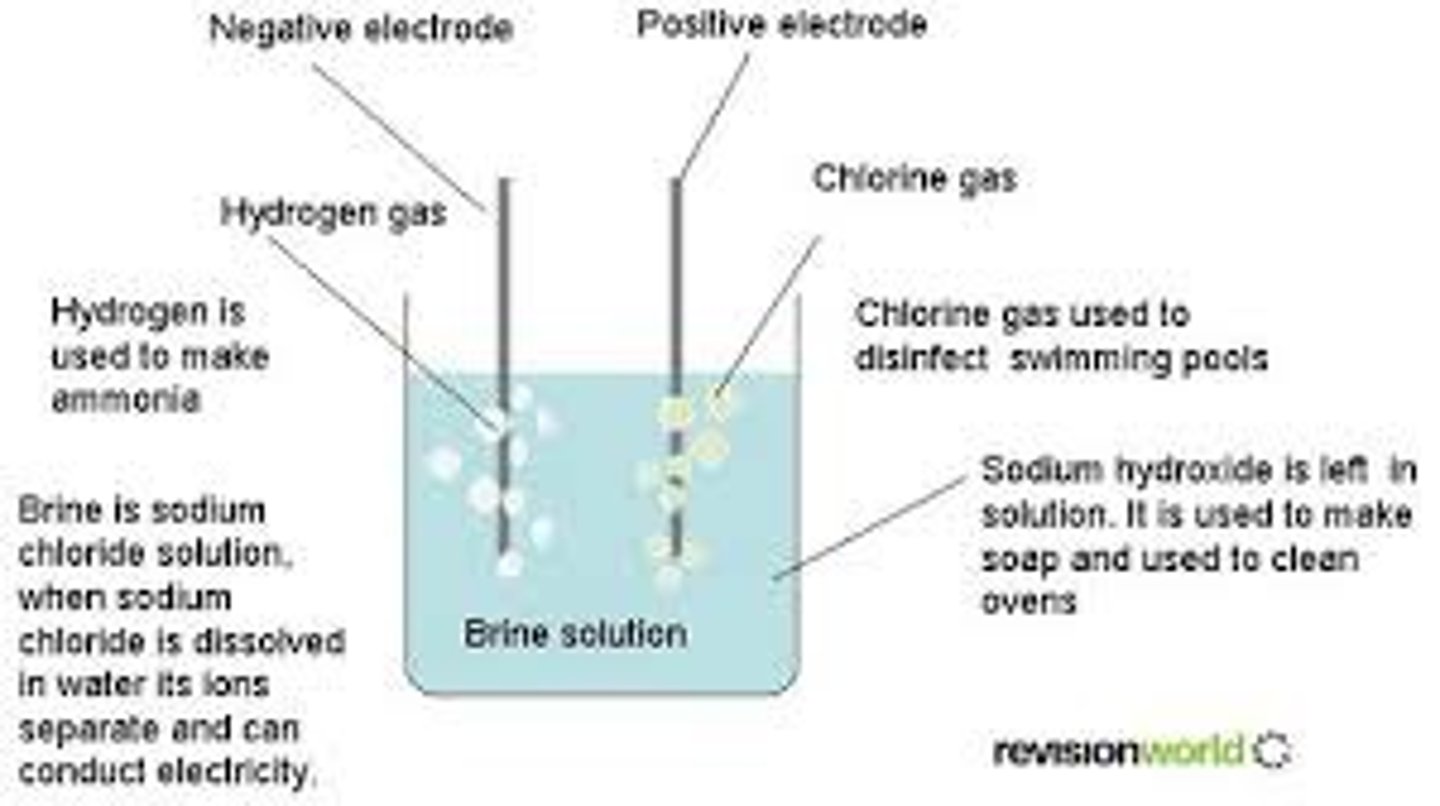
electrolysis
in electrolytic cell with water, hydrolysis will occur creating H2 at the cathode and O2 at the anode
in electrolytic cell with NaCl, hydrolysis will occur creating H2 at cathode and Cl2 at anode
redox titration
cerimetry- Ce4+ + e- => Ce3+
adding Ce4+, observing increase in pH on y-axis
reaction rate
factors that increase it:
1. increased temperature
2. decreased activation energy
3. not affected by concentration of reactants!
rate law
an equation that relates the reaction rate to the concentration of reactants, typically in the form: rate = k[A]^m[B]^n.
solubility equilibrium
the state in which the rate of dissolution of a solute is equal to the rate of crystallization, resulting in a constant concentration of the solute in solution.