Rev test 2 Metals, Redox, ELECTROLYSIS
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
Draw a labelled diagram to show the bonding and structure in a sample of aluminium metal. (3)
M1: (for a) 3x3 grid or bigger (of ions)
M2: ion(s) AND electron(s) labelled
M3: (drawing shows) Al3+ ion in fixed lattice position AND delocalised electron(s)
IGNORE: if no. of electrons = 27
IGNORE: (unless) less than 15 (electrons)
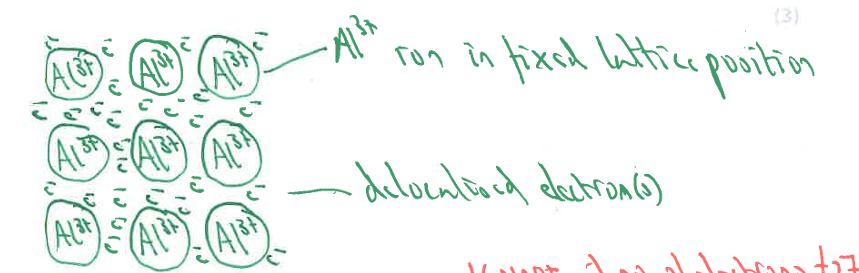
Explain why aluminium is a good conductor of electricity. (2)
M1: delocalised electrons present (since metallic bonding)
M2: these electrons are Mobile / free to move
Suggest why aluminium is ductile. (2)
M1: layers of atoms / ions
M2: can slide past each other
Explain what is meant by the term electrolysis. (2)
M1: passage of an electric current through an electrolyte
M2: accompanied by redox reactions at the electrodes / OR (resulting in) decomposition of the electrolyte
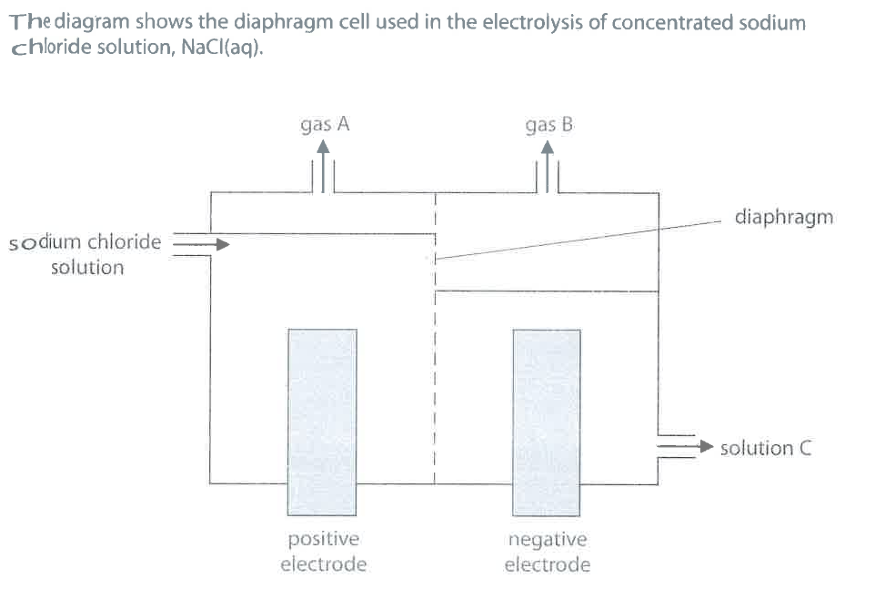
Identify gas A, gas B and solution C. (3)
M1 (gas A): chlorine / Cl2
M2 (gas B):
H2/ hydrogenM3 (solution C): sodium hydroxide / $NaOH$
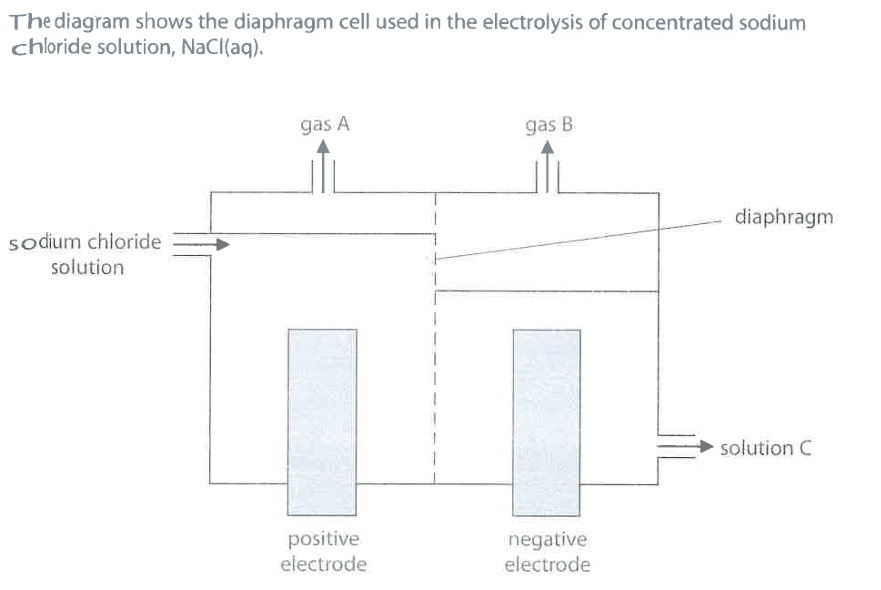
Why does the sodium chloride have to be molten before it will conduct electricity? (1)
M1: so that the IONS are free to move / Mobile
The ionic half-equation for the formation of sodium is $Na^+ + e^- \rightarrow Na$ Write the ionic half-equation for the formation of chlorine from chloride ions. (2)
M1: 2Cl− on left and Cl2 on right
M2: +2e− on right (or
−2e−on left)Full equation: 2Cl−→Cl2+2e−
State two properties that make iron a suitable metal for saucepans. (2 marks)
M1 malleable
M2 good conductor of heat
M3 high melting point
State two properties that make aluminium a suitable metal for power cables. (2 marks)
M1 ductile
M2 good conductor of electricity
What is meant by the term exothermic? (1)
M1: heat is given out to the surroundings
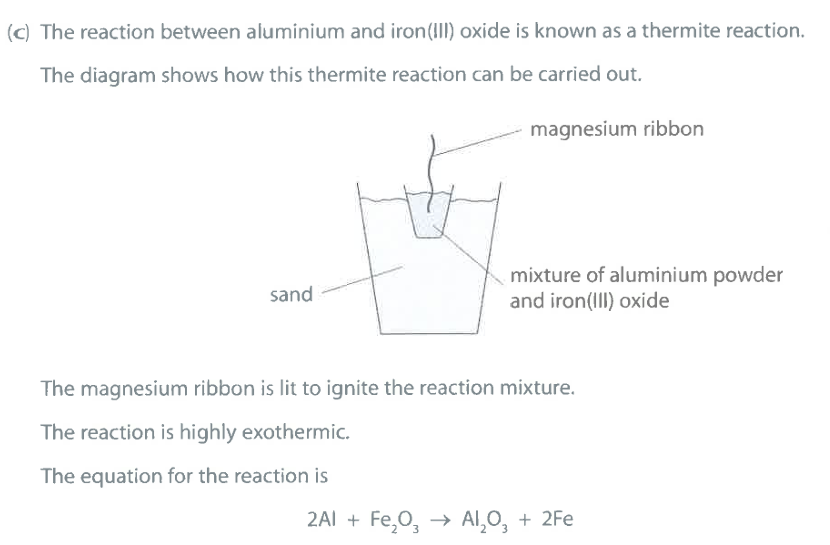
What does the reaction suggest about the reactivity of aluminium compared to the reactivity of iron? Explain your answer. (2)
M1: Al is more reactive than Fe
M2: Al displaces Fe / OR Al removes oxygen from $Fe_2O_3$
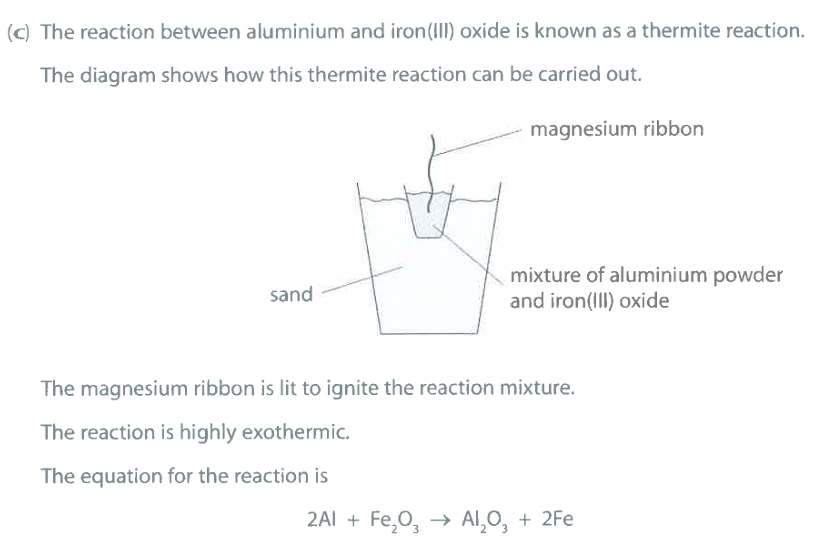
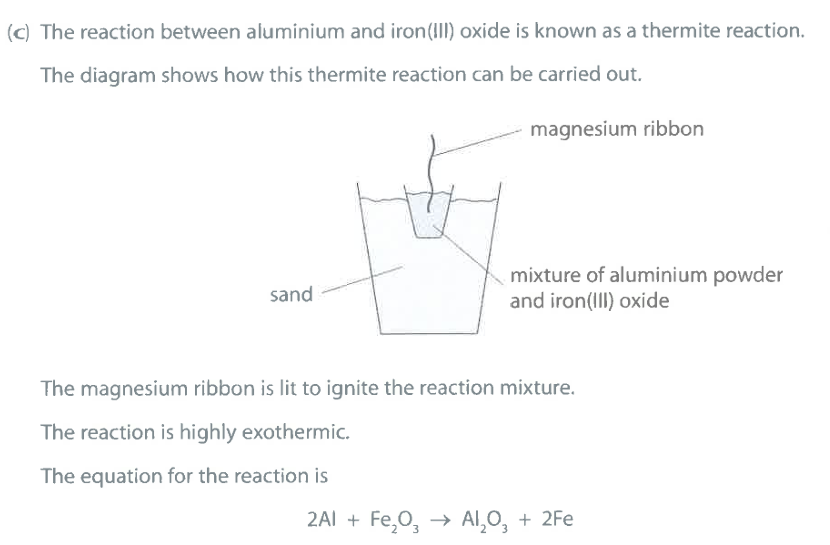
Which element is oxidised in this thermite reaction? Give a reason for your answer. (2)
M1: Al is oxidised
M2: Al is gaining oxygen
Ignore: loss of electrons
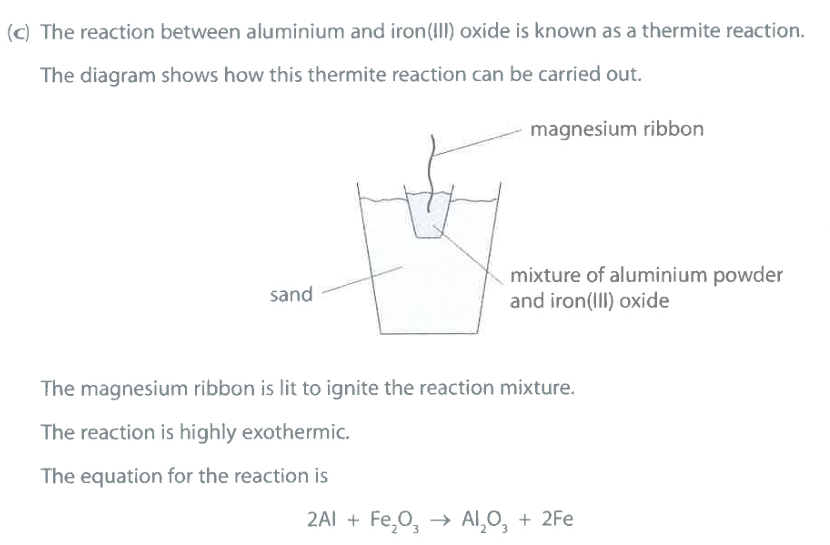
Give the chemical name for rust. (1)
M1: hydrated iron(III) oxide
Suggest a suitable element for the inert electrodes. (1)
M1: carbon or platinum
REJECT: "graphite"
3 properties of metals
Any one of the following:
Good conductors of heat; [1 mark]
Good conductors of electricity; [1 mark]
Malleable; [1 mark]
Ductile; [1 mark]
High density; [1 mark]
Have basic oxides / hydroxides; [1 mark]
High melting point; [1 mark]
Sonorous; [1 mark]
Shiny; [1 mark]
Hard; [1 mark]
Strong; [1 mark]
[Total: 3 marks]
The most common correct answer to this question is about metals being good conductors of electricity
Careful: Good conductor on its own may not be enough to achieve the mark
Explain why mercury is a liquid and other metals are solids. (2)
In mercury, the particles can move / flow; [1 mark] Comments about the spacing and energy of the particles are ignored
In solid metals, the particles do not move
OR
In solid metals, the particles vibrate around / are in fixed positions; [1 mark]
It is asking you to compare the movement of particles in a liquid and a solid