H3 Toth- Cytoskeleton I - microtubules
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
describe the 3 kinds of cytoskeleton
actin (micro) filaments- all eukaryotes, polar, use ATP, dynamic, 7nm
tubulin microtubules- all eukaryotes, polar, use GTP, dynamic, 25nm
intermediate filaments (multiple kinds)- only animals, apolar, don’t use ATP/GTP, less dynamic, 10nm
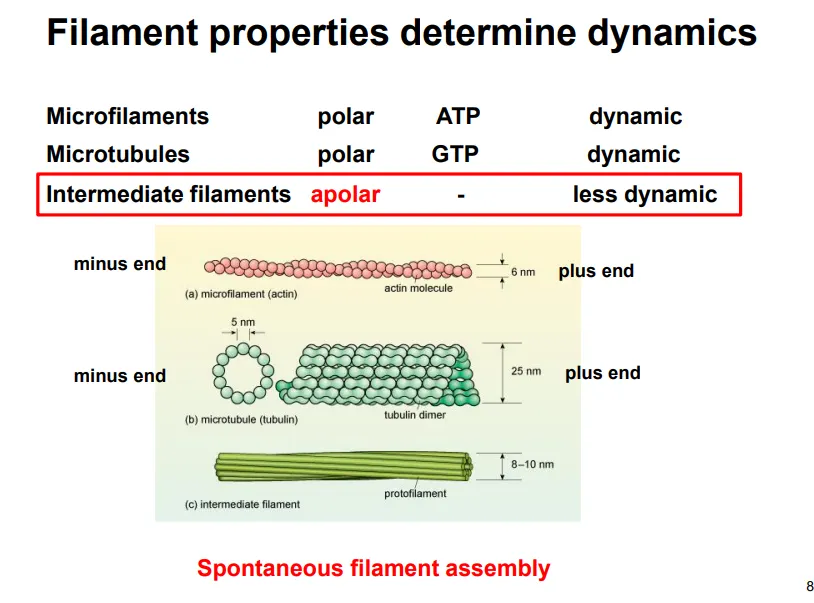
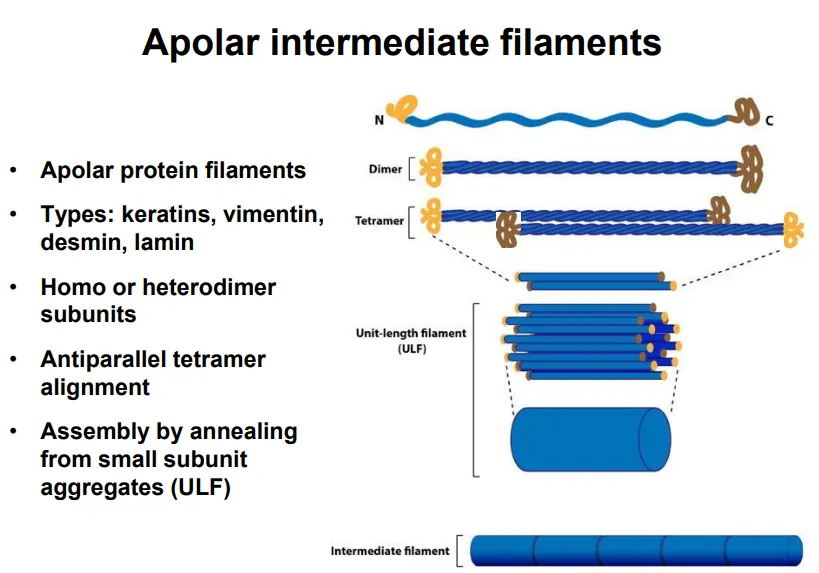
describe intermediate filament structure and function (give examples)
apolar (the two ends have the same properties)
long strands with globular C and N termini assemble into coiled-coil dimers
dimers assemble antiparallel into tetramers
these line up into smaller subunits, which anneal into long filaments
eg. keratin, vimentin, desmin, lamin
mainly used for structural support, determining and maintaining the cell and nucleus shape (so they aren’t needed in plants)
this is because they have a high tensile strength and are resistant to compression and bending
eg. lamins form a lattice structure which covers the inner side of the nuclear envelope as a site for chromatin anchorage
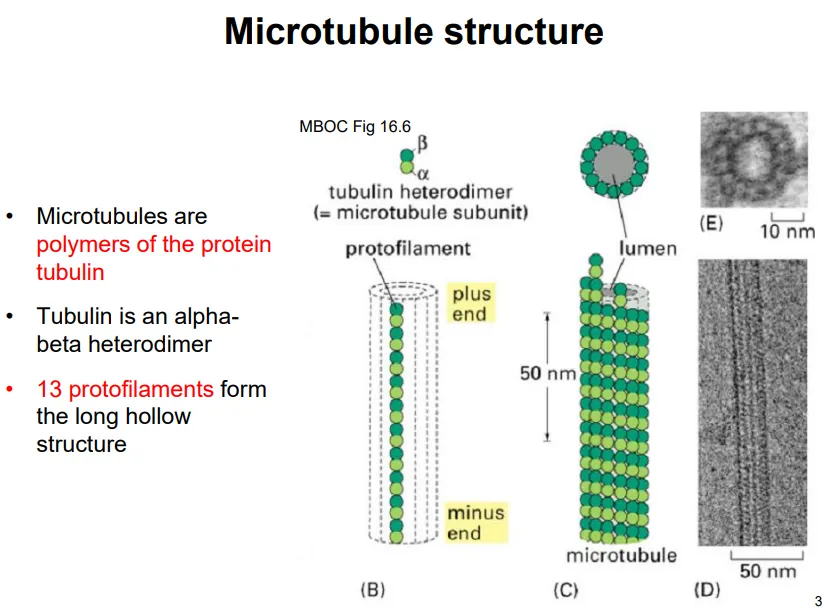
describe the structure of microtubules
tubulin monomers are heterodimers of two subunits: alpha and beta
the heterodimers assemble end to end, so that the alpha and beta subunits alternate, into long strands called protofilaments
13 of these protofilaments assemble into a hollow cylinder
the plus end is the beta end, and the minus end is the alpha end
each of the subunits has a GTP-binding site, but only beta-tubulin can hydrolyse it- the GTP bound to alpha-tubulin lends stability and proper folding
decribe microtubule formation, polymerisation and depolymerisation
self-assembly from scratch is difficult because 13 subunits need to interact laterally, which is energetically unfavourable
in cells gamma-tubulin ring complexes are used in initiation to act as structural templates for the alpha-tubulin of the heterodimers to bind (at the minus end)
these complexes are anchored at particular organelles
tubulin monomers, bound to GTP, are added to the plus end
the GTP provides strong lateral interactions for stability
once a monomer associates, it triggers GTP hydrolysis by the beta-tubulin subunit
this is very slow, so new subunits can incorporate before GTP hydrolysis is achieved, creating a GTP cap at the plus end
the GDP-bound heterodimer has a different conformation, which can induce dissociation if the GTP cap isn’t maintained (when the hydrolysis rate > polymerisation rate), resulting in shrinkage, and vice versa
this is dynamic instability, which can result in complete depolymerisation if there are no GTP-bound dimers to rescue the growth

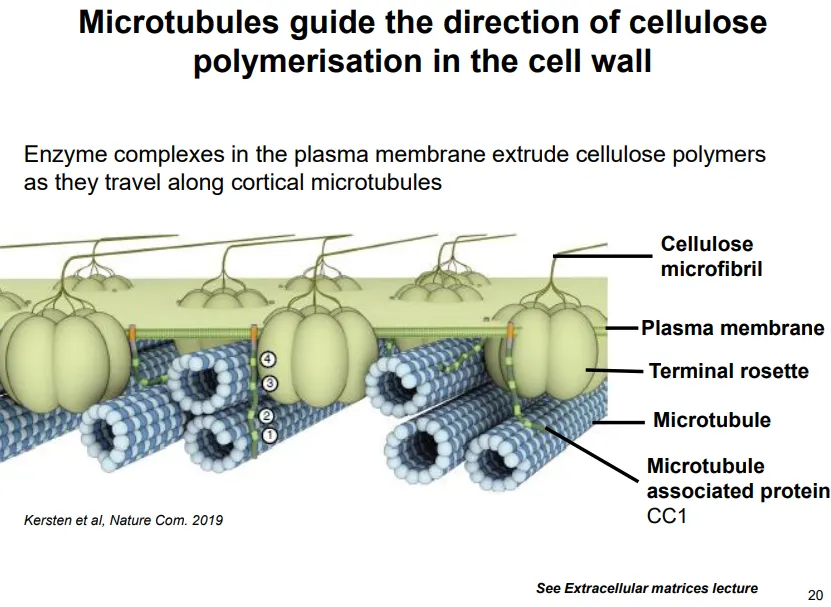
what is the function and organisation of microtubules in plant cells?
in young cells microtubules radiate out from the nucleus
in larger growing cells (interphase) they associate and run parallel with the membrane
these guide cellulose deposition in the correct orientation
enzyme complexes in the plasma membrane extrude cellulose polymers as they travel along parallel membrane anchored microtubules
what is the function and organisation of microtubules in animal cells?
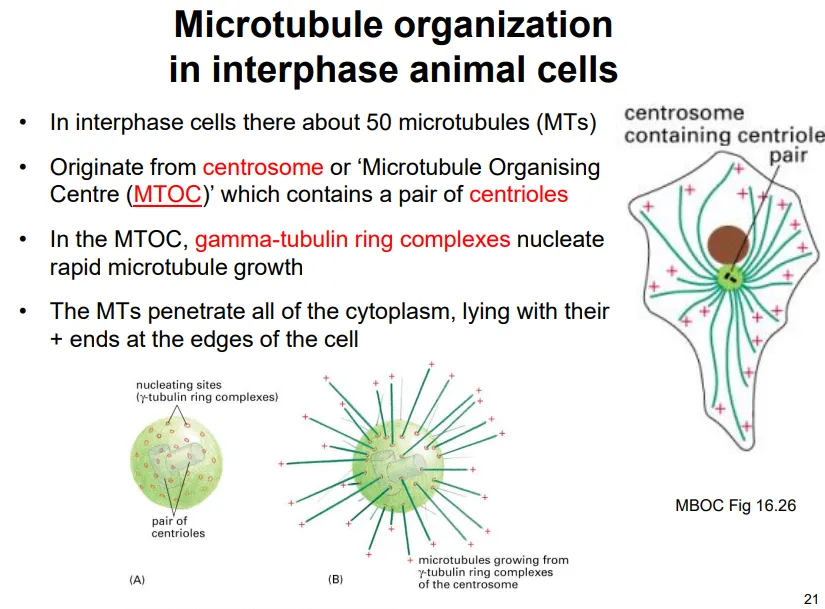
microtubules radiate out from the centrosome (the microtubule organising centre, MTOC), which contains a pair of centrioles- the centrosome has many gamma-tubulin ring complexes for initiation
most microtubules constantly polymerise and depolymerise out from the centrosome due to dynamic instability
this is because hitting the membrane perpendicularly causes the loss of the GTP cap and results in depolymerisation
we believe this functions to help the cell to explore and sense changes in the plasma membrane
microtubules also act as tracks for motor proteins to move organelles and transport vesicles around the cell eg. the endomembrane system
the spindle fibres used in mitosis are microtubules
(eukaryotic) cilia contain microtubules in a 9+2 formation for movement eg. sperm, and signalling/sensing eg. rod cells
how is microtubule growth regulated?
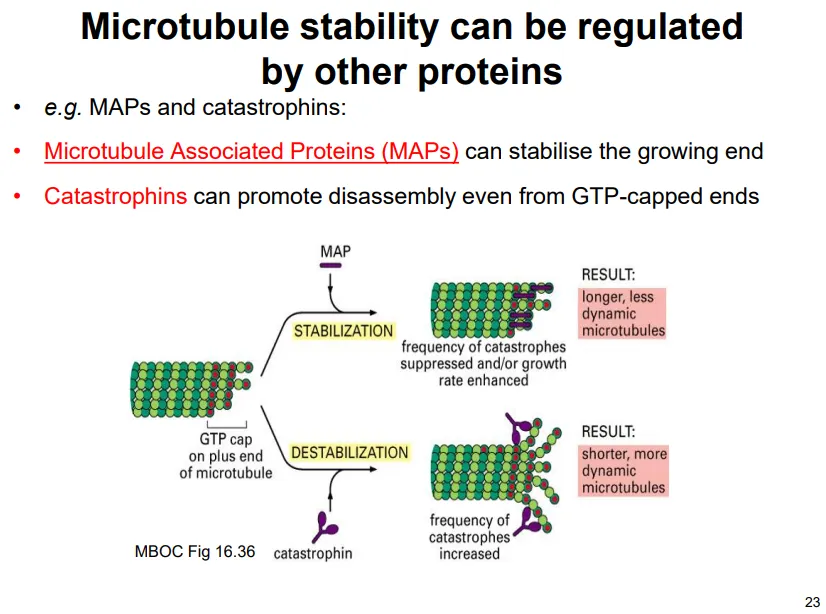
most of the time dynamic instability isn’t observed because of protein action:
microtubule associated proteins (MAPs) stabilise the plus end to promote growth
catastrophins destabilise the plus end to promote depolymerisation
how are organelles moved around the cell by microtubules?
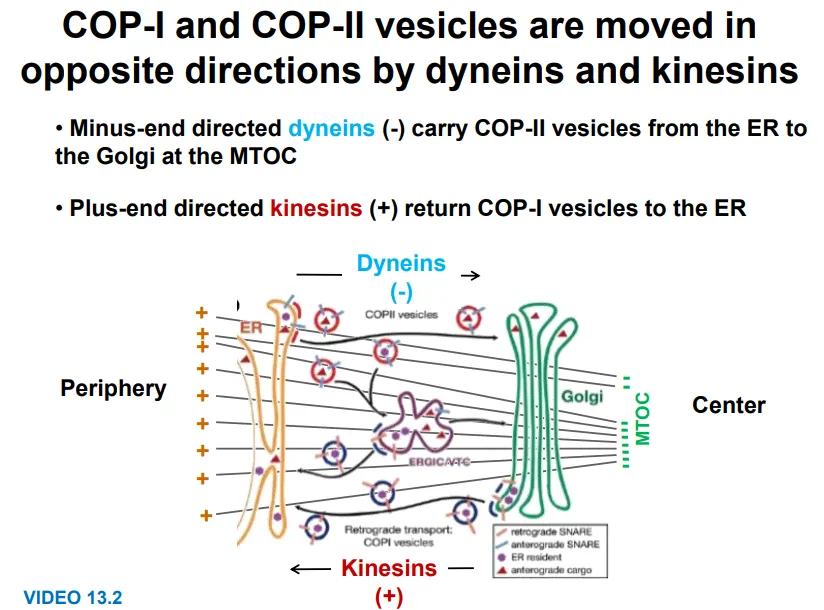
motor proteins bind to vesicles and organelles and move along the microtubules
kinesins move towards the plus end (outwards)- eg. for COP-I vesicles golgi → ER
these are dimers with a cargo-binding tail domain, a stalk and a two head domain, which moves along microtubules in 8nm steps, each using one ATP (binding and hydrolysis cause conformational changes for moving the heads forwards)
dyneins move towards the minus end (inwards)- eg. for COP-II vesicles ER → golgi
these are more complicated, sometimes involving accessory proteins for binding, but using ATP to make irregular movements
how are microtubules organised in eukaryotic cilia and how do they bend?
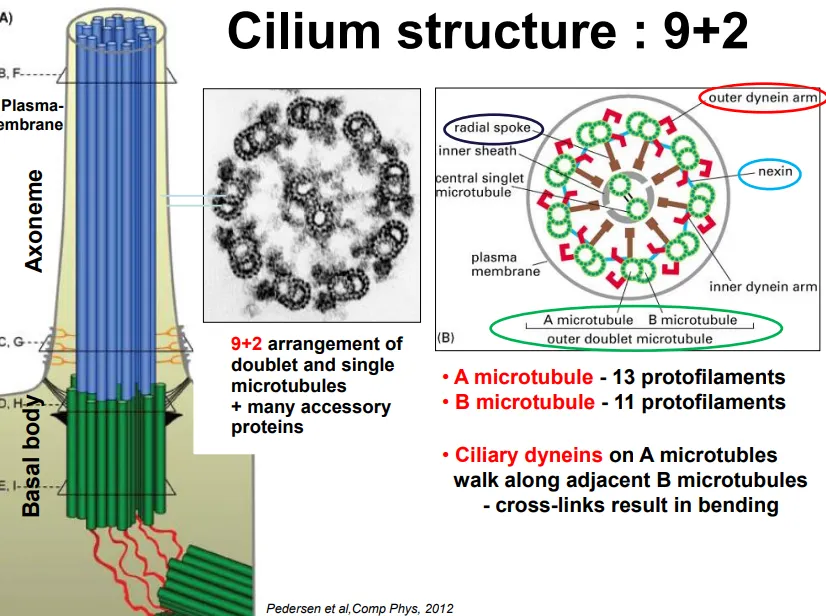
the basal body (originating from centrioles, but not in plants) extrudes microtubules in a 9+2 arrangement
9 doublets (13 protofilament cylindrical A microtubule + 11 protofilament attached B microtubule) are arranged in a ring, with 2 singlets in the centre
the doublets are connected by nexin proteins and have radial spokes towards the centre
the microtubules are always enclosed by the plasma membrane
ciliary dynein (a motor protein complex) moves along the doublet protofilaments due to ATP
this would cause the filaments to slide, but due to the nexin linking proteins and controlled activity, this causes bending
in short cilia this produces a force perpendicular to the long axis, but in long cilia this symmetric beat produces a parallel force (in a sinusoidal wave)
compare bacterial and eukaryotic cilia proteins, positions and movement
