Ch. 23 - Chromatography
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Chromatography is a _______.
Separation technique that operates on the same principle as liquid-liquid extraction.
❑Separation is performed in a column instead of a separatory funnel
❑One phase is held in place (stationary phase) while the other moves
(mobile phase) past of it.
Chromatography is based on the _______.
affinity of all analytes within the stationary phase vs the mobile phase
Important terminology
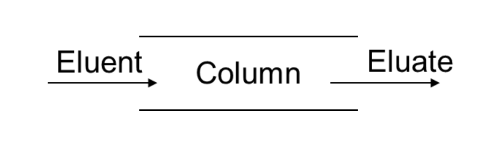
❑Eluent: fluid entering the column.
❑Eluate: fluid emerging from the end of the column.
❑Elution: the process of passing liquid or gas through a
chromatography column.
Mobile Phase
fluid moving through column
Liquid (methanol, water, etc.)→ Liquid Chromatography (LC)
Gas (He, H2, etc.)→Gas Chromatography (GC)
Stationary Phase
fixed inside the column
option 1: A viscous (gel-like) liquid chemically bonded to the inside of a capillary tube or onto the surface of solid particles in a column.
option 2: Solid particles packed into a column
Chromatographic separations
The differences in the extent to which solutes are partitioned between the mobile phase and the stationary phase
Cs: concentration of solute in stationary phase
CM: concentration of solute in mobile phase
This formula is not commonly used
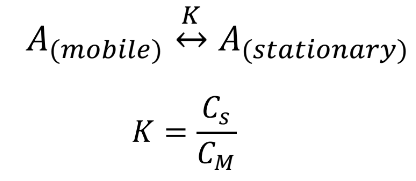
Retention time
More commonly used than the partition Coefficient (K)
Tr: retention time
length of time solute spends in column (from injection to when peak shows up)
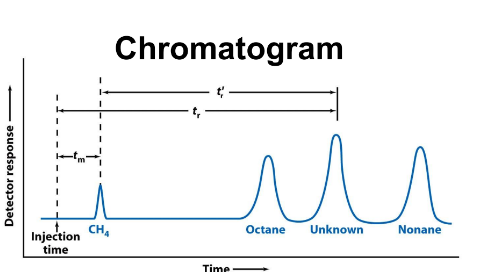
tm
Minimal possible retention Time →Time needed for mobile phase to travel through column
All solutes spend equal time in mobile phase
tr’
adjusted retention Time →Time solutes spend in stationary
phase
tr’ = tr - tm
Relative Retention/Separation Factor, a
❑Compares adjusted retention times (tr’) of two solutes
❑Measure of how well the column will separate two solutes
❑Fairly independent of flow rate.

The larger a, the better the _______.
separation between two components (peaks are farther away from each other)
Retention Factor (K)
❑The longer a component is retained by the column (stationary phase),
the greater is the retention factor.
❑Higher k indicates solute retained longer.
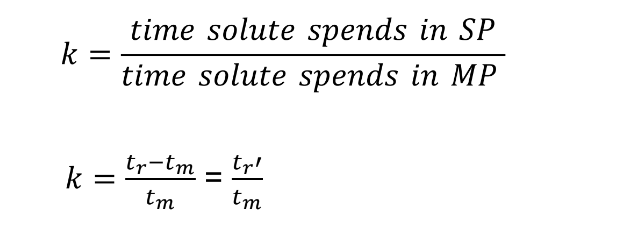
How can we determined how well compounds are separated by chromatography?
Difference in elution times: The farther apart the peaks are the better their separation
Width of peaks: The sharper the peaks the better separation
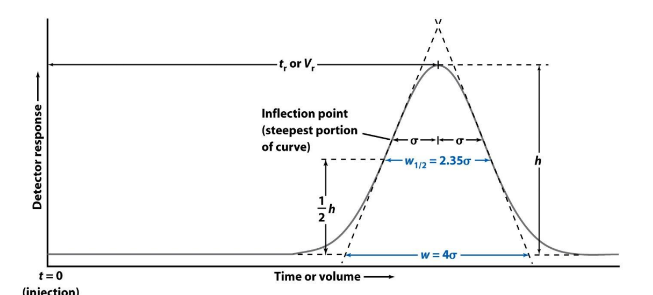
Common measures of peak width
❑w1/2 : width measured at a height equal to half of the peak height
❑w: width at the baseline between tangents drawn to the steepest
parts of the peak.
❑Solute peaks→Gaussian peak shapes → use population st.dev to describe peak
width.
• W1/2 = 2.35 sigma
• W = 4 sigma
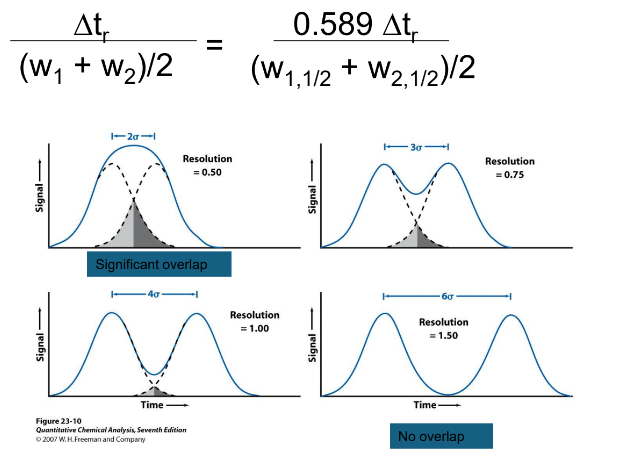
Resolution
The ability of a column to distinguish two solutes from each other
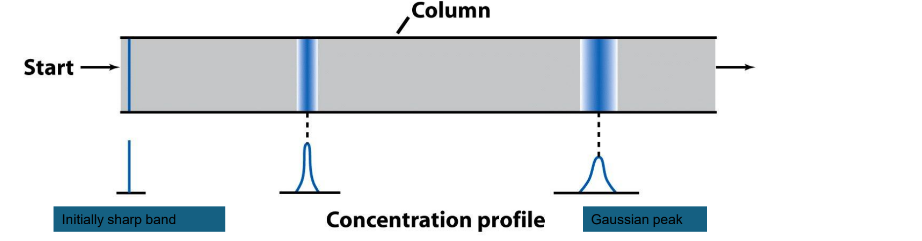
Broadening
A band of solute broadens as it moves randomly through a
chromatography column from a region of high solute concentration
to a region of low solute concentration.
Factors causing band spreading or broadening
A. Multiple flow paths
B. Longitudinal Diffusion
C. Equilibration time
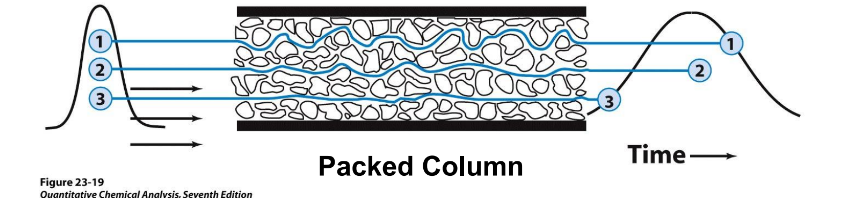
Multiple Flow paths (A)
Molecules choose varying path lengths through a packed column →
varying retention times → wider peaks → band broadening
open tubular column = no packed materials inside →molecules follow same path
Factor causing band spreading or broadening
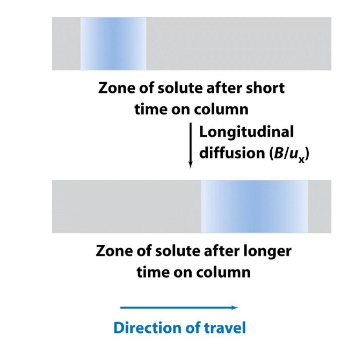
Longitudinal Diffusion (B)
Diffusion along the column axis.
diffusion meaning: molecules move from areas in the band of molecules that are high in concentration to areas that are low in concentration
Factor causing band spreading or broadening
Equilibration Time (C)
❑It takes a finite moment for a solute to enter and to leave the SP.
❑Band in MP will be slightly ahead of SP → Solutes in SP are left behind by the solutes traveling along in MP → a cycle of equilibrium takes time when the MP is slightly ahead
❑Plate height from finite equilibration time is also called “Mass Transfer Term”
Factor causing band spreading or broadening
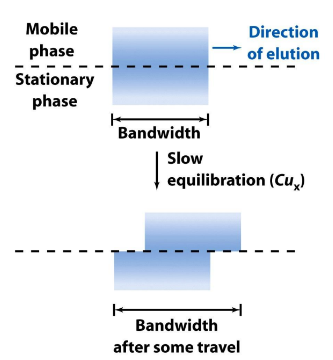
Plate Height (H) describes the relationship between the _______.
width of a solute band to the distance (x) it has traveled
The smaller H → the narrower the bandwidth (smaller st. dev) → better separations

Number of Theoretical Plates (N)
want a large # of Theoretical plates
N: Plate Count or Number of Theoretical Plates
L: length of the column (cm)
H: Plate Height

How to solve for N using baseline width and ½ height width
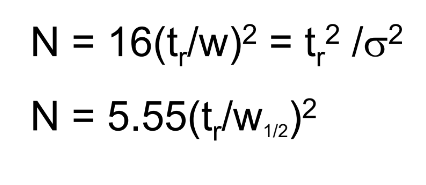
Plate Height (H)
Length of column required for one equilibration of solute between the MP and SP
How improve column efficiency
small volumes for liquid-liquid extraction→ small H
multiple extractions liquid-liquid extraction→Large N
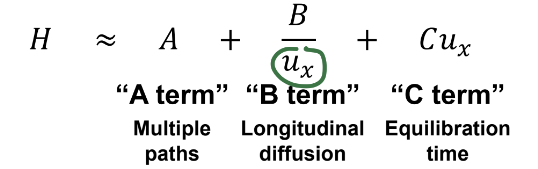
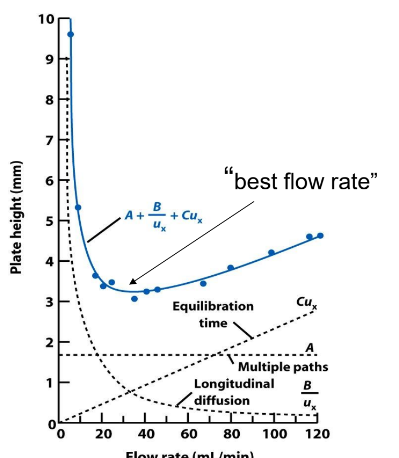
Van Deemter Equation for Plate Height
Describes how all terms have a different relationship with flow rate
A term: no dependence on flow rate.
B term: increase ux to minimize longitudinal diffusion.
C term: decrease ux to minimize band spreading due to
equilibration time
Ux = linear flow rate
Van Deemter specifics
❑Packed columns: terms A, B, C do not equal zero
❑For packed columns, band broadening can be minimized by small particle diameters (ex: 1.8 or 3.5 micrometers = small particles maximize separation efficiency)
❑Open tubular columns : A = zero
Overloading
Too much solute is applied to the column → Gradual increase then sharp decrease
Produces a gradual rise and an abrupt fall of the chromatographic peak.
As [S] increases → solute becomes more soluble in the SP. (too much Analyte → solubility of Analyte increases)
S is so soluble in the overloaded zone that little solute trails behind the peak.
Avoid this to make sure peak is good and to avoid contamination from left-over solute
![<ul><li><p>Too much solute is applied to the column → Gradual increase then sharp decrease</p></li></ul><ul><li><p>Produces a gradual rise and an abrupt fall of the chromatographic peak.</p></li></ul><ul><li><p>As [S] increases → solute becomes more soluble in the SP. (too much Analyte → solubility of Analyte increases)</p></li></ul><ul><li><p>S is so soluble in the overloaded zone that little solute trails behind the peak.</p></li></ul><ul><li><p>Avoid this to make sure peak is good and to avoid contamination from left-over solute</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/4019031e-0937-4dad-a270-8b7b8abf9be1.png)
Tailing
❑Arises when small quantities of solutes are retained more strongly than large quantities → increase then gradual decrease
❑Some sites retain solute more strongly than other sites.
❑Silica surfaces of columns and SP particles have hydroxyl
groups that form hydrogen bonds with polar solutes → leading
to tailing.