CHEM 2312 FINAL EXAM
1/203
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
204 Terms
What are the 7 things to check for when analyzing stability vs reactivity of a compound?
the period/row
charges
the group
hybridization
aromaticity
inductive effects
field effects
electronegativity increases from (left/right) to (left/right) and from (top/bottom) to (top/bottom)
left to right and from bottom to top
the more s-character the (less/more) electronegativity
more
state Aufbau’s principle
fill orbitals starting from lowest to highest
state Hund’s rule
for degenerate orbitals, fill of parallel spins w/ single occupancy before going up
main group elements are surrounded by how many valence electrons when forming covalent bonds?
8
which type of elements does having 8 valence electrons when forming covalent bonds work well for?
second period elements
equation for finding formal charge
# valence electrons - (# shared electrons + # LP)
how many bonds does carbon, nitrogen, boron, oxygen and halides prefer?
C → 4
B & N → 3
O → 2
Halides → 1
what is the formal charge on Boron bonded to 4 single bonds?
-1
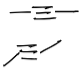
which one is proper alkyne geometry?
the top one
state the geometry associated with each of the following number of electron pairs:
2
3
4
5
6
2 → linear
3→ trigonal planar
4→ tetrahedral
5→ trigonal bipyramidal
6→ octahedral
state the geometry of the following geometries if you were to remove a lone pair from the subsequent geometry
trigonal planar
tetrahedral
trigonal bipyramidal
octahedral
trigonal planar: bent
tetrahedral: trigonal pyramidal → bent
trigonal bipyramidal: sawhorse → Tshape → linear
octahedral: square pyramidal → square planar → T shape → linear
state the degrees between atoms of a linear, trigonal planar and tetrahedral geometry
linear → 180
trigonal planar → 120
tetrahedral → 109.5
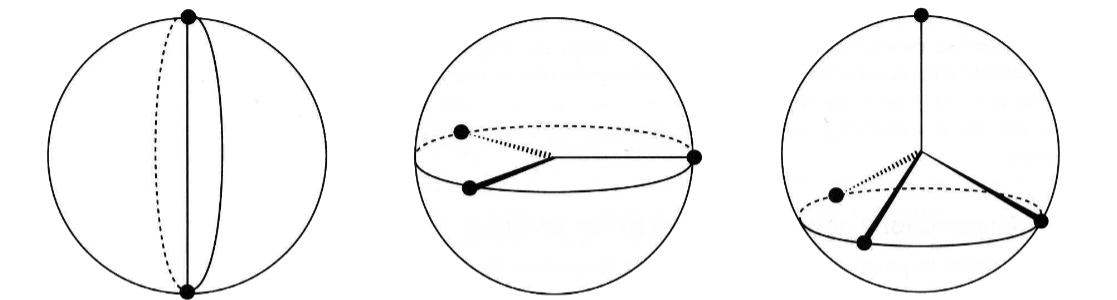
name the following geometry names to the following images
linear, trigonal, tetrahedral
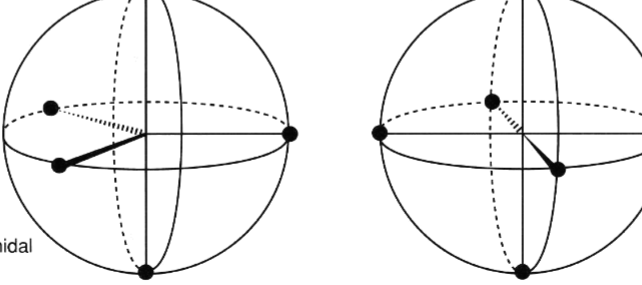
name the following geometry names to the following images
trigonal bipyramidal, octahedral
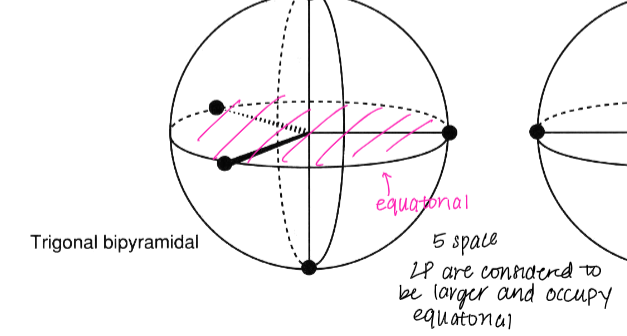
the trigonal bipyramidal geometry is associated with the ( what # - space) and its electrons are (smaller/larger) and occupy (equatorial/ not equatorial)
5 -space, larger, equatorial
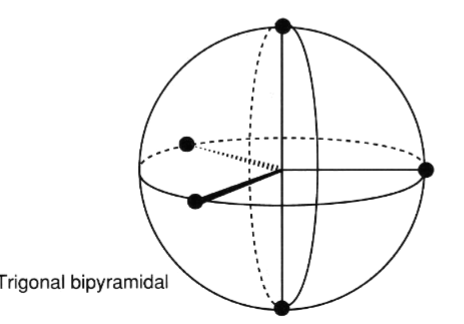
label where the electrons would be positioned
what are atomic orbitals?
what are the different types of orbitals?
the probability of the location of finding electrons.
s, p, d, and f
what are molecular orbitals?
atomic orbitals on different atoms overlap located next to each other to form molecular orbitals
draw the node shape for each of the orbitals:
s
Py
Pz
Px
d
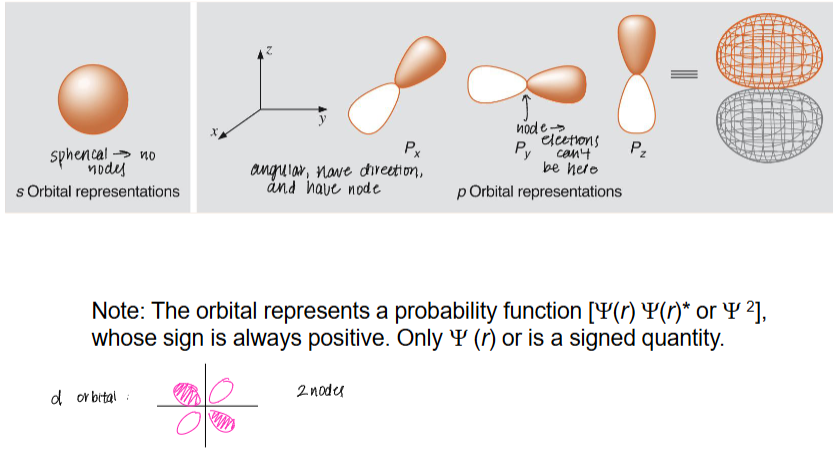
angular (have/don’t have direction) and (have/don’t have) nodes
have, have
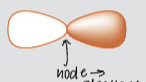
what is a node?
a place where electrons can’t be
locate the node in the following image
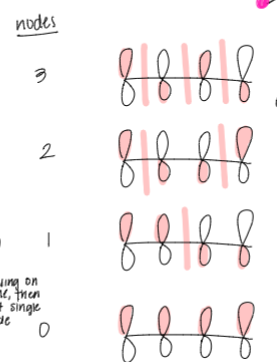
how many nodes does the d orbital have? draw the d orbital and draw the nodes
draw the following orbitals, draw the resulting orbitals when they are mixed together, and draw the orbitals that are leftover in each interaction. Give the names of all orbitals.
1 s orbital ± 1 p orbital
1 s ± 2 p orbitals
1 s orbital ± 3 p orbitals
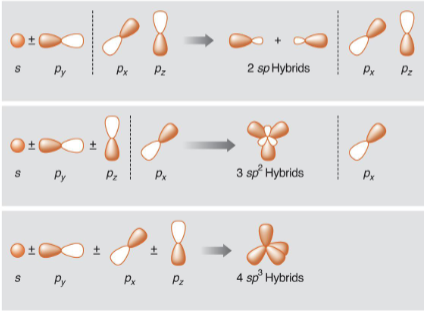
what are hybrid orbitals?
linear combinations of atomic orbitals
with hybrid orbitals, are phases arbitrary?
yes
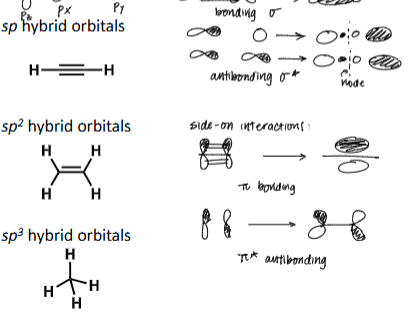
Draw 2 atomic orbitals that results in sigma bonding interaction and state whether the bond interaction is constructive or destructive
Draw 2 atomic orbitals that results in anti sigma bonding interaction and state whether the bond interaction is constructive or destructive
Draw 2 atomic orbitals that results in pi bonding interaction and state whether the bond interaction is constructive or destructive
Draw 2 atomic orbitals that results in anti sigma bonding interaction and state whether the bond interaction is constructive or destructive
true or false: adding an electronegative atom lowers the energy of the LUMO
true
true or false: adding an electronegative atom decreases the energy of all orbitals, whether unfilled or filled
true
2 bonds being subtracted from each other results in (bonding/antibonding) and (no node/a node)
antibonding, a node
2 bonds being added to each other results in (bonding/antibonding and (no node/ a node)
bonding, no node
true or false: 2 filled orbitals can react if they have proper geometry
true
the reaction between 2 filled orbitals results in (favorable/unfavorable) (sigma/pi/anti sigma/ anti pi), (increasing/decreasing) the energy more than (sigma/ anti sigma/ pi/ anti pi) (increasing/ decreasing) in energy
unfavorable, anti sigma, increasing, sigma, decreasing
true or false: the addition of an electronegative atom results in the increase of the bonding interaction and lowers the antibonding interaction.
true
draw all bonding and antibonding orbitals for sigma and pi
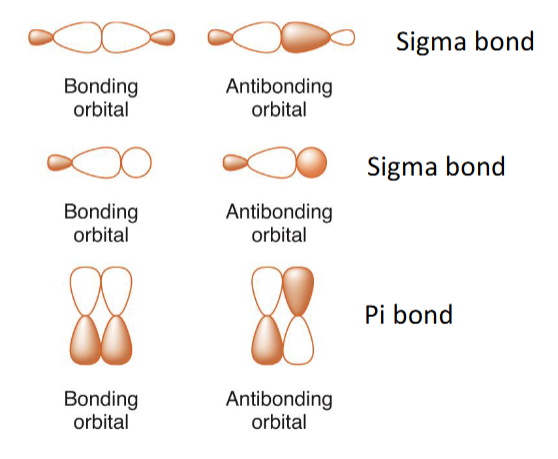
going from binding to antibonding, how many nodes do you increase by?
1
when orbitals overlap:
bonding orbital (stabilize/ destabilize), and they do (up/down in energy)
antibonding orbital (stabilize/ destabilize), and they go (up/down in energy)
stabilize, down
destabilize, up
electrophiles look for (filled/empty) orbitals and they go to (pi*/pi/sigma*/sigma) before (sigma*/sigma/pi/pi*)
empty, pi*, sigma*
true or false: pi* are lower in energy than sigma *
true
electrophiles look for the (LUMO/HOMO)
LUMO
true or false: heteroatoms are more electronegative than carbon
true
true or false: electronegative atoms have lower atomic orbitals
true
true or false: polar molecules (A-B) bonds are stronger than the average bond strengths of individual bonds of (A-A) and (B-B)
true
true or false: the larger the electronegativity difference, the more polarized the bonds
true
electrophiles want to have (higher/lower) antibonding orbitals
lower
functional groups with electronegative atoms tend to be electron (winthdrawing/donating)
withdrawing
describe what inductive effect is
withdrawing electrons to the more electronegative atoms
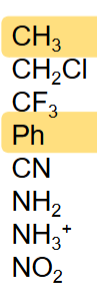
rank the following compounds from most to least electronegative
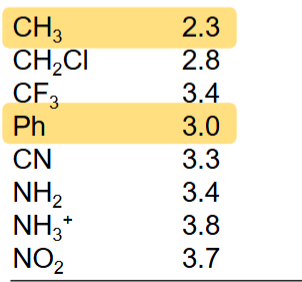
s orbital are (less/more) electronegative than p orbitals
more
the best Lewis structure is the one that puts the (negative charge/positive charge) on the most electronegative atom
negative charge

state whether the following guidelines for resonance structures are true or false:
all true
more covalent bonds on a resonance structure makes the structure (more/less) stable
more
true or false: resonance structures where all of the atoms have a complete valence shell of electrons are stable and make larger contribution to the hybrid
true
LP that are perpendicular to the p atomic orbitals (are/are not) a part of the pi system
LP that are in a p orbital (are/ are not) a part of the pi system
are not, are
true or false: nucleophiles have HOMO and electrophiles have LUMO for bond making
true
Name 3 sources of nucleophilicity and rank them in the order of importance
1) LP, 2) pi bonds of C-C double or triple bonds 3) sigma bonds of grinards and hydrides
name 3 sources of electrophilicity
lewis acids that lack full octet
sigma bonds of LG-E
pi bonds → polarized double/triple bonds or C-C double/triple bonds attached to an electrophilic atom
give 3 examples of lewis acids that lack a full octet
carbocations, aluminum compounds and boron compounds
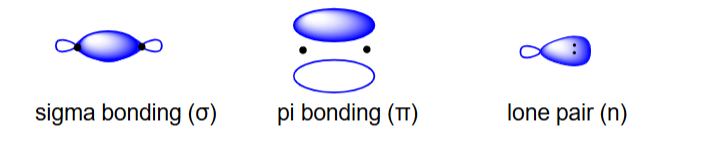
draw and state the three types of bonding interaction orbitals that are associated with nucleophiles
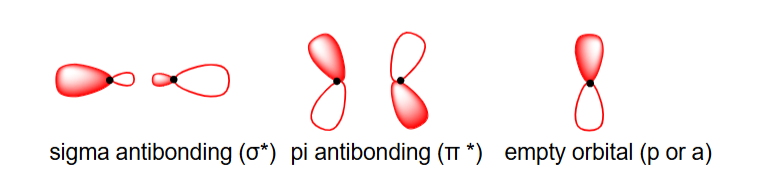
draw and state the three types of bonding interaction orbitals that are associated with electrophiles
true or false: reactions take place between a filled orbital (nucleophile) and an unfilled orbital (electrophile)
true
Draw the SN2 reaction and the orbital associated with it
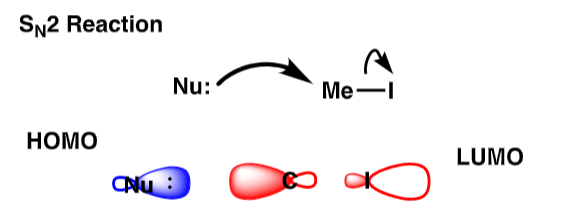
true or false: putting electron density into an antibonding orbital breaks the bond
true
define what a Bronsted-Lowry acid and base is and give the chemical equation between an acid and a base, labelling the conjugate acid and conjugate base
Bronsted-Lowry acids can donate H ions
Bronsted-Lowry bases can accept H ions
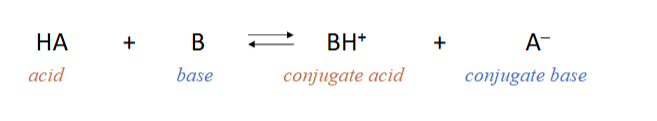
the smaller the pKa, the (stronger/weaker) the acid and the (stronger/weaker) its conjugate base
stronger, weaker
draw the following compounds and their conjugate bases, alongside the pKa values of the acids:
hydronium ion, acetic acid, ammonium ion, phenol, diethylmalonate, water, ethanol, acetone, ethyne, and ammonia

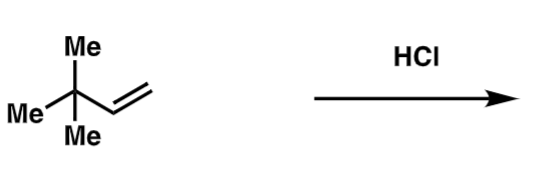
draw the mech and provide the product to the following reaction
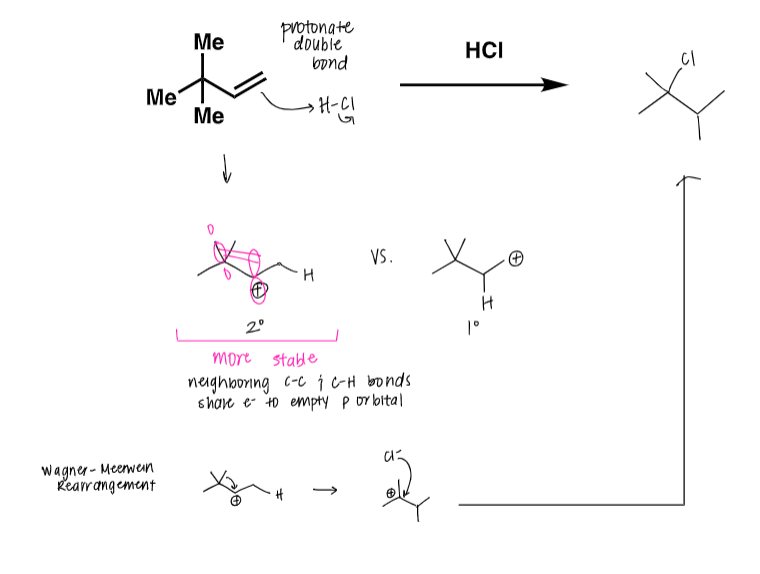
true or false: in the more stable carbocation position of a secondary prime structure, the neighboring C-C and C-H bonds share the electrons to empty the p orbital
true
true or false: when creating a carbocation, put the carbocation on the more substituted side of the former C=C bonds
true
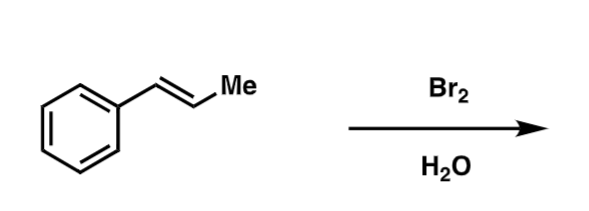
draw the product and mech for the following reaction, and identify the most nucleophilic position the and electrophile
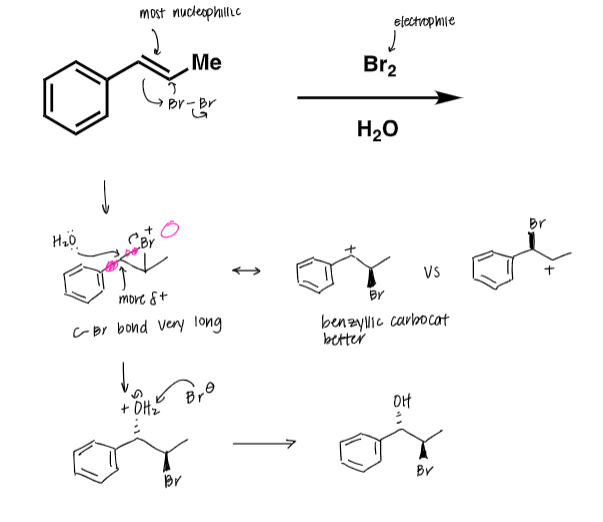
draw the benzylic and allylic carbocation positions
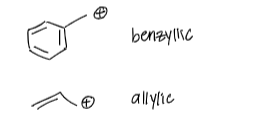
true or false: it is better to put the carbocation charge at the benzylic and allylic positions
true
define the following types of dienes:
isolated diene
cumulative diene
conjugated diene
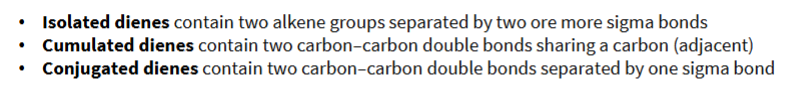
true or false: the 2 C=C bonds in a cumulative diene do not interact with each other (meaning they don’t overlap one another).
true
true or false: you need a double bond, single bond, double bond to be a conjugated olefin
true
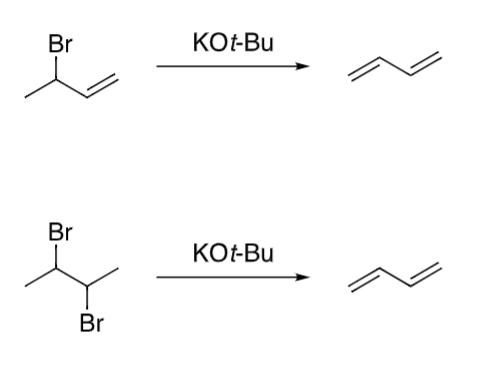
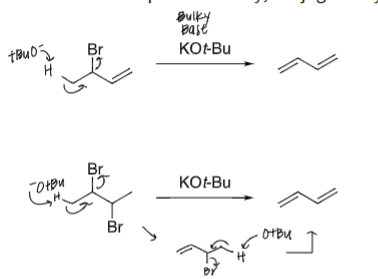
draw the product and mech for the following reactions
the C-C bond in conjugated diene are (shorter/longer) than typical sp3 C bonds
shorter
conjugated dienes are (more/less) stable than isolated dienes
more
true or false: the more substituted olefines are more stable
true
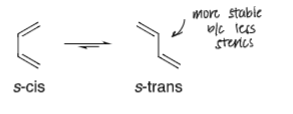
draw out the cis and trans conformations of a conjugated diene and state which one is more stable
when drawing the p orbitals of a conjugated diene, they should be drawn (not interacting/interacting)
interacting
draw out the molecular orbitals of conjugated systems, including the number of nodes on each level, and identify where the ground state is
draw out the phasing of orbitals on the LUMO and the HOMO
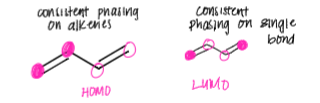
draw an orbital diagram of a conjugated diene, labelling the pi bonding MO and the antibonding MO and recall where the node would be on the antibonding MO
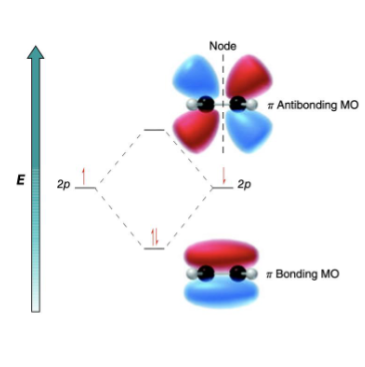
true or false: pi MO’s of conjugated systems are made up of 2 parallel p orbitals
true
w/ increasing strength (the larger the conjugated system = the more MO’s) the HOMO-LUMO energy gap (enlarges, shrinks)
shrinks
for polyenes with an even number of C’s, the HOMO resembles the (dominant Lewis structure/ a-w diradical structure) and the LUMO resembles the (dominant Lewis structure/ a-w diradical structure)
dominant Lewis structure, a-w diradical structure
what types of compounds can participate in conjugation?
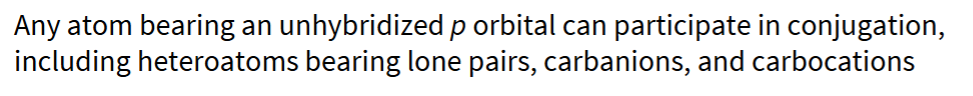
name the 5 structural elements of conjugated systems

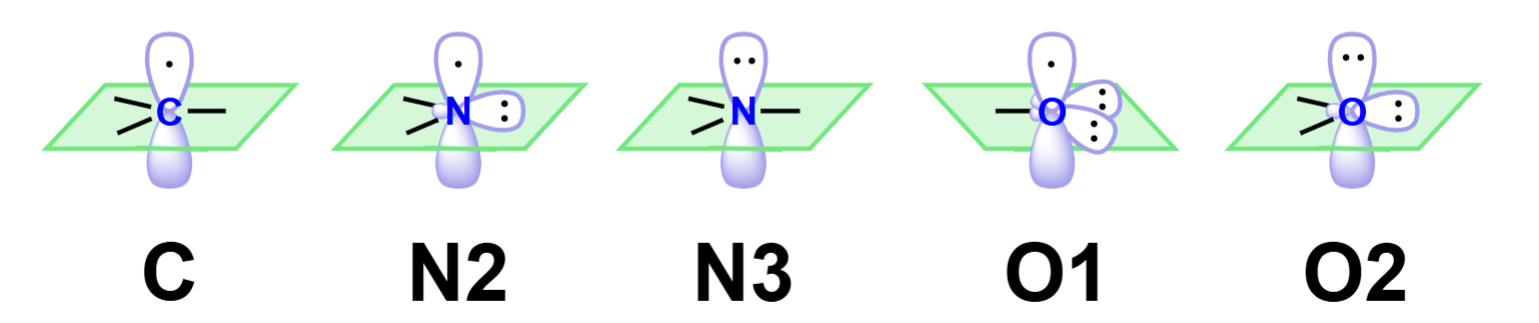
State if the following LP will be a part of the conjugated system or not
The lone pair on C will be a part, the LP on the top of N2 can participate but the other LP can’t, the LP on N3 cannot, the LP on O1 cannot, and the top LP on O2 can but the side LP cannot.

draw the following product and draw its mech
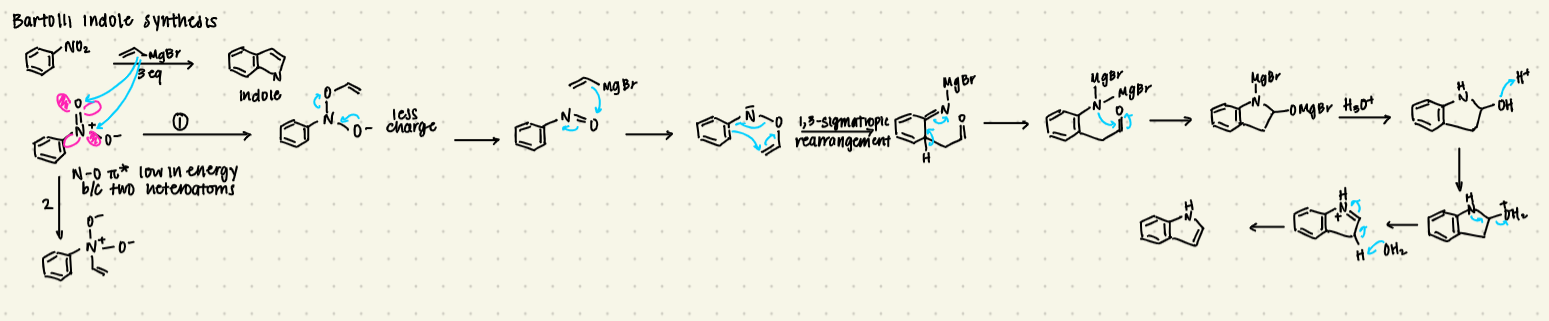
define what an electron donating group does in terms of HOMO and electron pushing

draw the resonance structure of an EDG and identify the nucleophile
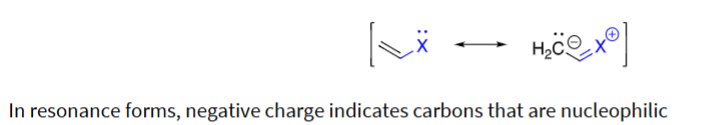

show the mech for the following grabbing an electrophile
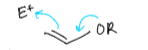
draw the mech for this grabbing an electrophile
state what an electron withdrawing group is in terms of HOMO and electron pulling
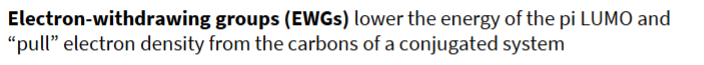
draw the resonance structure of an EWG and identify the electrophilic C
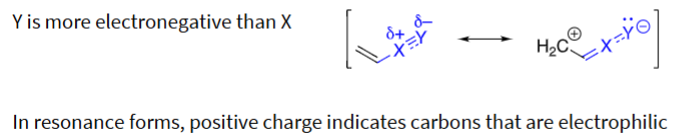
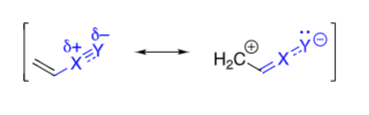
in an EWG, the Y is (more/less) electronegative than X
more