ADMET: Metabolism: Reactions: Phase I
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
Objective
What kind of reactions occur in Phase I reactions?
- Oxidations
- Reductions
- Hydrolyses
Major goal of Phase I is not to just deactivate the drug, but to also form...
functional groups "handles"
Objective
What are the kinds of oxidation reactions? (6)
- Hydroxylations (Aromatic hydrocarbon hydroxylation, Aliphatic hydrocarbon hydroxylation)
- Epoxidations
- Dealkylations (N-dealkylations, O-dealkylations, S-dealkylations)
- Heteroatom oxidations (N-oxidation, S-oxidation)
- Deamination
- Dehalogenation
What reactions occur in Phase II conjugations?
Conjugations
Why do we care about metabolic reactions? (6)
- they affect drug action in the body
- required to activate most prodrugs
- create active metabolites
- create toxic metabolites
- responsible for many drug-drug and drug-food interactions
- subject to pharmacogenetic variation
Aromatic hydroxylation makes molecules ______________ polar and provides a _________________ for Phase II conjugation reactions
- more polar
- functional "handle"
Hydroxylation commonly occurs at the __________________ site on the ring, with _____-________________ being most common in man
- least hindered
- para-hydroxylation
T/F: The site of hydroxylation cannot be changed. Site of hydroxylation cannot be influenced by other substituent properties, binding orientation
FALSE
site of hydroxylation CAN be influenced by other substituent properties, binding orientation
Hydroxylation is believed to proceed through formation of.....
arene oxides/epoxides
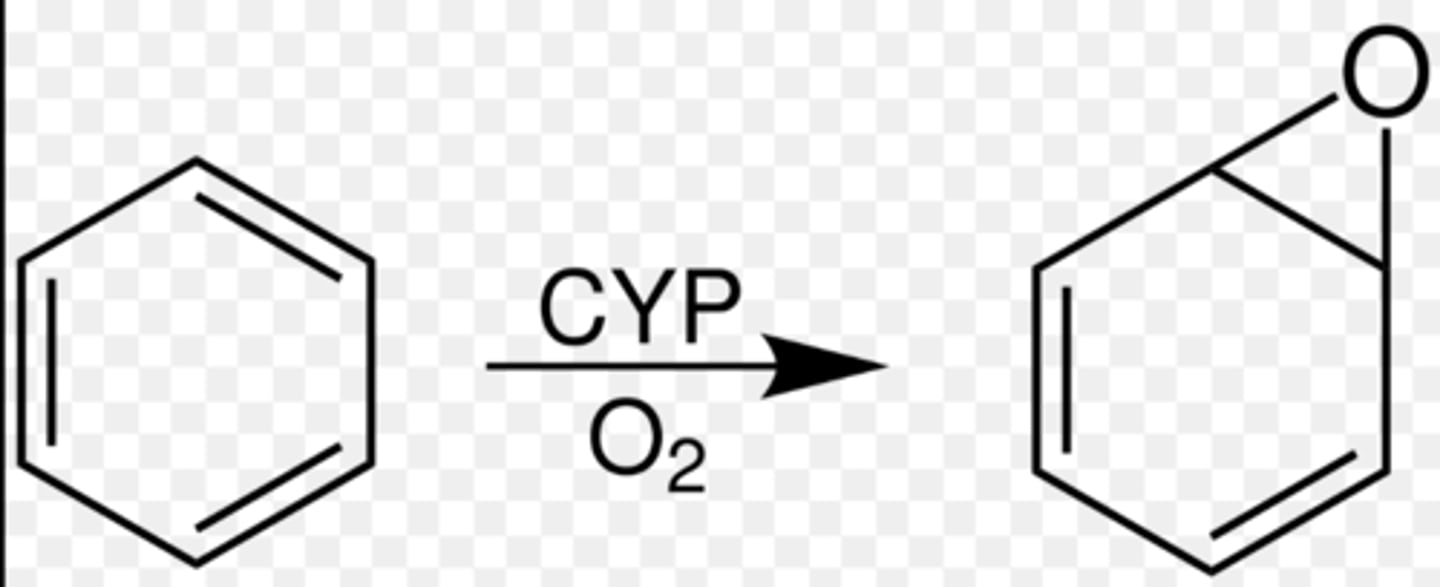
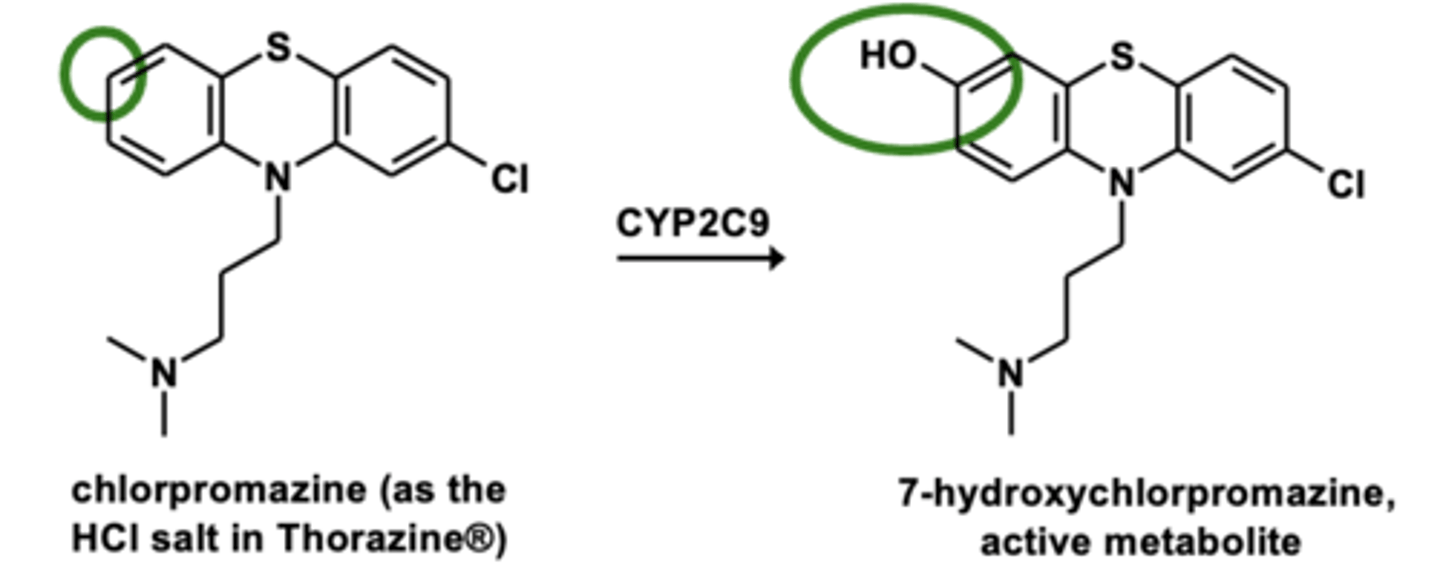
Aromatic hydroxylation is the addition of....
the addition of -OH group to aromatic ring
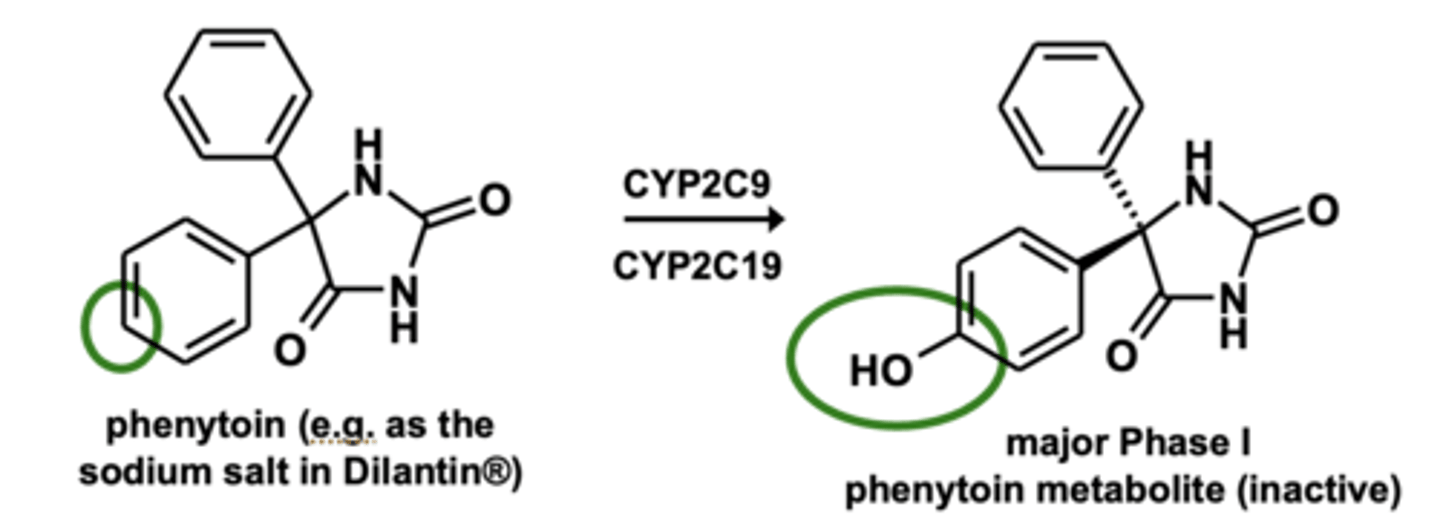
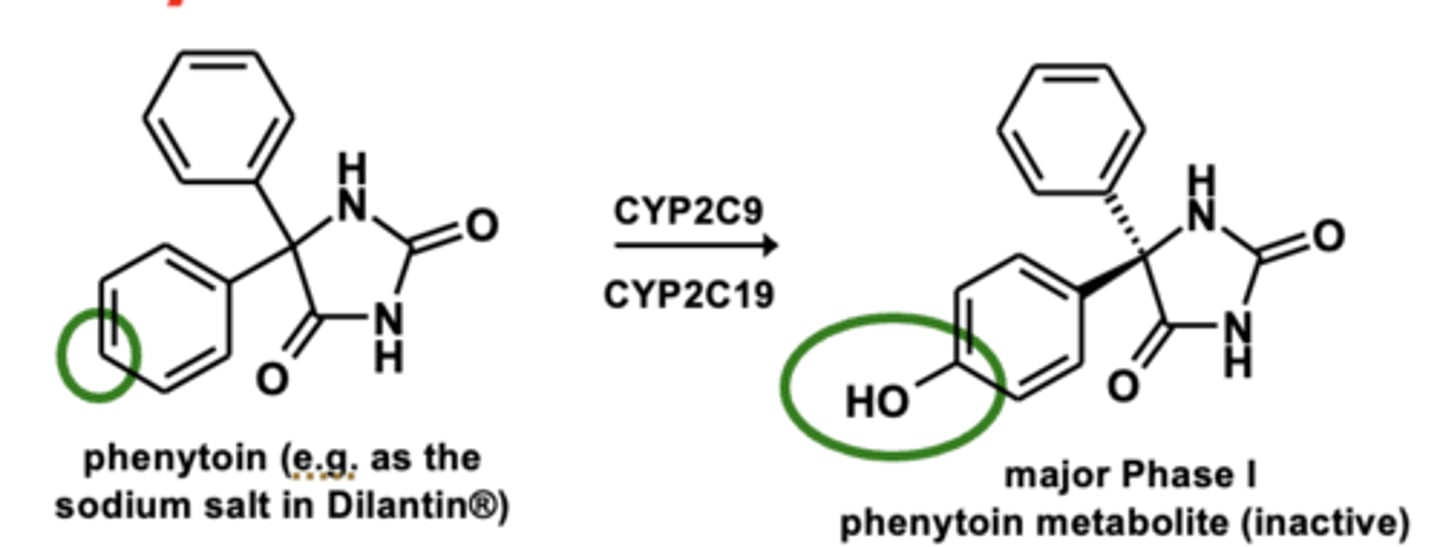
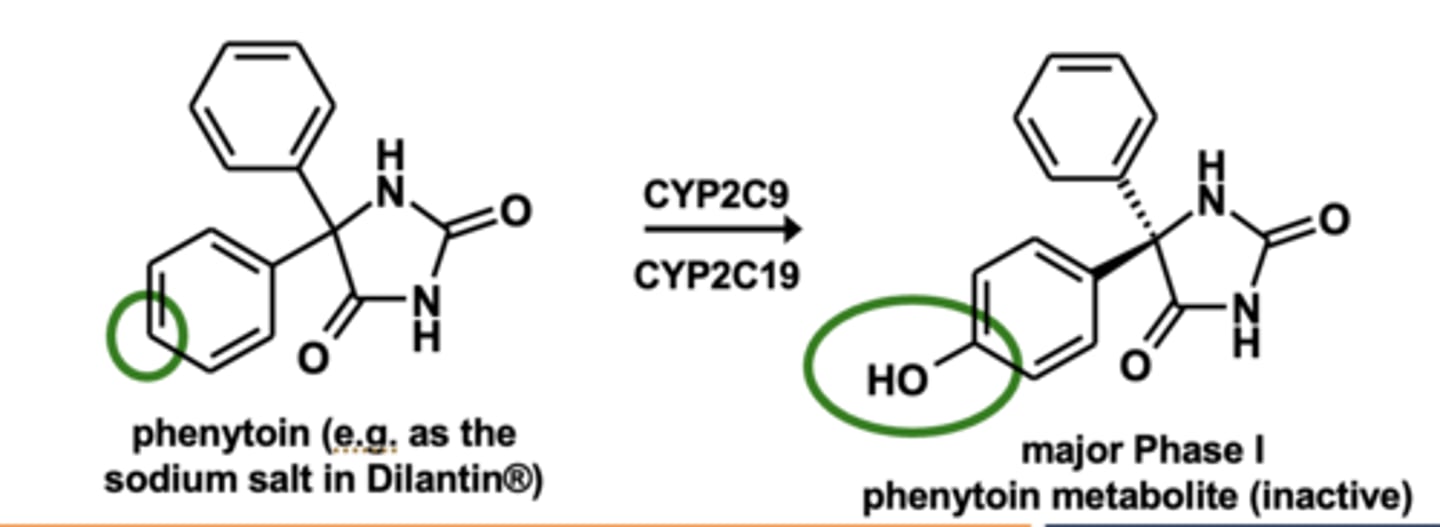
Example of aromatic hydroxylation: Phenytoin metabolism
Example of aromatic hydroxylation: Estradiol metabolism

Example of aromatic hydroxylation: Chlorpromazine metabolism
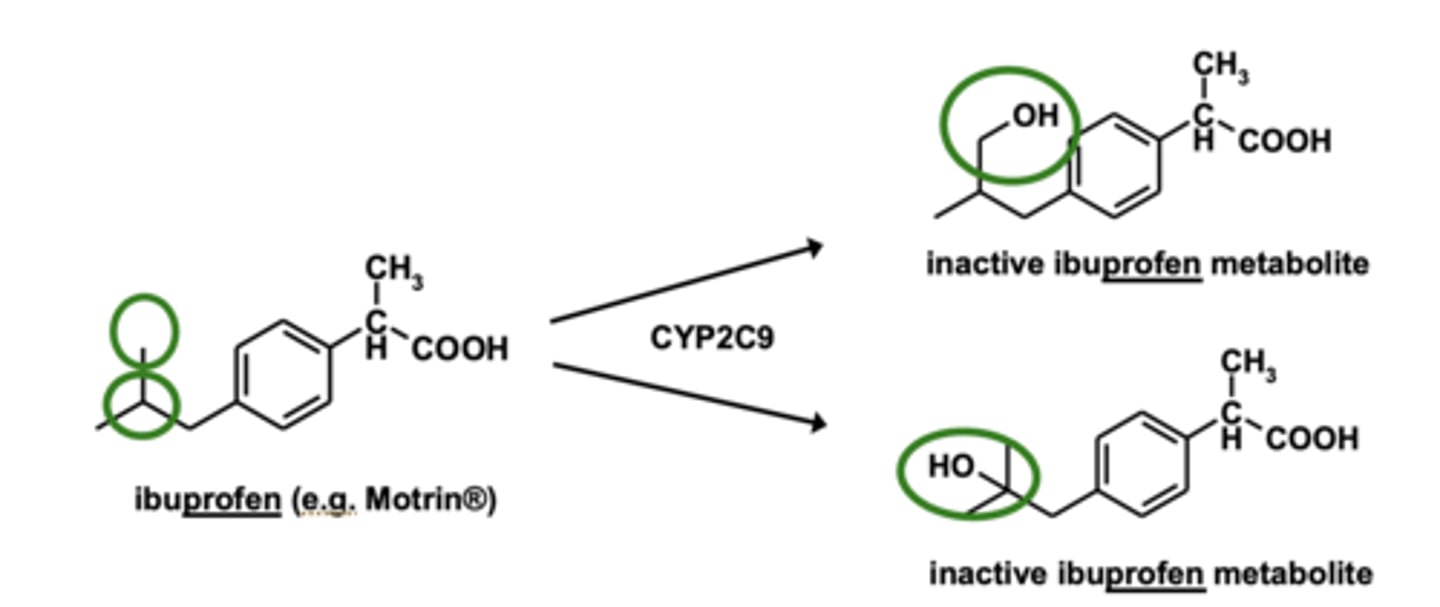
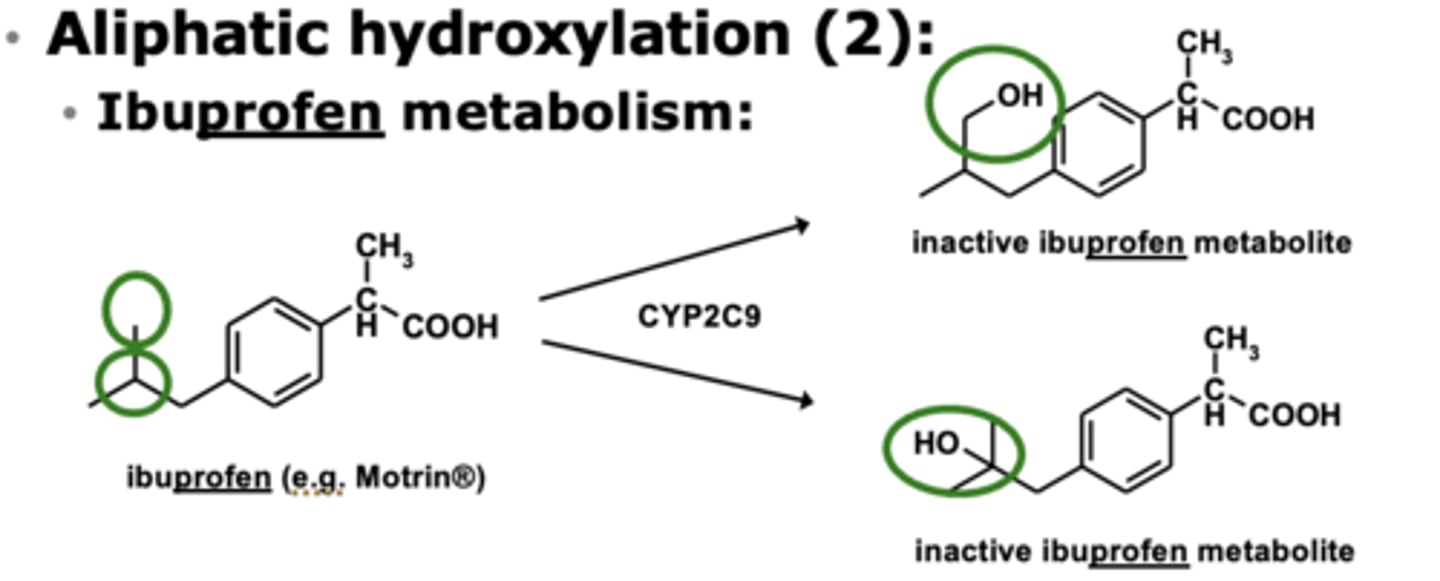
Where is the principal site of aliphatic hydroxylation? Where can this be different?
on terminal/ultimate or penultimate carbon of chain
- can be different for alkyl side chains attached to aromatic rings
Aliphatic hydroxylation is the addition of....
the addition of -OH group to carbon chain
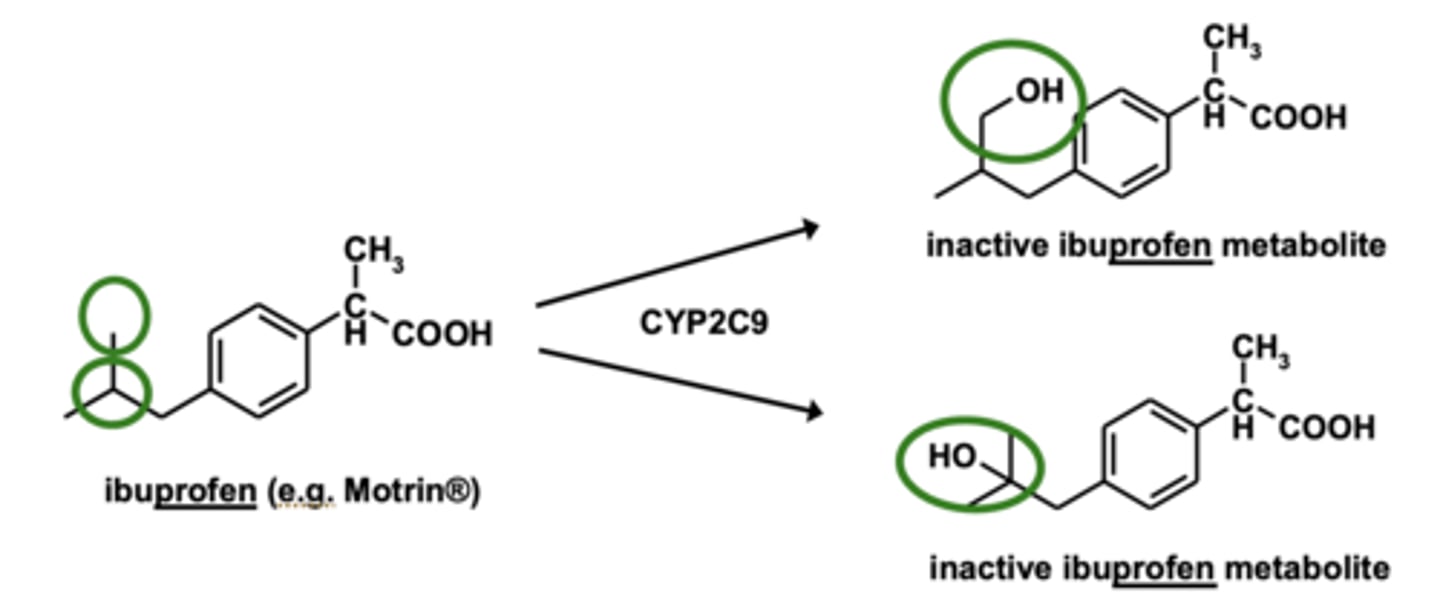
Aliphatic hydroxylation may be followed by....
further oxidation
(oxidation to -COOH group)
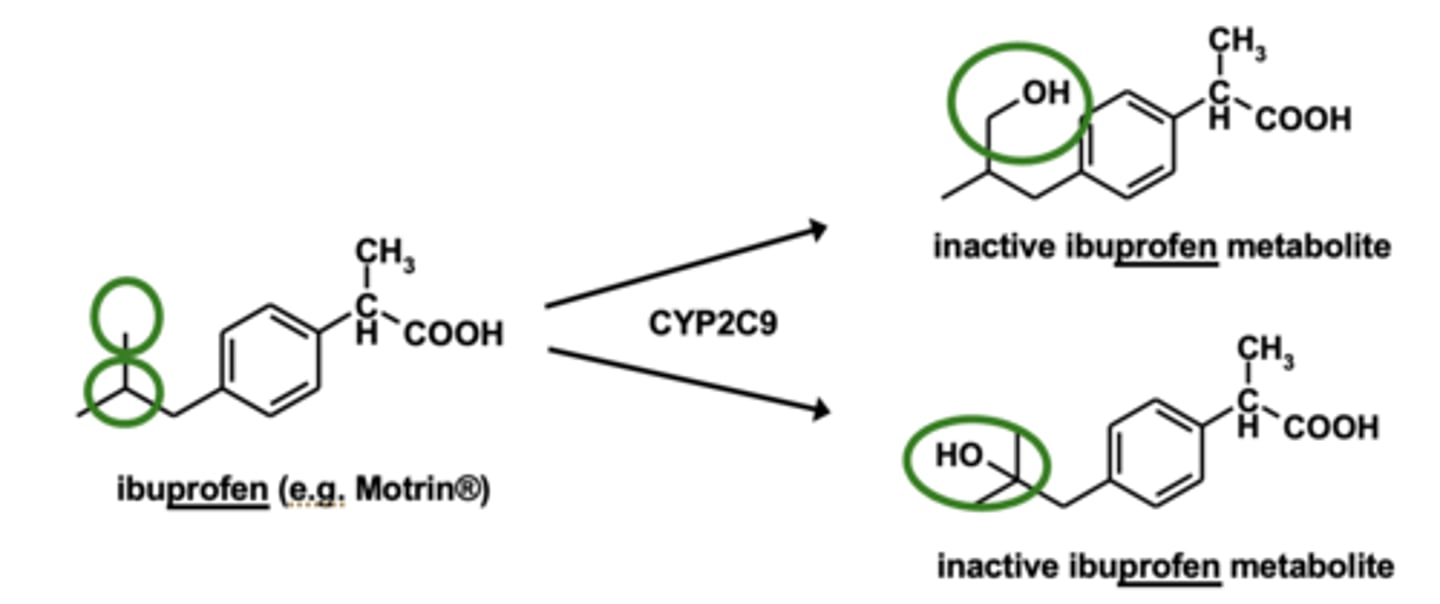
Example of aliphatic hydroxylation: Ibuprofen metabolism
Epoxidation is a reaction mechanism similar to....
aromatic hydroxylation
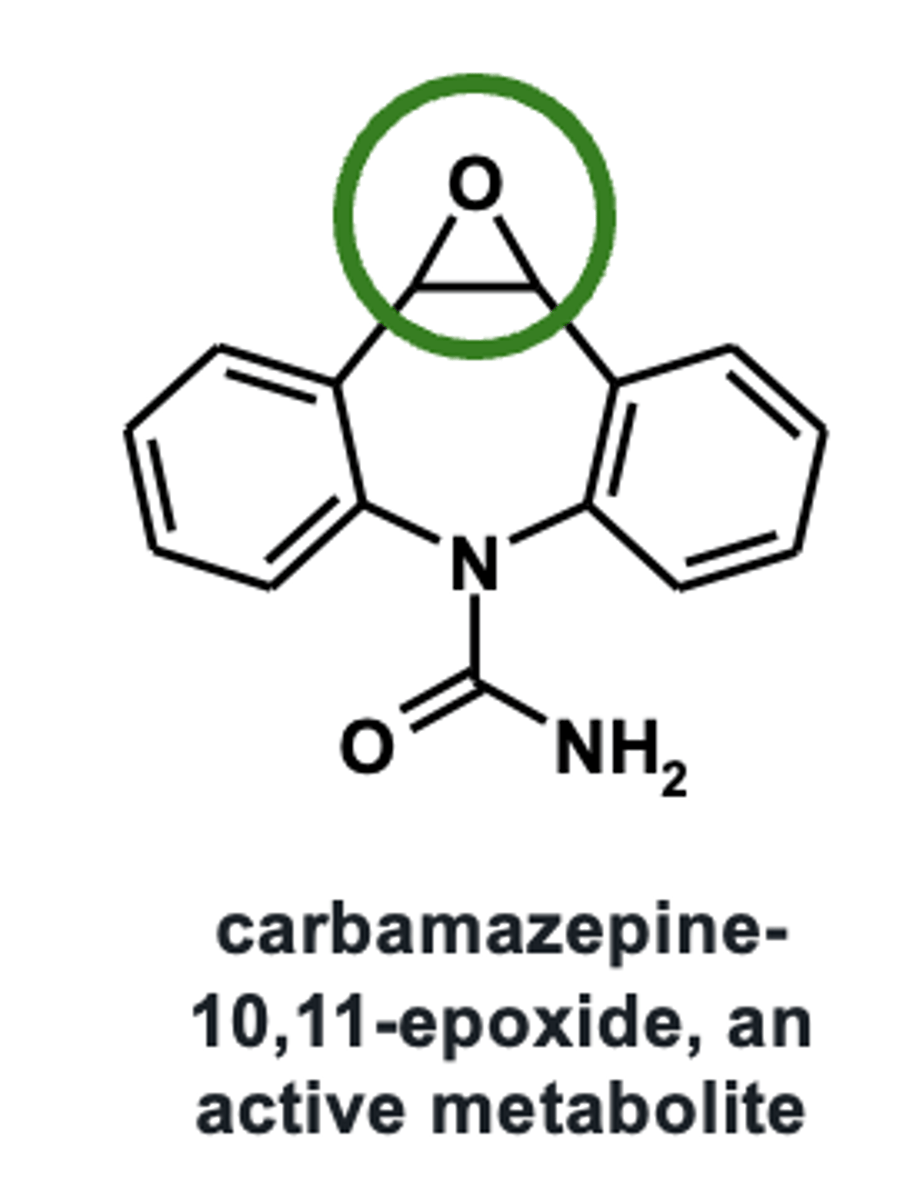
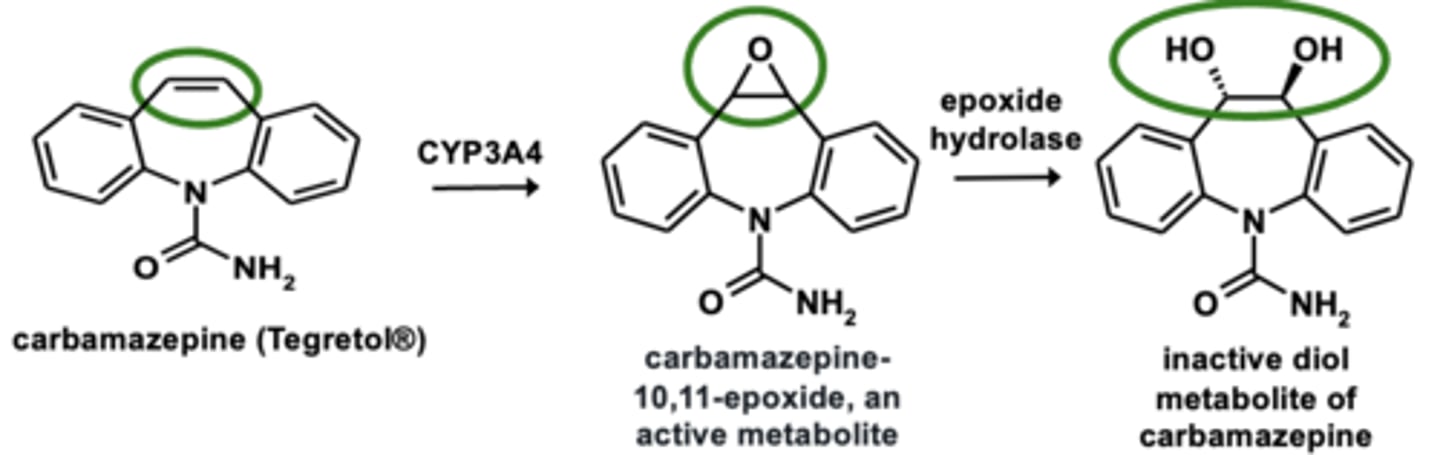
Epoxidation can lead to the formation of ________________ similarly to __________ hydroxylation via arene oxide formation
- reactive/toxic products
- aromatic hydroxylation
Example of epoxidation: Carbamazepine metabolism
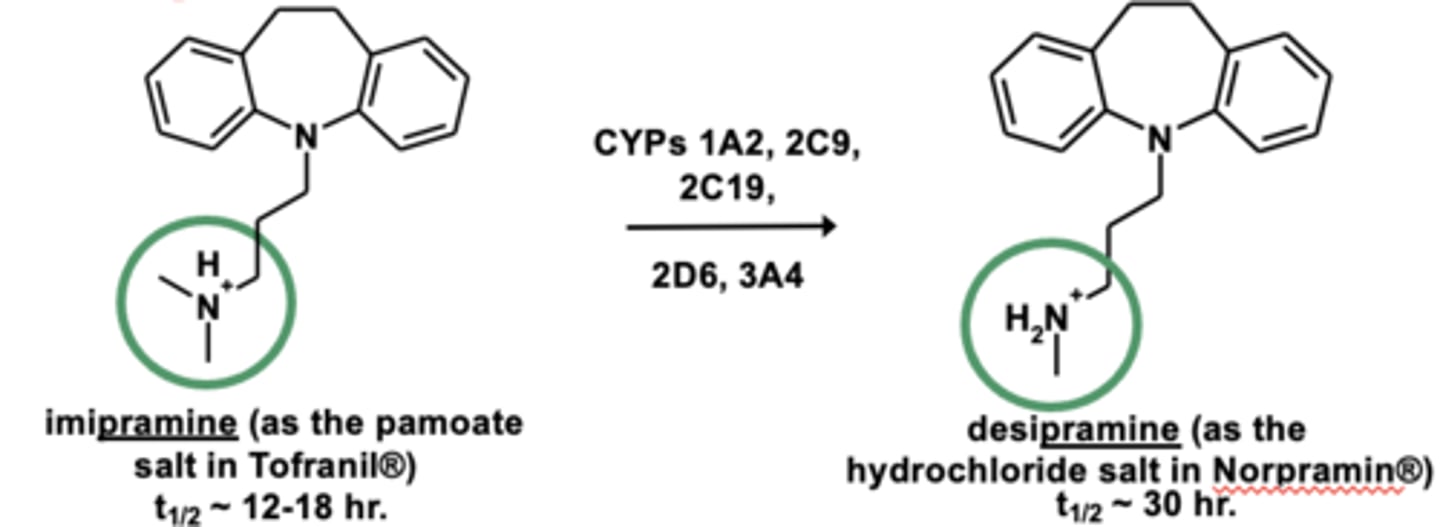
N-dealkylation is the dealkylation of....
3° -> 2° -> 1° amines (the removal of alkyl group)
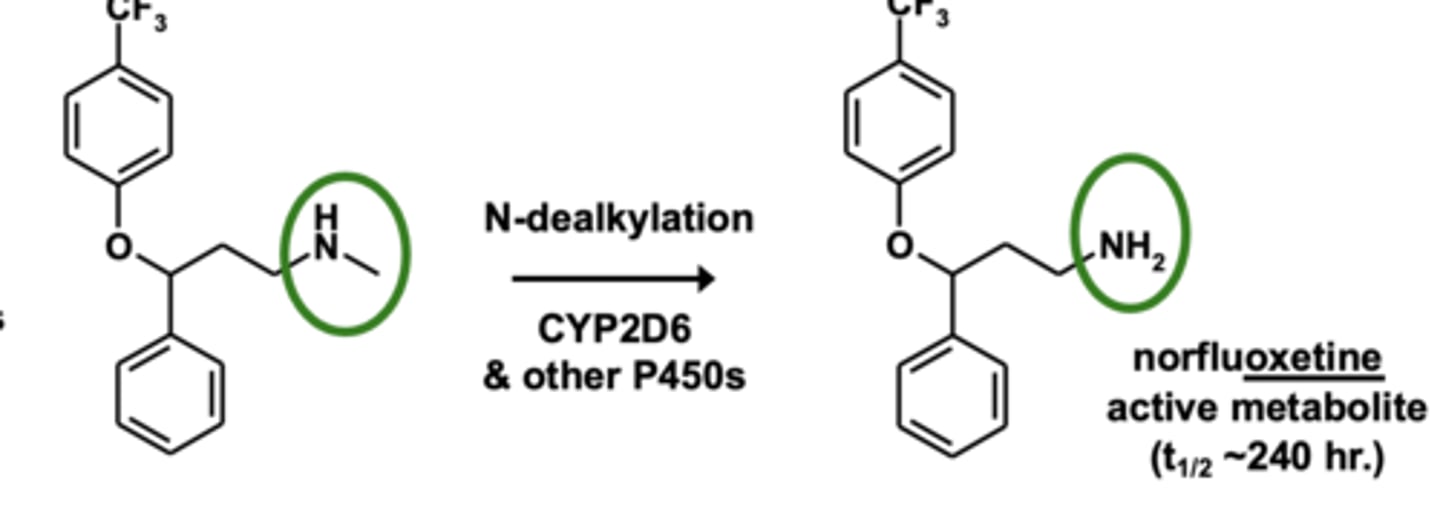
N-dealkylation is one of the most ____________ and most ___________ reactions in drug metabolism
- most common
- most important
If multiple alkyl groups are available, N-dealkylation usually starts with the....
smaller group
N-dealkylation requires the presence of an _____________ on the alkyl group
α-hydrogen on the alkyl group
Example of N-dealkylation: Impramine metabolism
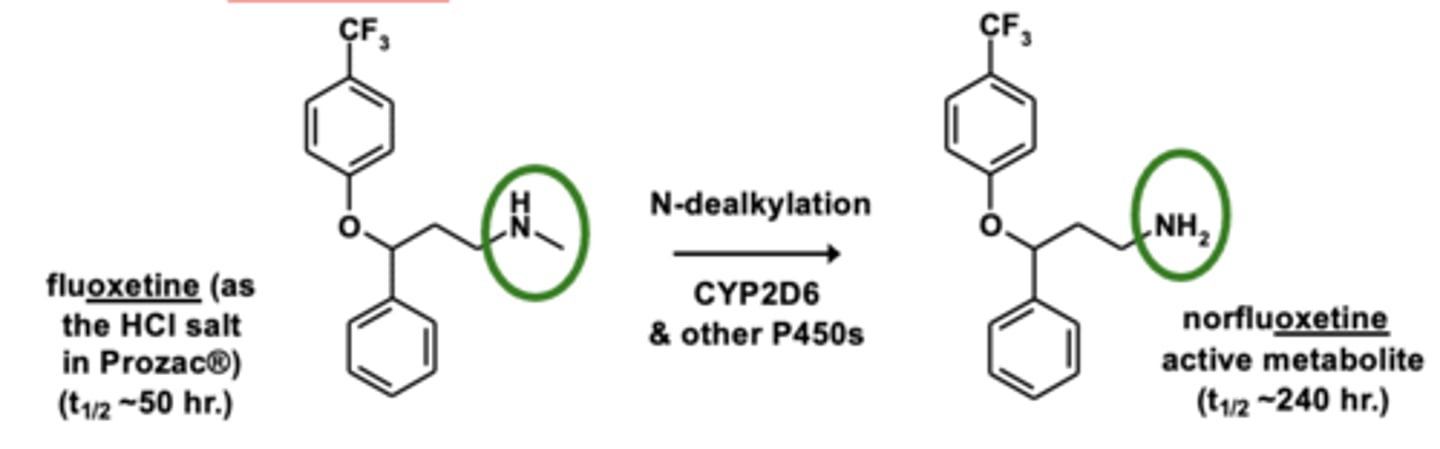
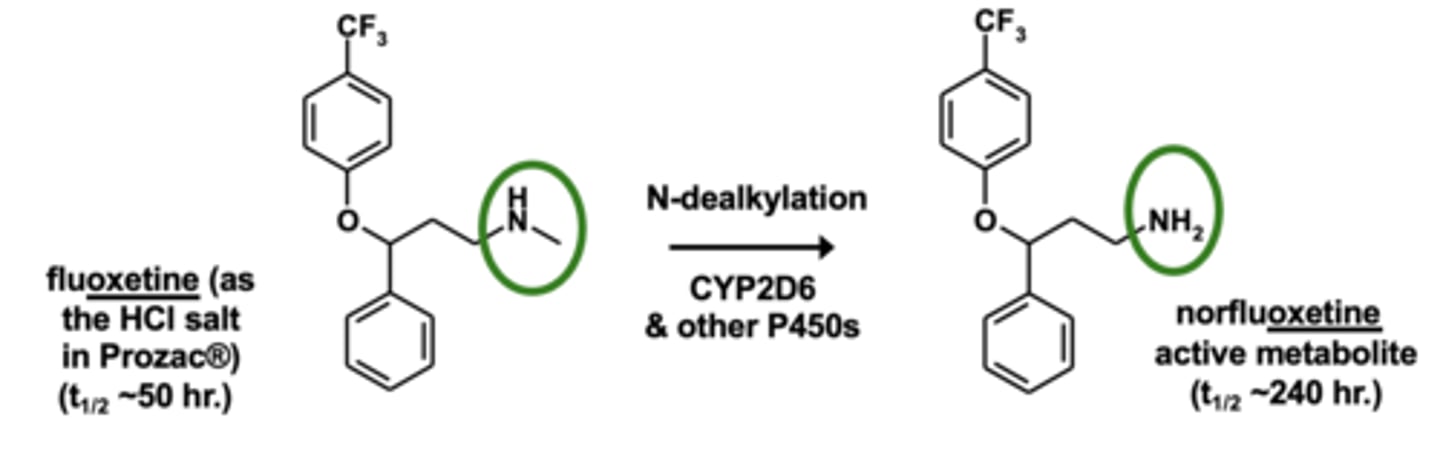
Example of N-dealkylation: Fluoxetine metabolism
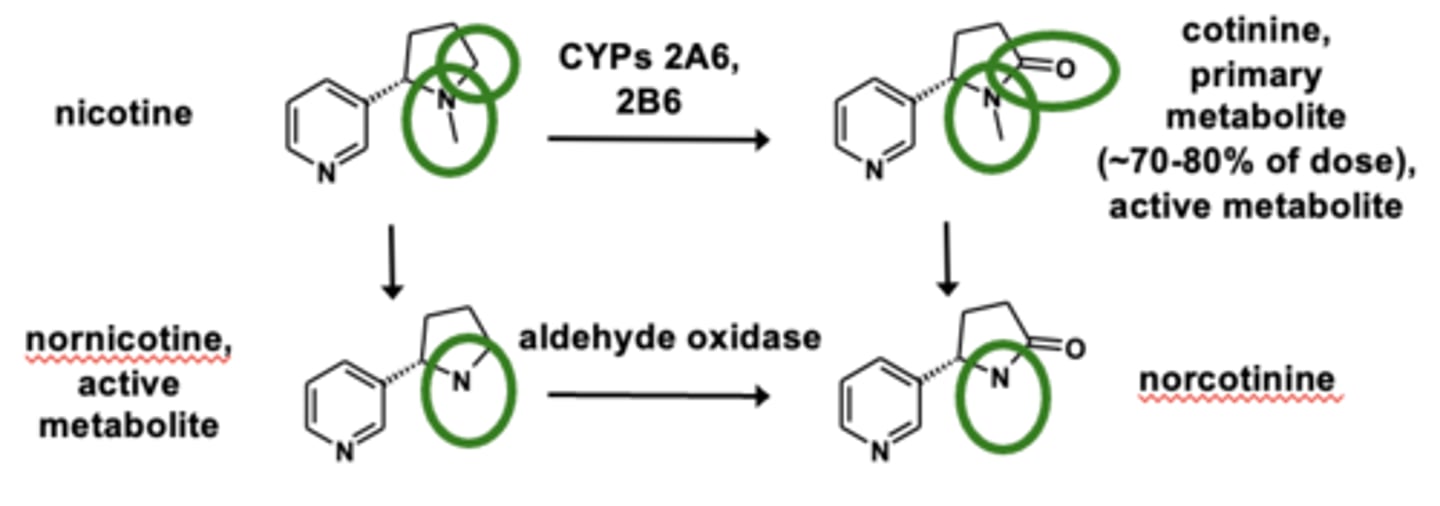
Example of N-dealkylation: Nicotine metabolism
O-dealkylation is....
the dealkylation of oxygen groups (removal of alkyl group from O)
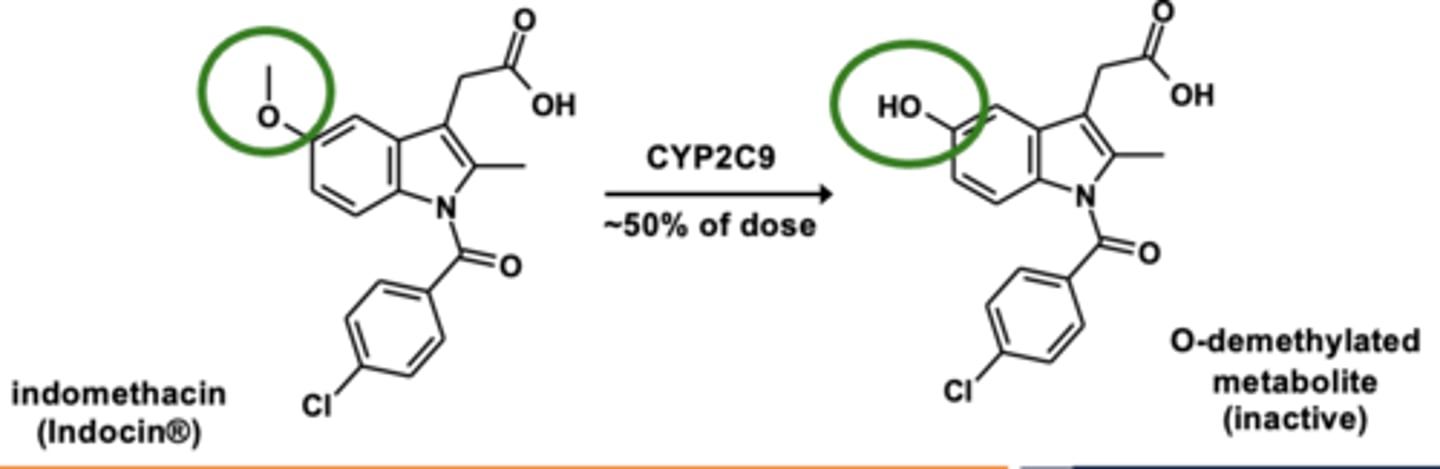
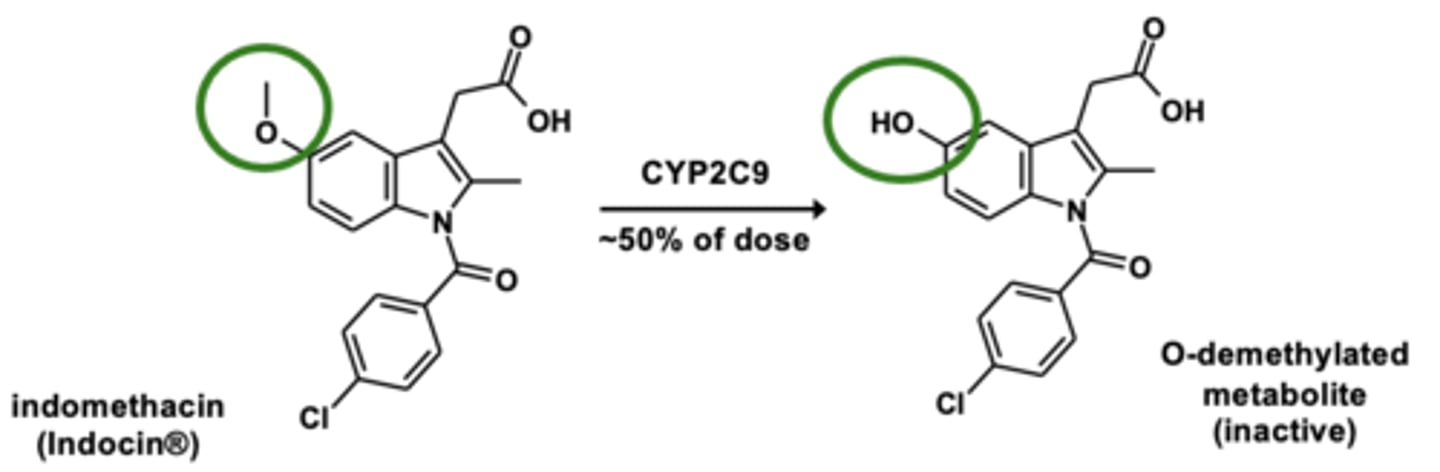
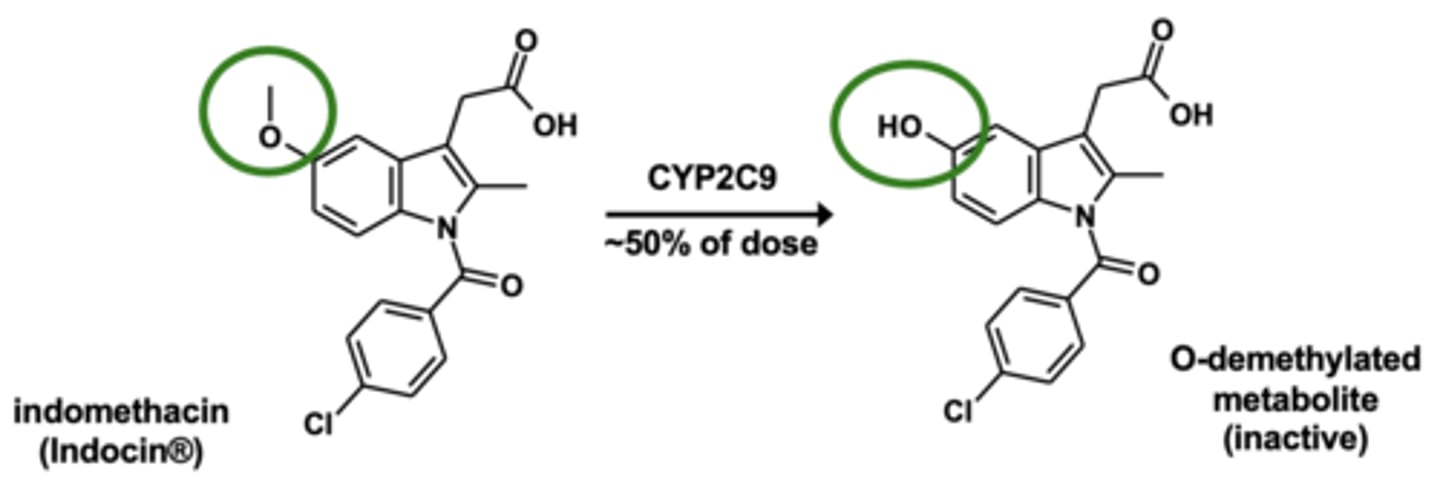
Example of O-dealkylation: Indomethacin metabolism
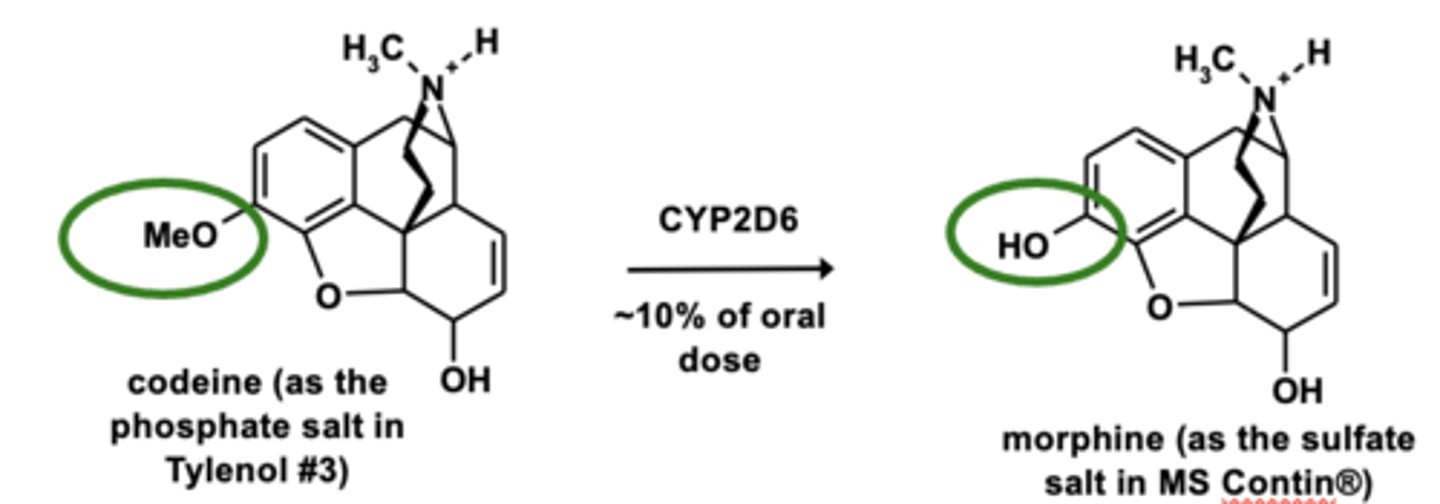
Example of O-dealkylation: Codeine metabolism
T/F: S-dealkylation is more common than O-dealkylation and N-dealkylation
FALSE
- NOT as common as O-dealkylation or N-dealkylation
S-dealkylation is similar to the mechanisms of...
O-dealkylation and N-dealkylation
N-oxidation: N atoms in both _______ and _______ can be oxidized
- both amines and amides
What can N-oxidation lead to the generation of?
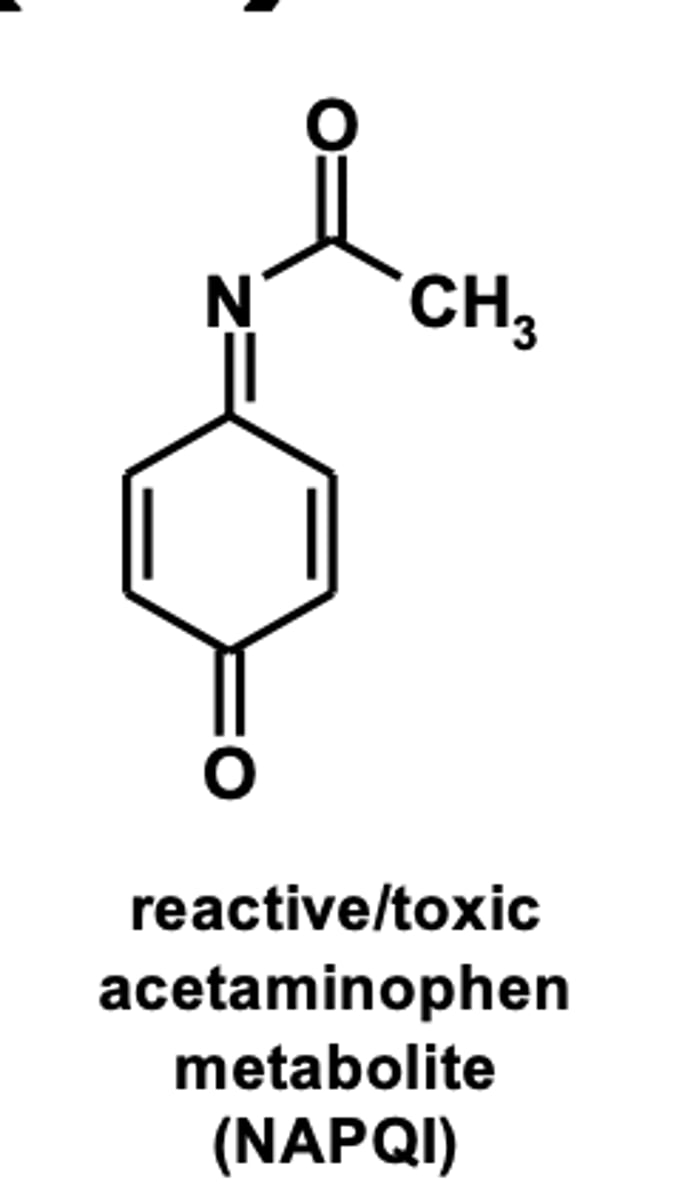
reactive/toxic electrophilic species
(ex: formation of benzoiminoquinone reactive metabolite (NAPQI ) in acetaminophen metabolism)
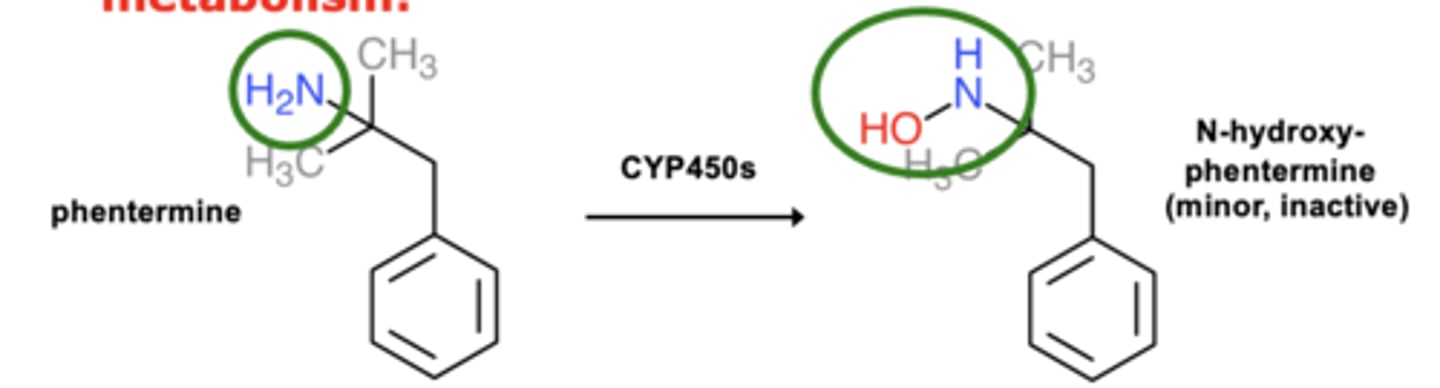
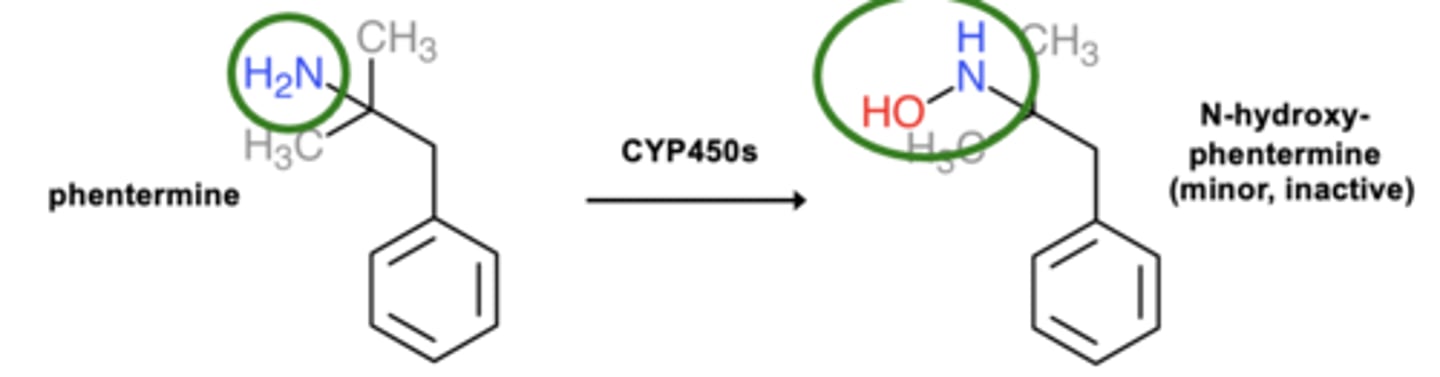
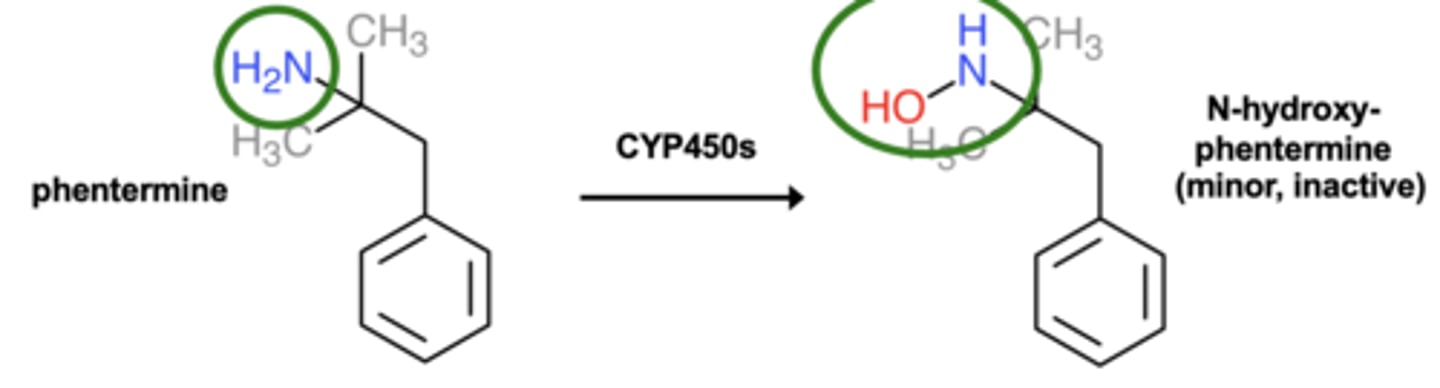
Example of N-oxidation: Phentermine metabolism
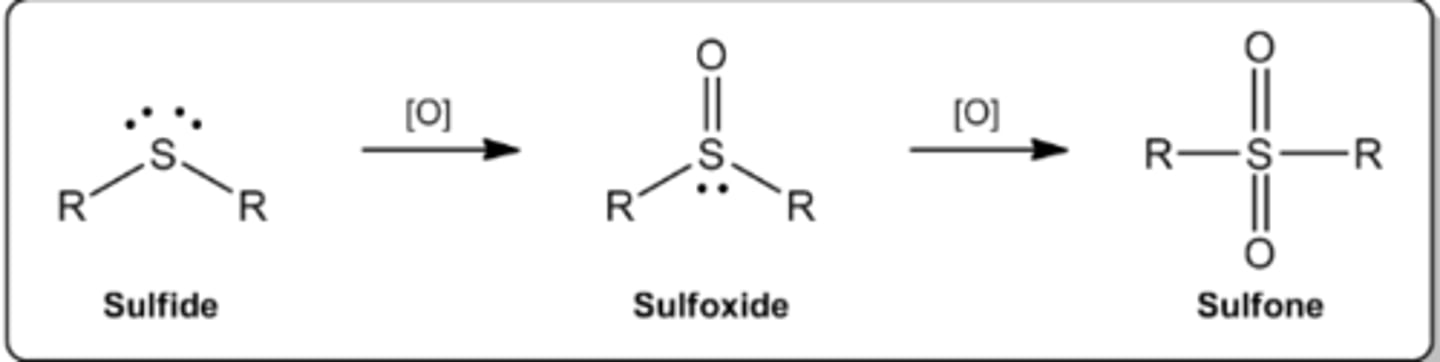
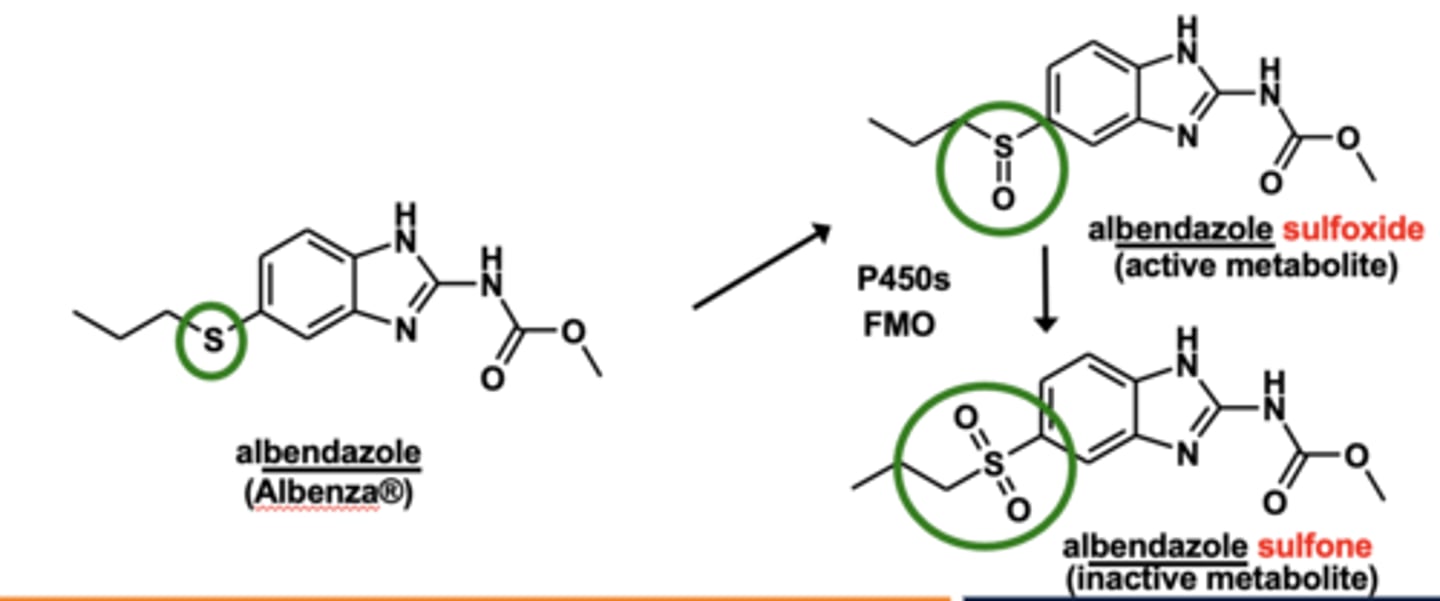
S-oxidation sees the oxidation of thioether up to ___________. Or the sulfoxide up to the ____________.
- thioether (R-S-R) -> sulfoxide (R-S(=O)-R)
- sulfoxide (R-S(=O)-R) -> sulfone (R-SO2-R)
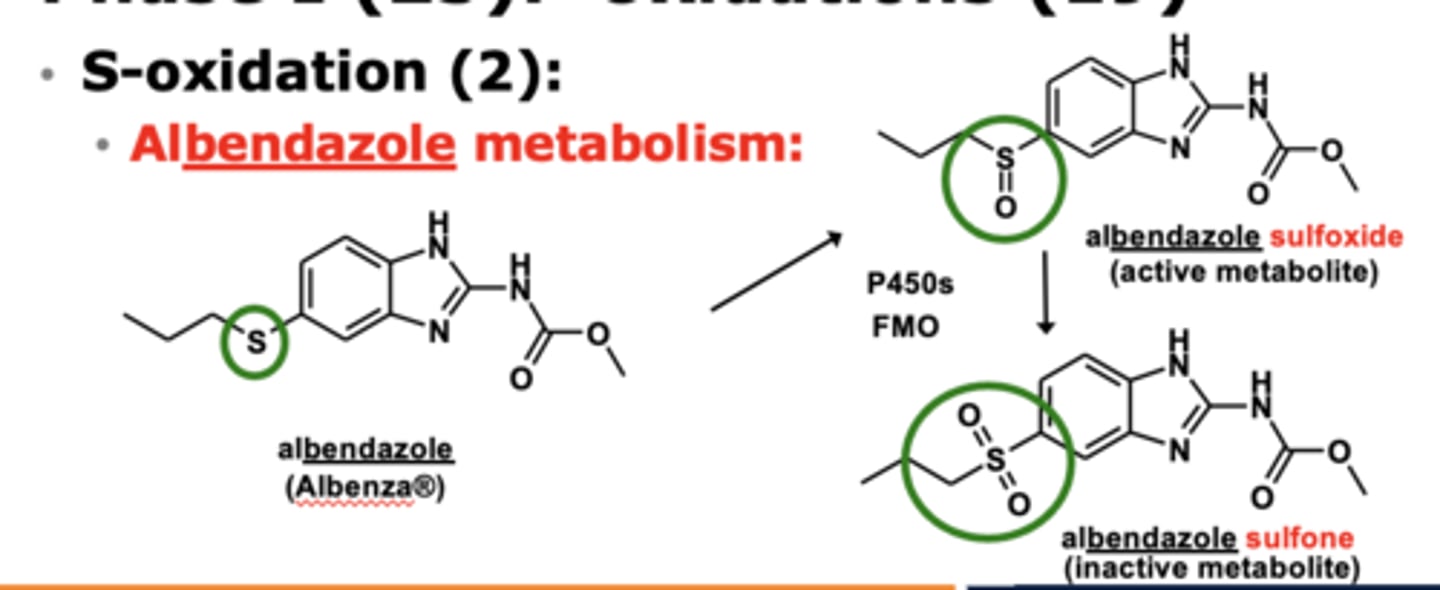
What enzymes are involved in S-oxidation? (2)
- P450 enzymes
- flavin monooxyfenase (FMO)
*FMO plays the predominant role here
Example of S-oxidation: Albendazole metabolism
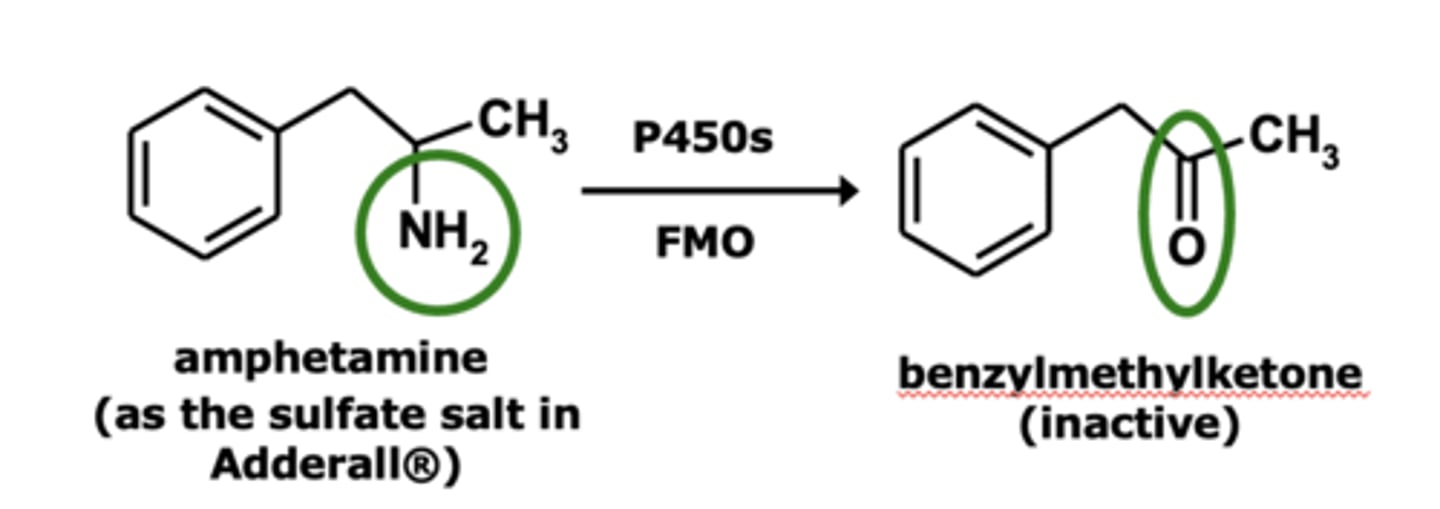
Deamination is the loss of.....
loss of N from a primary amine
Deamination results in the formation of a __________ or ____________
- ketone
- aldehyde
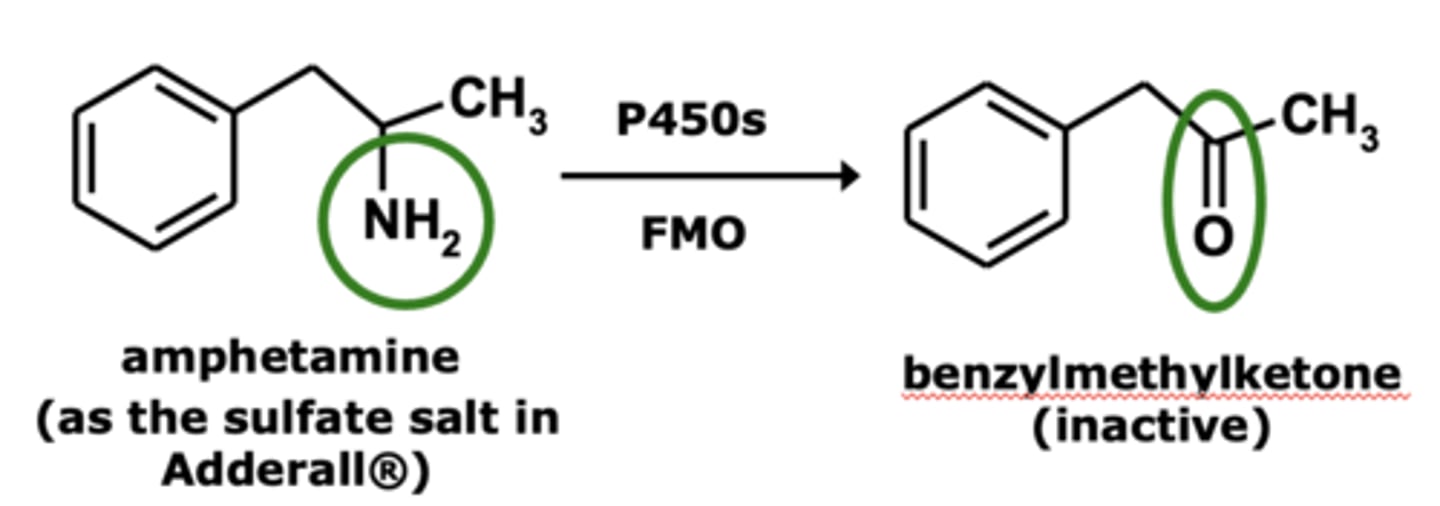
Deamination occurs with ____________ aliphatic and _____________ amines having at lease one __-_____________
- primary aliphatic and arylalkyl amines
- α-hydrogen
Deamination is mechanistically similar to...
N-dealkylation
Example of deamination: Amphetamine metabolism
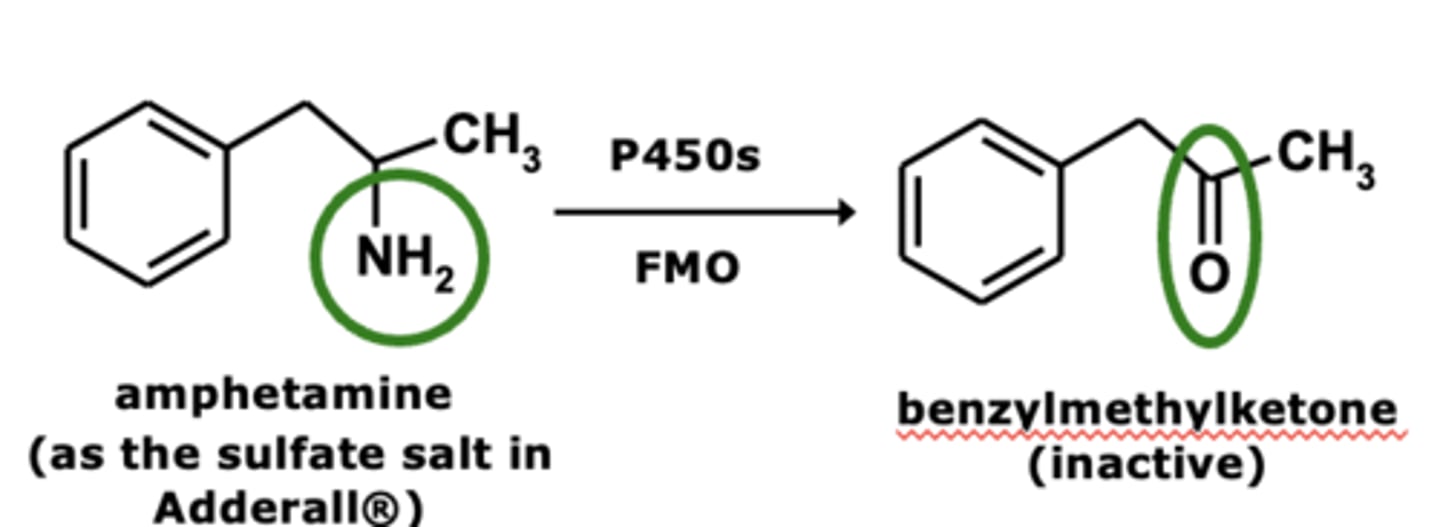
Halogenated hydrocarbons include.... (4)
pesticides, insecticides, general anesthetics, commercial solvents
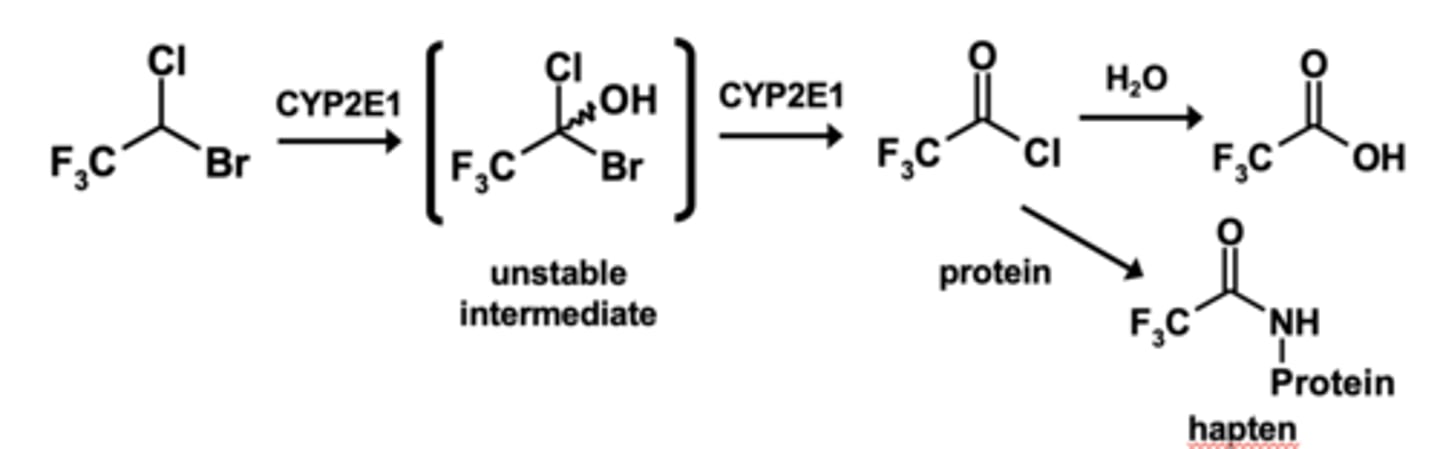
What is a key enzyme of dehalogenation ?
Key CYP enzyme 2E1
In dehalogenation there is a possibility of ______________________ being formed via metabolic processes
reactive intermediates
What form of hepatitis is associated with dehalogenation?
"Halothane-associated" heaptitis
Example of dehalogenation: Halothane* metabolism
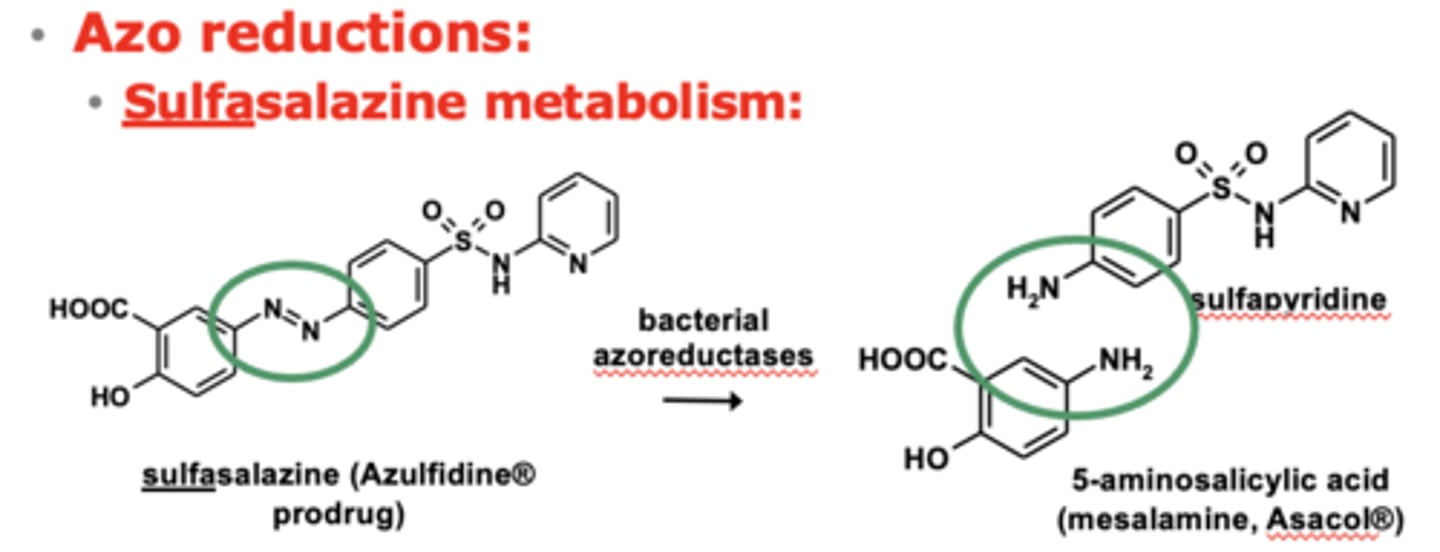
What are the different reductions reactions? (3)
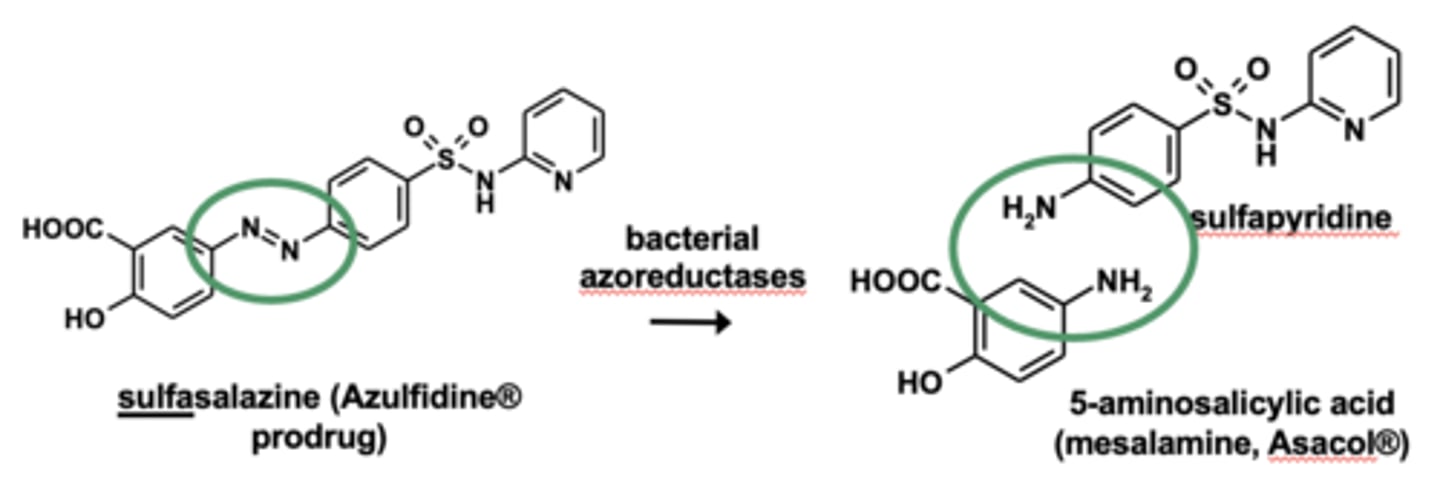
- Azo reductions
- Nitro reductions
- Carbonyl reductions
Example of azo reductions: sulfasalazine metabolism
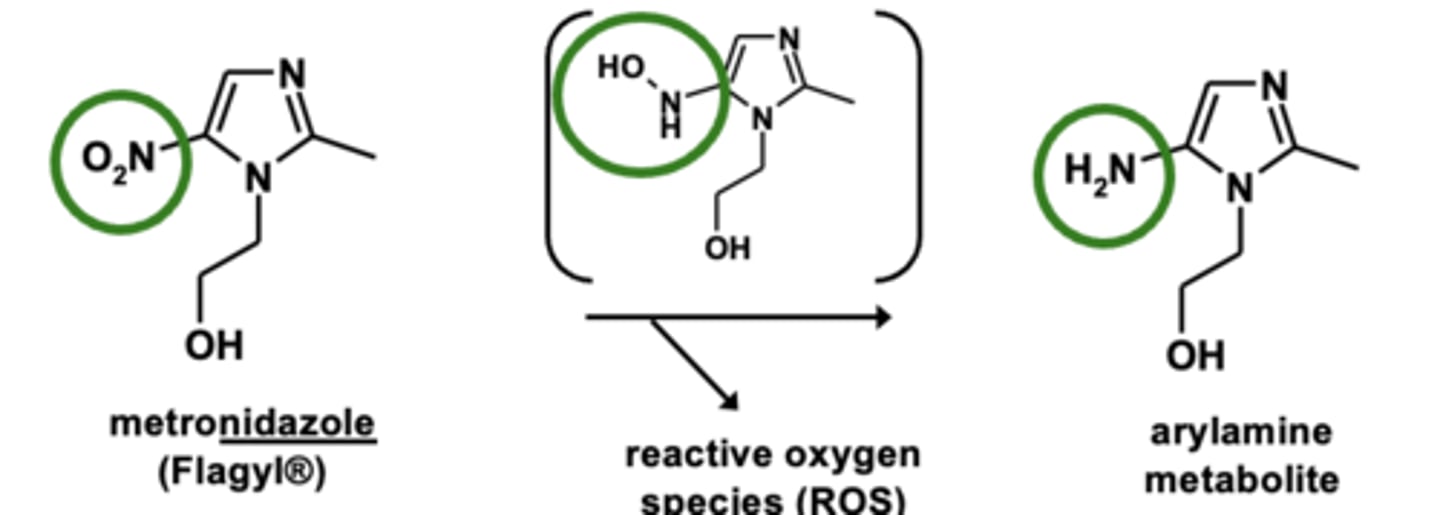
Example of nitro reductions: Metronidazole metabolism
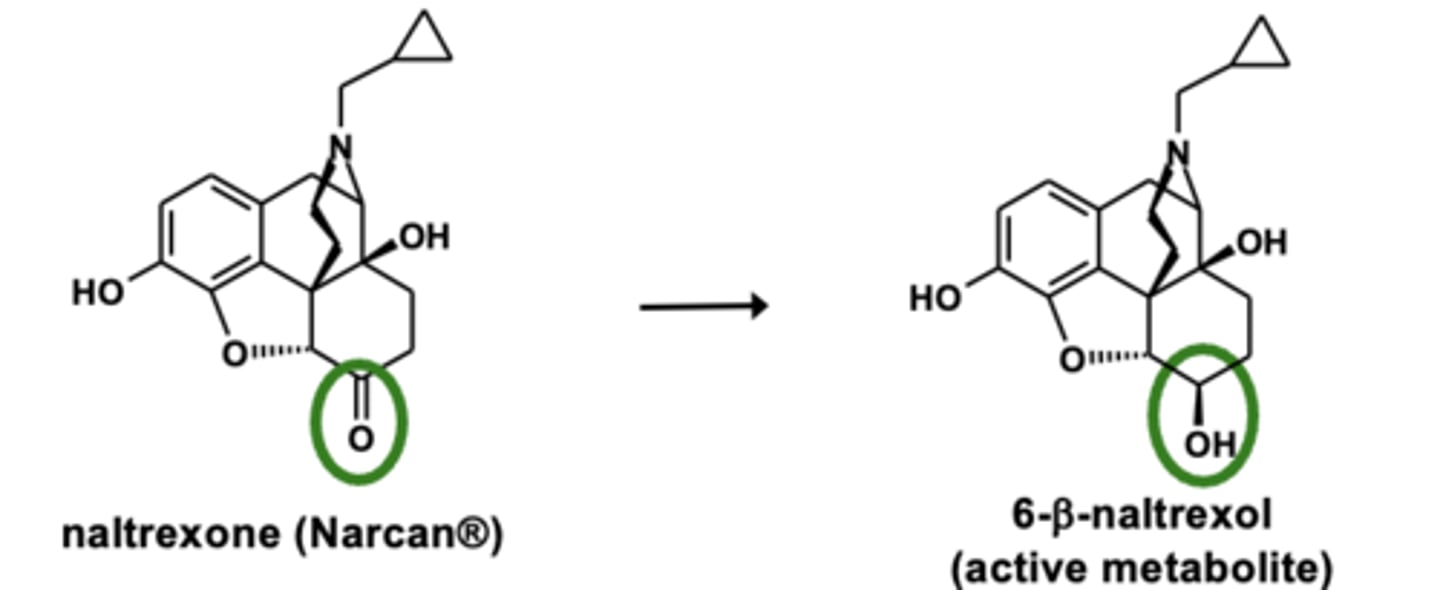
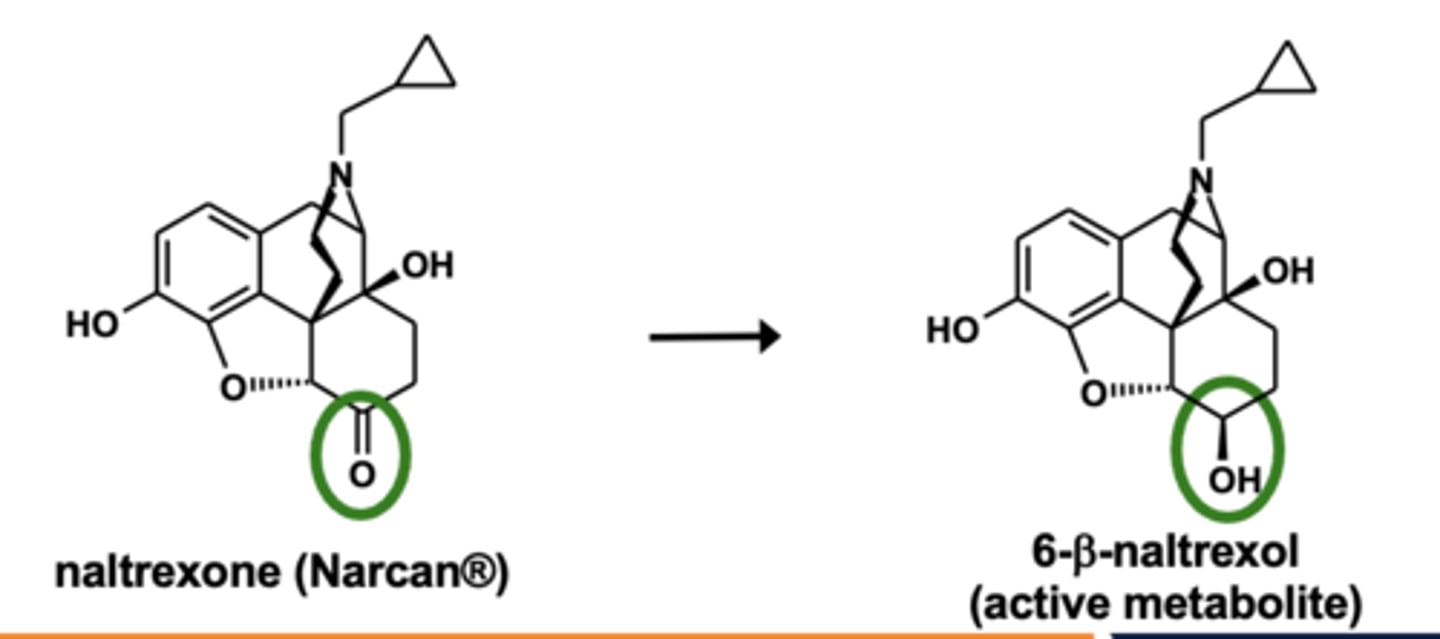
Example of carbonyl reductions: Naltrexone metabolism
What are the different hydrolyses reactions? (3)
- Ester hydrolyses
- Amide hydrolyses
- Carbamate hydrolyses
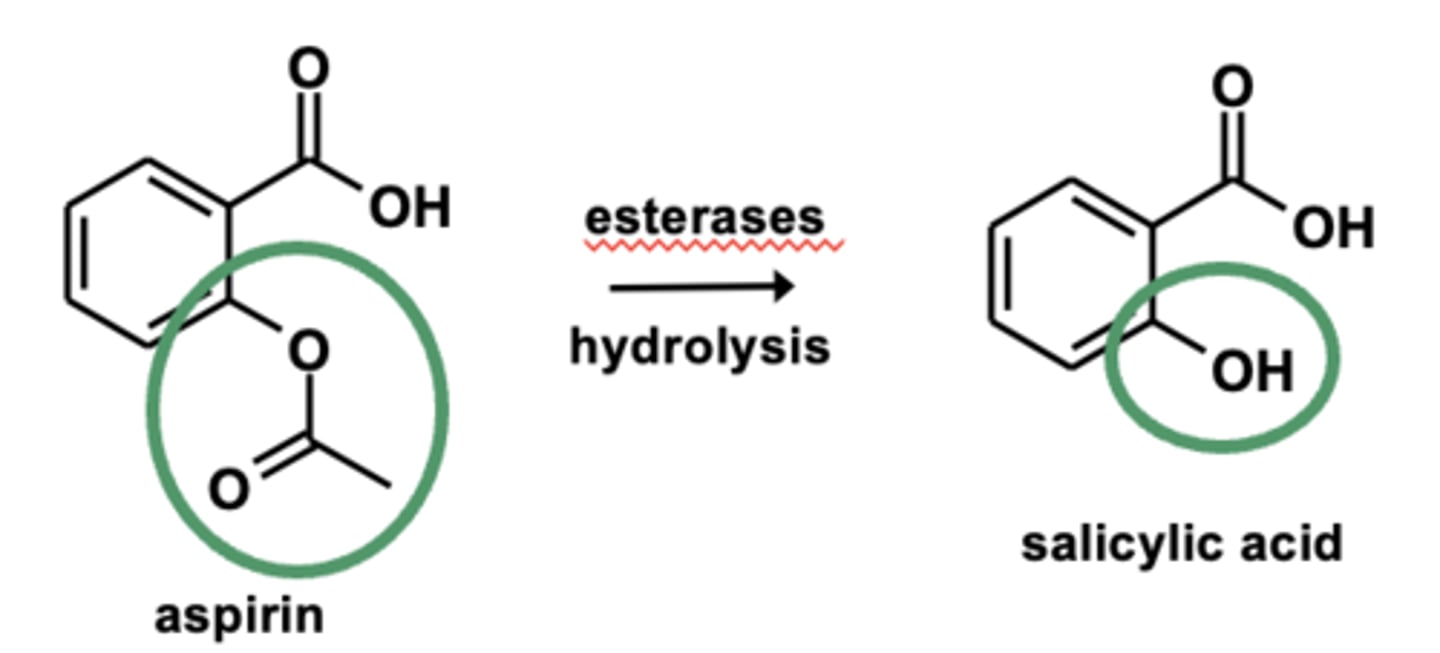
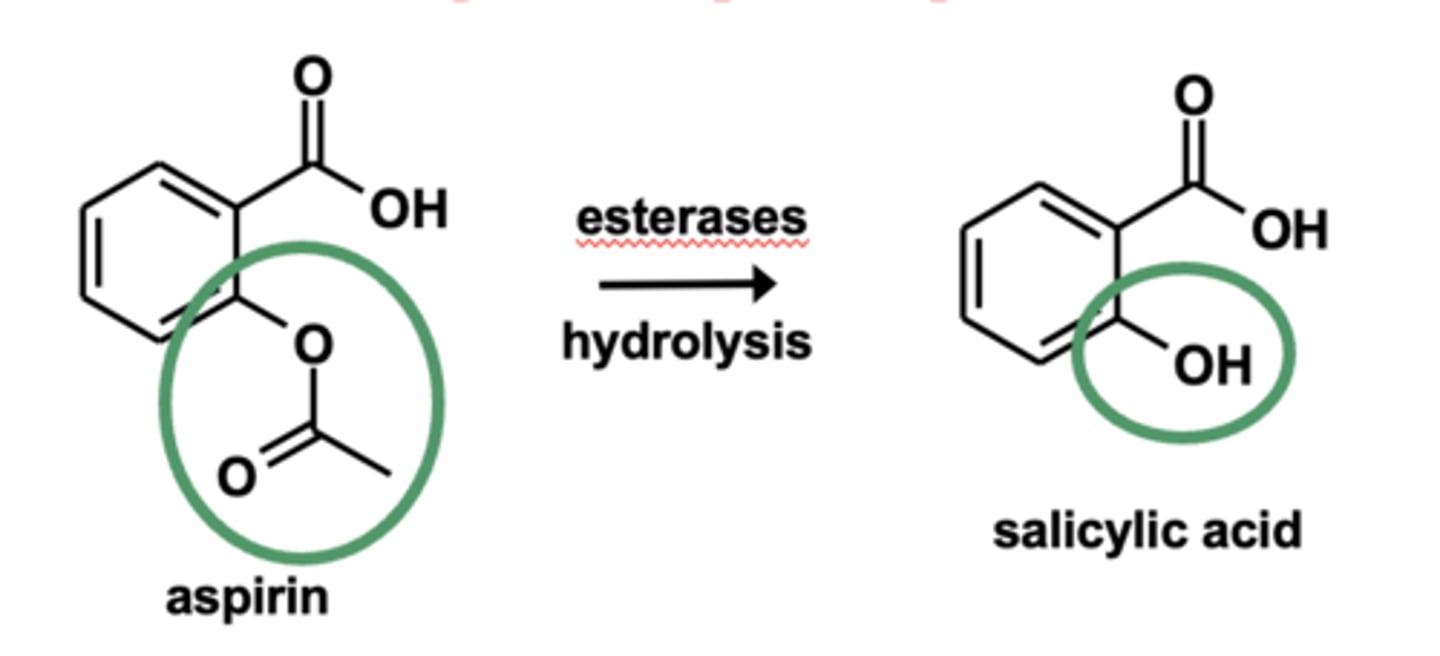
Example of ester hydrolyses: Metabolism of aspirin (ASA)
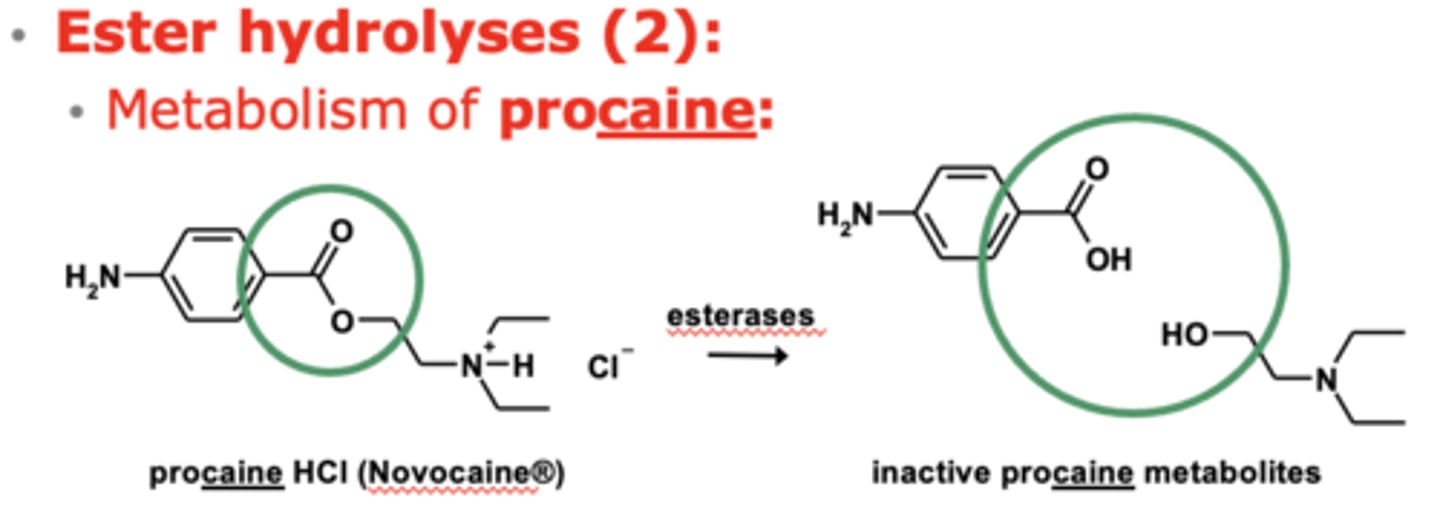
Example of ester hydrolyses: Metabolism of procaine
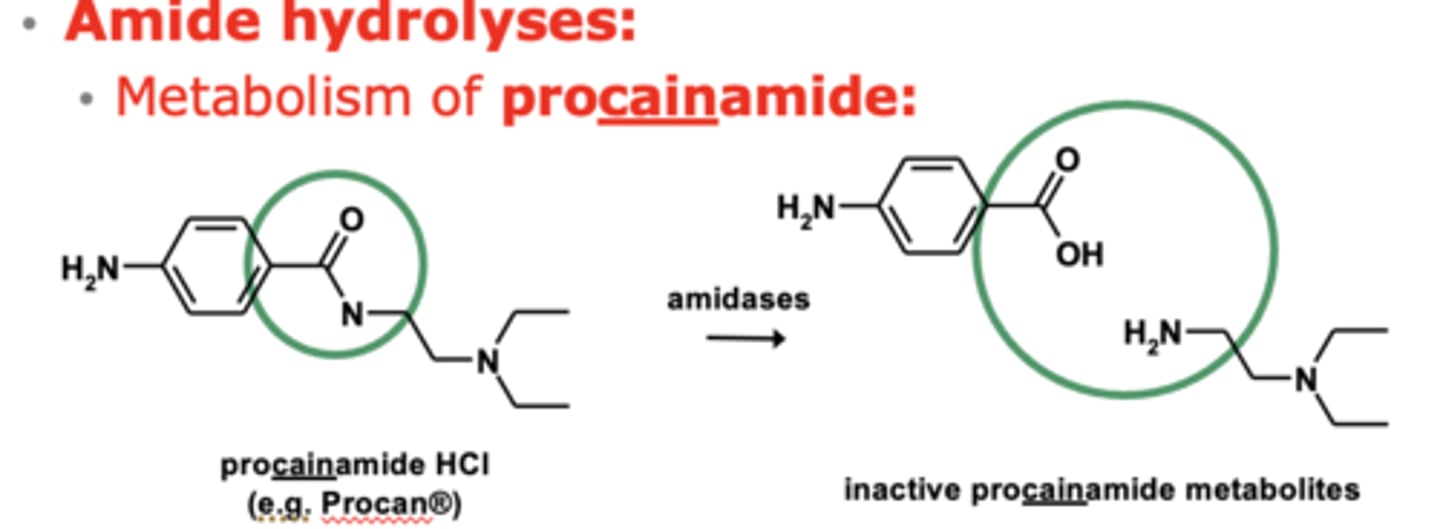
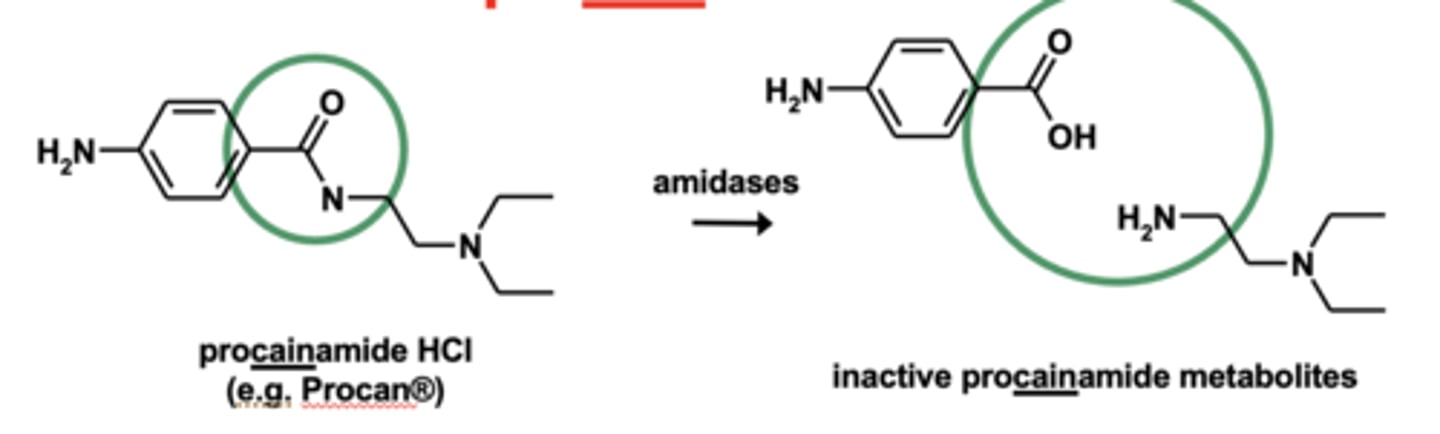
Example of amide hydrolyses: Metabolism of procainamide
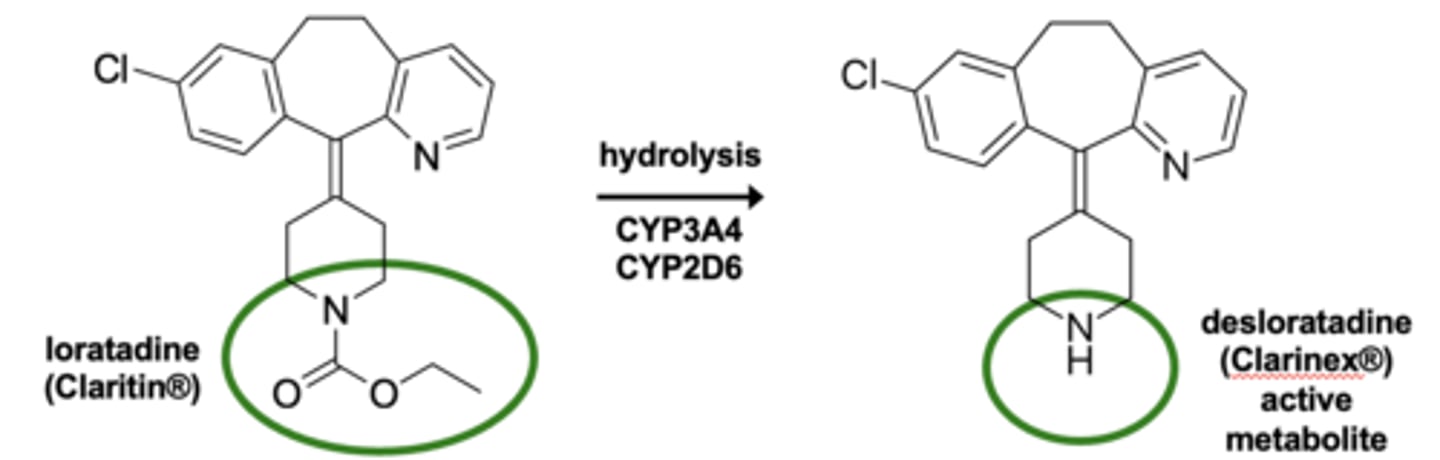
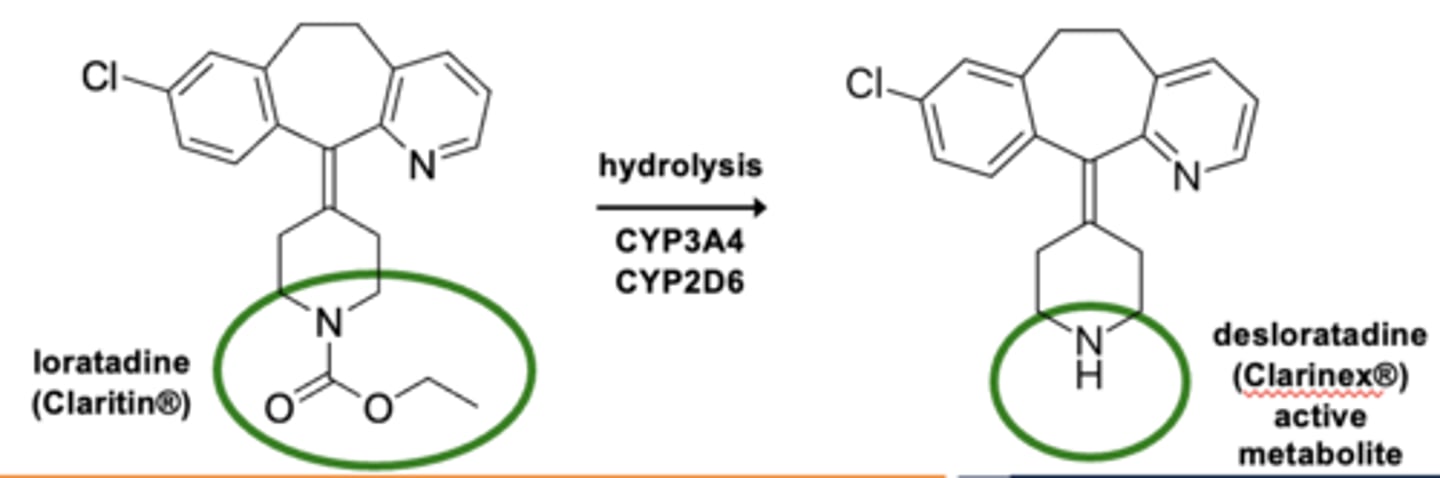
Example of carbamate hydrolyses: Metabolism of loratidine
Aromatic hydroxylation
Aliphatic hydroxylation
Epoxidation
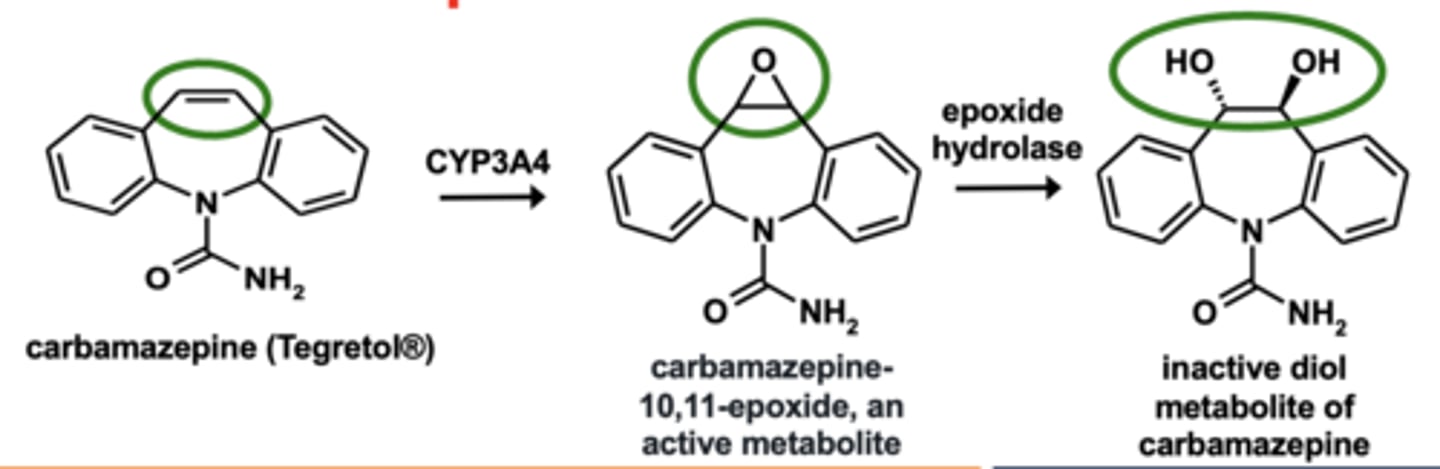
N-dealkylation
2/3 amines -> 1/2 amines
O-dealkylation
N-oxidation
S-oxidation
Deamination
Dehalogenation
Azo reductions
Nitro reductions
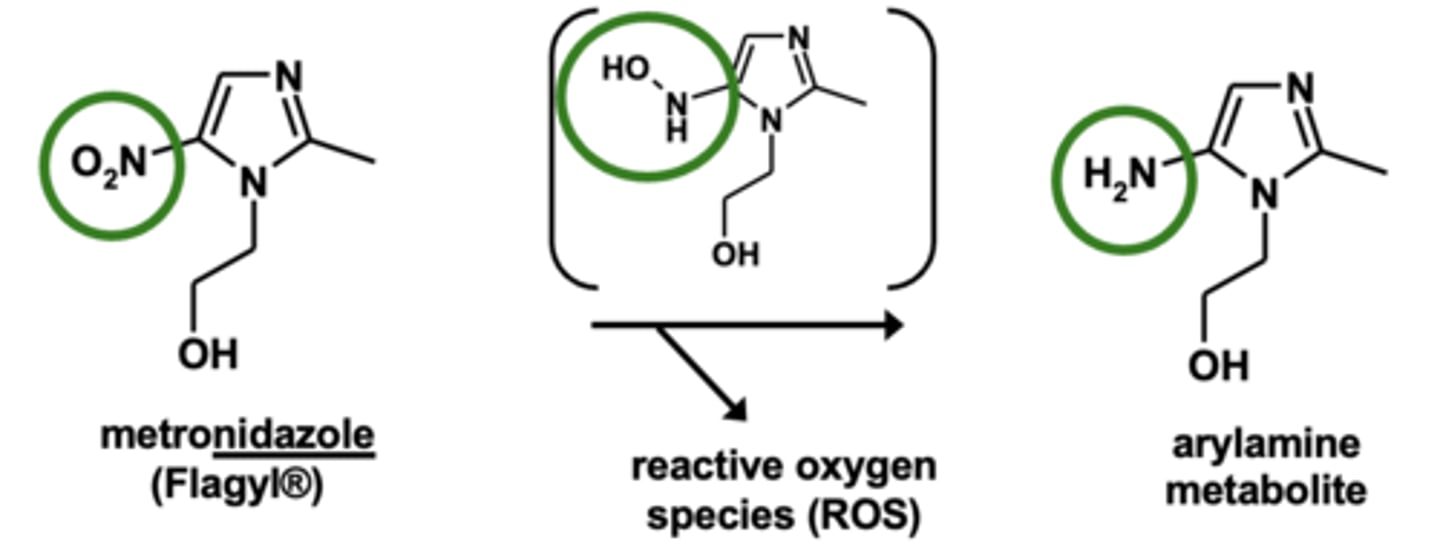
Carbonyl reductions
Ester hydrolyses
Amide hydrolyses
Carbamate hydrolyses