13A ERGs & Hereditary Disease
1/61
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
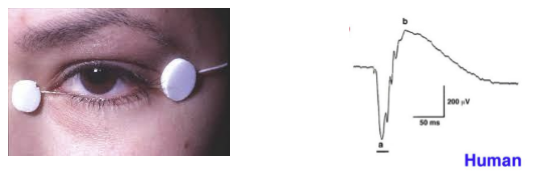
What does an electroretinogram (ERG) measure, and how is it recorded?
An ERG measures the summed electrical activity of the retina in response to light stimuli.
Recorded using a corneal or periocular electrode: historically a contact‑lens electrode with speculum; now commonly a thin wire along the lower eyelid connected to an amplifier.
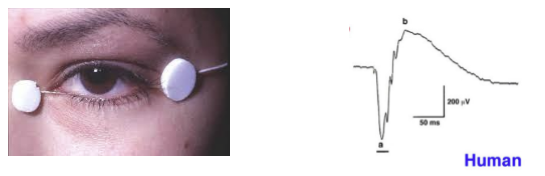
What creates the ERG waveform, and which components are clinically important?
Light stimulation produces a multi‑component retinal waveform, representing summed responses of photoreceptors, bipolar cells, and Müller cells.
Clinically, the two main components assessed are:
a‑wave → photoreceptor (mostly rods/cones) activity
b‑wave → ON bipolar + Müller cell activity
What is the origin and physiological basis of the ERG a‑wave?
The a‑wave reflects photoreceptor hyperpolarization due to a reduction of the dark current when light hits rods/cones.
→ Serves as a marker of photoreceptor functional integrity.
What cells generate the ERG b‑wave, and what does it represent?
The b‑wave is generated mainly by depolarization of ON‑bipolar cells, with contribution from Müller cells (K⁺ buffering).
→ Reflects post‑synaptic retinal response to photoreceptors, essentially photoreceptor output.
Why is the ERG b‑wave so large in a dark‑adapted retina despite having ON and OFF bipolar pathways?
In dark adaptation, ON‑bipolar cells are strongly activated when light removes glutamate inhibition.
OFF‑bipolar cells contribute little because their response requires increased glutamate (which light suppresses).
→ Responses do not cancel; the ON pathway dominates → large b‑wave.
Which retinal layers contribute most to the flash ERG, and why do inner retinal neurons (e.g., RGCs) contribute little?
Flash ERG is dominated by outer retinal activity:
a-wave: photoreceptors
b-wave: ON bipolar cells and Müller cells
Inner retinal neurons (e.g., RGCs) contribute minimally because they produce small, asynchronous action potentials that do not summate well into a measurable corneal signal.
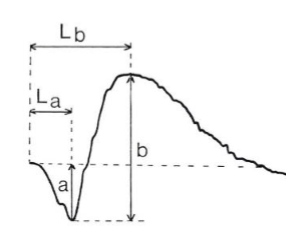
What ERG components are clinically measured, and what does “implicit time” represent?
Clinically measure:
a‑wave amplitude (baseline → trough)
b‑wave amplitude (a‑wave trough → b‑wave peak)
b‑wave implicit time = time from stimulus onset to b‑wave peak
→ Useful for detecting delays in retinal signal transmission (common in ischemia, retinal dystrophies).
What does a photopic (light‑adapted) ERG measure, and why?
Photopic ERG uses light‑adapted conditions, where rods are minimally active.
→ Primarily measures cone system responses, including cone‑driven bipolar cell activity.
Used for assessing cone dystrophies, macular disease, and photopic pathway dysfunction.
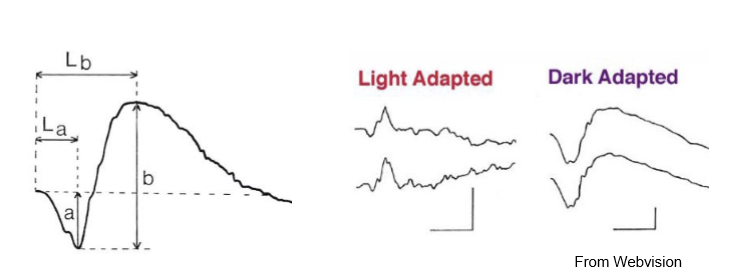
What does a scotopic (dark‑adapted) ERG measure, and why does dark adaptation matter?
Dark adaptation (~30 min) maximizes rod sensitivity and enables a mixed rod–cone response to a flash.
Scotopic ERG therefore assesses rod photoreceptor function and rod bipolar responses.
Useful in diagnosing rod‑predominant dystrophies (e.g., retinitis pigmentosa).
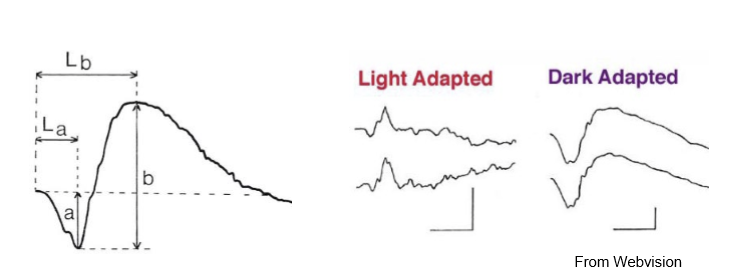
How do scotopic and photopic ERGs differ in physiology and clinical interpretation?
Scotopic: Rod‑dominant, high sensitivity, large b‑wave; tests night vision pathways.
Photopic: Cone‑dominant, faster implicit times; tests daylight & color‑vision pathways.
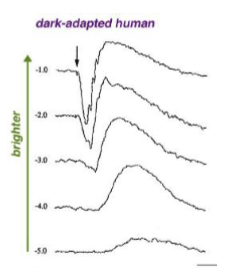
How can ERG testing isolate rod responses and why does light intensity matter?
Use a dim flash → stimulates rods only (cones less sensitive).
As flash intensity increases, both amplitude and implicit time systematically change (cones respond faster, rods saturate).
→ Useful for assessing rod pathway integrity under scotopic conditions.
How can ERG testing isolate cone responses?
Suppress rods by:
Light adaptation with a bright background, or
Using a 30‑Hz flicker stimulus (rods cannot follow high‑frequency flicker).
→ Yields a cone‑specific ERG, useful for diagnosing cone dystrophies and photopic pathway defects.
How can colored stimuli help differentiate rod and cone responses in the ERG?
Different spectral sensitivities:
Rods peak at ~498 nm (blue‑green).
Cones have S/M/L sensitivities across spectrum.
Using colored flashes allows preferential activation of specific photoreceptor classes.
→ Helps isolate S‑cone, cone‑mixed, or rod‑dominant responses.
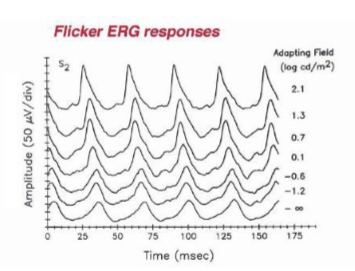
What characterizes the 30‑Hz flicker ERG, and which retinal pathway generates it?
30‑Hz flicker ERG = rapid light pulses presented to a light‑adapted subject.
Rods cannot follow >15 Hz, so the signal is generated entirely by cones.
Why are standardized ERG protocols used, and which organization sets these standards?
Standardization ensures inter‑clinic consistency in ERG results.
Protocols specify stimulus luminance, background luminance, flicker frequency, and recording conditions.
Set by the International Society for Clinical Electrophysiology of Vision (ISCEV).
What are the steps to prepare and un an ISCEV-standard ERG?
Dilate pupils and dark adapt the pt for 30 min
Put electrode on under dim red light
Use dim flash to get rod response
Use bright flash to get maximal (mixed) response
Increase background light to suppress rods, get cone response to single flash
Get flicker response (same flash as previous 2 responses, but flickering at 30 Hz)
What retinal layers are primarily assessed with standard full‑field ERGs in clinical practice?
Full‑field ERGs mainly assess the outer retina, in particular, issues that affect most or all of the outer retina.
What is RETeval?
A hand-held ERG device that can be used to aid in diagnosis/monitoring of retinal hereditary diseases in addition to Diabetic Retinopathy.

What does a full‑field ERG measure, and what are its main limitations?
Full‑field ERGs use flashes that illuminate the entire retina, producing signals that represent overall retinal activity (global rod and cone function).
Limitation: Poor at detecting focal or localized defects, especially macular‑only pathology.
What is a focal ERG and when is it used?
A focal ERG uses a small, localized flash (e.g., confined to the macula).
→ Designed to detect regional dysfunction, especially macular disease, which may be missed on full‑field ERG.

What is a multifocal ERG (mfERG), and how does it stimulate different retinal areas?
mfERG presents an array of ~240 hexagonal elements that flicker on/off in a pseudorandom pattern (about half illuminated at any time).
An algorithm extracts local ERG responses for each hexagon.
→ Provides a topographic map of retinal function across the posterior pole.
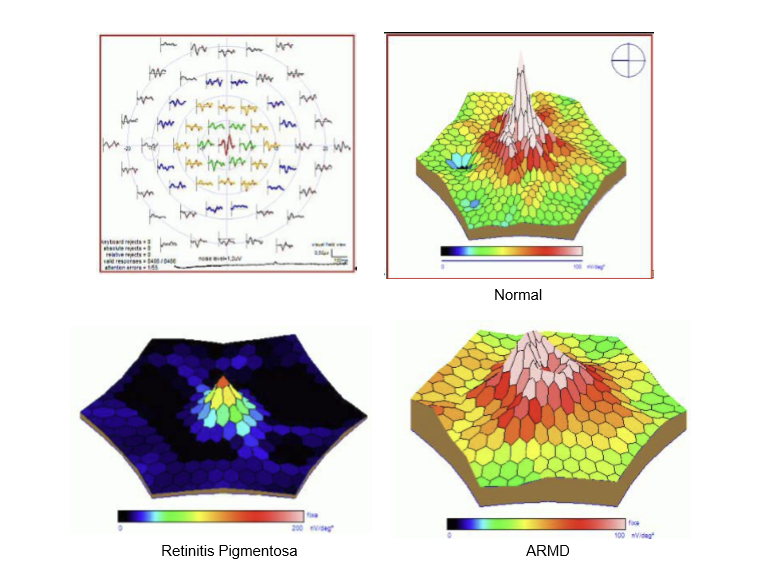
How does a multifocal ERG differ from full‑field ERG in clinical utility?
mfERG: Evaluates localized cone‑mediated macular function; excellent for detecting central or patchy retinal abnormalities.
Full‑field ERG: Measures global rod/cone function; best for generalized retinal diseases (e.g., retinitis pigmentosa).
→ They complement each other depending on whether the disease is diffuse or focal.
How are multifocal ERG (mfERG) waveforms generated, and how do they differ from full‑field ERG recordings?
mfERG waveforms are mathematically derived, not direct electrical recordings.
They come from pooling and subtracting hundreds of local responses obtained while the patient views flickering hexagonal stimuli.
→ Allows simultaneous sampling of many small retinal areas.
What is the major clinical advantage of the mfERG technique?
mfERG can identify localized functional deficits (especially macular and parafoveal dysfunction) that full‑field ERGs may miss.
→ Provides a topographic map of retinal function.
Why is patient fixation important during mfERG testing?
mfERG requires minimal eye movements so that each hexagon consistently stimulates its intended retinal region.
Poor fixation → mislocalization of responses and inaccurate functional mapping.
What feature of the mfERG stimulus pattern allows separation of responses from hundreds of retinal locations?
The hexagons flicker in a pseudorandom sequence, allowing response extraction through cross‑correlation algorithms.
→ Each retinal location has a unique temporal signature that can be isolated mathematically.
What are the primary clinical uses of ERG and EOG in hereditary retinal diseases?
ERG and EOG are used to diagnose and monitor hereditary retinal and choroidal diseases.
ERG (Electroretinogram): Best for detecting outer retinal dysfunction, especially photoreceptor diseases (e.g., retinitis pigmentosa).
EOG (Electrooculogram): Less commonly used but valuable for RPE function, especially in Best’s vitelliform dystrophy.
Why is the EOG particularly important in diagnosing Best’s disease?
Best’s disease affects the retinal pigment epithelium (RPE), and the EOG directly measures RPE function (light‑dark ratio, Arden ratio).
→ EOG is abnormal early, even before clinical signs appear.
→ ERG is usually normal, since photoreceptors are relatively preserved.
What is the inheritance pattern of Best’s disease, and how does this affect EOG findings?
Best’s disease is autosomal dominant, but with variable penetrance.
Even asymptomatic adult carriers have an abnormal EOG, allowing early detection in family screening.
What is the typical ERG finding in Best’s disease, and why?
ERG is often normal, because photoreceptor function is preserved early in the disease.
The primary defect is in the RPE, which the ERG does not measure well.
→ EOG abnormalities are the key diagnostic marker.
What is the underlying pathological hallmark of Best’s disease?
Accumulation of lipofuscin within the RPE and sub‑RPE space, especially in the fovea.
→ Results from RPE dysfunction, often due to BEST1 gene mutation (autosomal dominant).
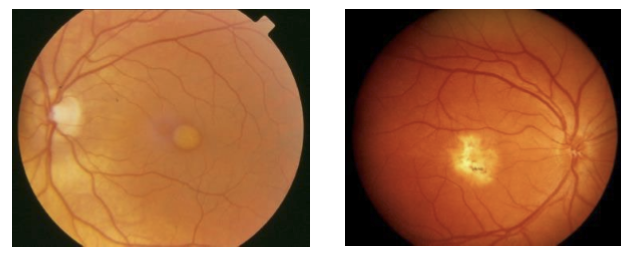
At what age do early clinical signs of Best’s disease appear, and what do they look like?
Initial pigment disturbances appear around ages 5–10.
The lesion evolves into the classic “egg‑yolk” (vitelliform) appearance in the macula.
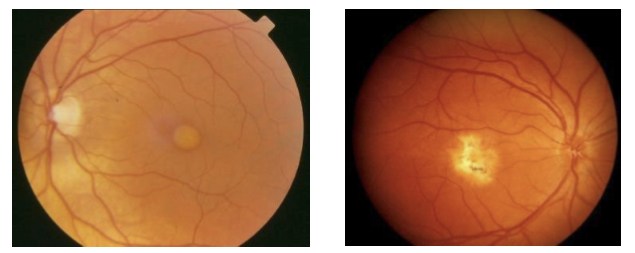
What visual symptoms and acuity changes occur during the vitelliform stage of Best’s disease?
Mild vision loss, typically 20/30 to 20/50 while the vitelliform (“egg‑yolk”) lesion is intact.
Photoreceptor function often remains relatively preserved at this stage.
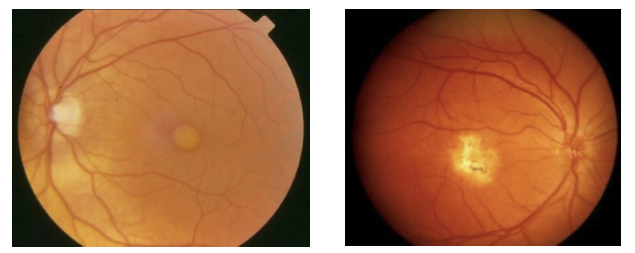
What happens as Best’s disease progresses beyond the vitelliform stage?
The lesion may rupture, leak, or hemorrhage, leading to:
Focal RPE degeneration
Choroidal atrophy
These changes can cause greater visual decline and scarring (atrophic stage).
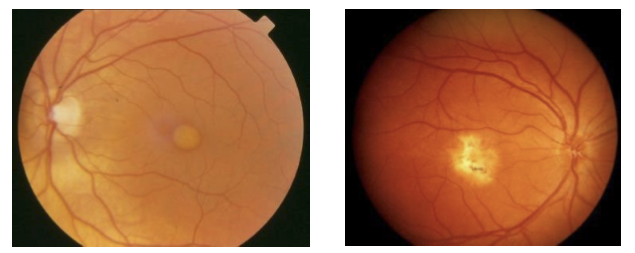
Why is the ERG useful in diagnosing early retinitis pigmentosa (RP)?
In early RP, the fundus may appear normal, so structural changes can be missed.
ERG detects functional abnormalities that precede visible fundus changes, especially in rod pathways.
→ Allows earlier diagnosis than ophthalmoscopy alone.
What are the earliest ERG abnormalities seen in RP?
Reduced scotopic (rod) ERG amplitudes (measured with blue light)
Prolonged scotopic implicit times
These reflect primary rod photoreceptor degeneration, characteristic of early RP.
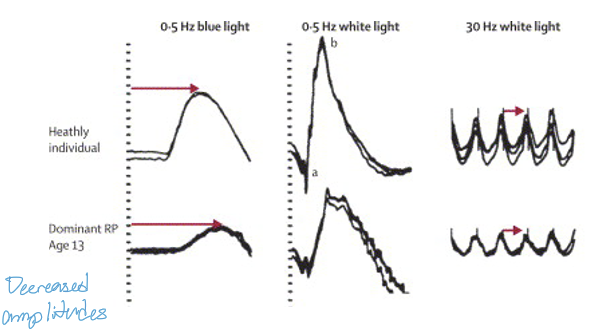
How does the ERG help monitor disease progression in RP?
Scotopic ERG amplitudes show a progressive decline as rods degenerate.
Later in the disease, photopic (cone) responses also diminish.
→ ERG provides an objective measure of retinal functional loss over time.
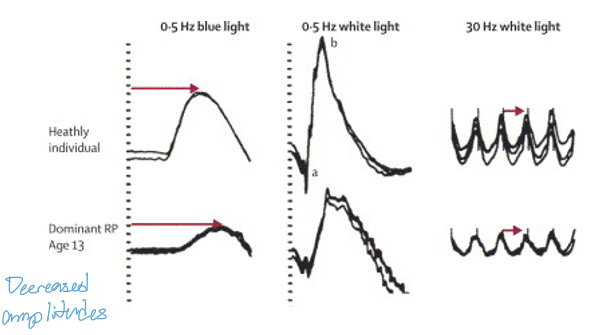
What ERG pattern is characteristic of advanced RP?
Near‑extinguished scotopic ERG (rod pathway loss) and reduced photopic ERG (secondary cone degeneration).
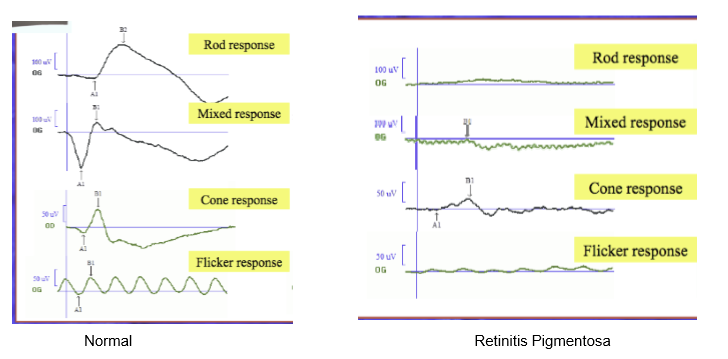
What is the ERG from a patient with late-stage RP?
Both cone and rod neurotransmission are severely affected.
What is the fundamental physiological defect in congenital stationary night blindness (CSNB)?
CSNB involves abnormal neurotransmission between photoreceptors and bipolar/horizontal cells.
→ Rod pathway signaling is disrupted, causing lifelong night blindness without progressive degeneration.
How does Type I (complete) CSNB present in terms of retinal pathway function?
Type I (“complete”) CSNB shows complete absence of rod pathway function.
→ ON‑bipolar cell signaling is specifically impaired.
ERG: Absent scotopic b‑wave with preserved a‑wave (classic electronegative ERG).
What are the genetic causes and inheritance patterns of Type I (complete) CSNB?
Two major patterns:
X‑linked CSNB: mutation in nyctalopin (NYX), a protein in ON‑bipolar cell dendrites.
Autosomal recessive CSNB: often due to mGluR6 (GRM6) mutations; more recently TRPM1 mutations identified.
All disrupt ON‑bipolar signaling.
Which molecular pathway is disrupted in both X‑linked and autosomal recessive complete CSNB?
Both forms cause a defect in mGluR6 and TRPM1 mutations.
Why do TRPM1 mutations cause complete CSNB?
TRPM1 is the ion channel required for ON‑bipolar cell depolarization in response to rod signals (mGluR6 pathway). Mutations block ON‑bipolar activation → rod pathway fails → night blindness.
Why does complete CSNB affect night vision but not usually cause progressive degeneration?
CSNB affects synaptic signaling, not photoreceptor survival. Rods are present but cannot communicate with ON‑bipolar cells.
Which gene accounts for ~50% of autosomal recessive complete CSNB?
TRPM1
What genetic defect causes Type 2 (incomplete) congenital stationary night blindness (CSNB2)?
Type 2 CSNB is X‑linked and caused by mutations in the gene encoding a photoreceptor-specific voltage‑gated Ca²⁺ channel (VGCC).
→ Without functional VGCCs, Ca²⁺ influx is reduced, preventing normal neurotransmitter release from photoreceptors.
How does the VGCC defect in CSNB2 affect photoreceptor signaling?
Photoreceptors require Ca²⁺ influx at their terminals to release glutamate.
A defective VGCC → no Ca²⁺ entry → no neurotransmitter release.
→ Both rod and cone pathways are impaired (rods more affected).
How does CSNB Type 2 (incomplete) differ functionally from Type 1 (complete)?
Type 1 (complete): ON‑bipolar signaling defect; rod pathway absent.
Type 2 (incomplete): Photoreceptor Ca²⁺ channel defect, causing rod > cone dysfunction.
Vision is less impaired in Type 2 because cones possess additional VGCC types that partially preserve function.
Why is vision generally better in CSNB Type 2 compared to Type 1?
Because cones have other functioning VGCCs, they retain partial neurotransmission.
→ Results in less severe visual impairment, especially under photopic conditions.
What is the primary symptom of congenital stationary night blindness (CSNB), and how does it differ from retinitis pigmentosa (RP)?
Primary symptom: Nyctalopia (night blindness).
Unlike RP, CSNB is non‑progressive (“stationary”).
→ Patients have lifelong, stable night vision deficits without photoreceptor degeneration.
What additional ocular findings may accompany CSNB?
Symptoms are variable and may include:
High myopia or hyperopia
Strabismus
Nystagmus (in some forms)
These reflect altered retinal signaling but the exact link to rod neurotransmission defects remains unclear.
How does visual acuity differ between Type 1 (complete) and Type 2 (incomplete) CSNB?
Visual acuity in CSNB can range from normal to severely reduced.
Type 2 (incomplete): Vision more often normal or mildly affected.
Type 1 (complete): More likely to show significant VA reduction due to complete rod‑ON pathway failure.
What is the inheritance pattern of X‑linked CSNB, and how does it influence clinical presentation?
X‑linked CSNB affects males predominantly, often with a strong family history.
Female carriers may have mild or no symptoms due to X‑inactivation mosaicism.
Why can the fundus appear normal in congenital stationary night blindness (CSNB)?
CSNB is a functional neurotransmission disorder, not a degenerative photoreceptor disease.
→ Structural retina appears normal on fundus exam, despite profound defects in synaptic signaling between photoreceptors and bipolar cells.
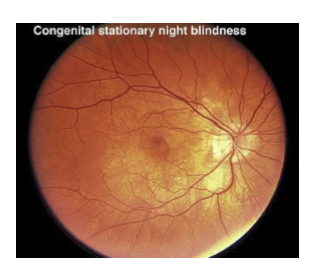
What aspect of retinal physiology is disrupted in CSNB, and how is this detected clinically?
CSNB involves impaired photoreceptor → bipolar cell neurotransmission.
→ Detected using full‑field ERG, which reveals characteristic abnormalities in the b‑wave (post‑photoreceptor response).
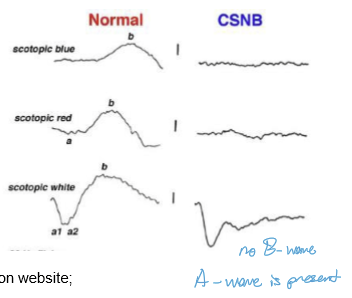
What classic ERG pattern is seen in CSNB, and why?
Bright‑flash ERG shows:
Normal a‑wave (photoreceptor hyperpolarization intact)
Absent or severely reduced b‑wave (ON‑bipolar pathway failure)
→ Produces the classic “electronegative ERG”, a hallmark of CSNB.
How does the ERG pattern help distinguish CSNB from degenerative retinal diseases like RP?
In CSNB: a‑wave normal, b‑wave absent → electronegative pattern, consistent with synaptic dysfunction.
In RP: reduced a‑wave and b‑wave, reflecting photoreceptor degeneration.
→ ERG helps differentiate functional vs. degenerative pathology.
What are the three principal functions of the retina?
Transduce visual images into electrochemical signals (phototransduction).
Perform initial processing of visual information (contrast, temporal filtering, center‑surround processing).
Convert the processed signal into outputs (ganglion cell action potentials) that can be interpreted by the brain.
What major concepts fall under “function of the outer retina,” and what structures are involved?
Outer retina (photoreceptors → bipolar cells → first synapse) underlies:
Color vision (cone types & circuitry)
Dark and light adaptation (photoreceptor and RPE mechanisms)
Detection thresholds (rod sensitivity, noise reduction)
Electrophysiology of the outer retina (EOG for RPE, ERG a‑ & b‑wave origins)
Which clinical electrophysiology tests assess outer retinal function, and what structures do they correspond to?
ERG: Photoreceptors, bipolar cells, Müller cells (a‑wave = PR; b‑wave = ON‑bipolar).
EOG: RPE function (Arden ratio).
→ Together assess outer retinal physiology and early dysfunction.
Why is understanding outer retinal physiology clinically relevant?
Many retinal diseases primarily affect the outer retina, including:
Retinitis pigmentosa
Cone/rod dystrophies
Best’s disease (RPE dysfunction)
CSNB (PR→bipolar synaptic transmission defects)
→ Physiology explains ERG/EOG findings and early functional changes.