Lecture 17: Disaccharides and Polysaccharides
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
examples of disaccharides
sucrose and lactose
formation of disaccharides and polysaccharides
condensation reaction via glycosidic linkage aka ether bond
lactose formed by
galactose and glucose where -OH group form one sugar and the -OH group from other sugar contribute to bond formation and water is released
Sugars are either
aldoses or ketoses, they contain carbonyl group
reducing end
in aldoses the aldehyde can be oxidized to an acid, ketose is also a reducing sugar
reducing ends in disaccharide
can only have 0 or 1 reducing end
ex: lactose has one reducing end
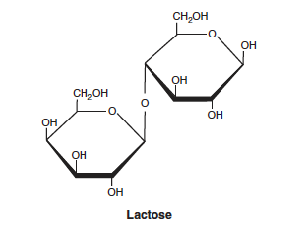
reducing end depends on
which -OH groups are involved in the condensation reactions
Whenever condensation occurs:
one of the sugars always donates the hydroxyl group bound to the anomeric carbon
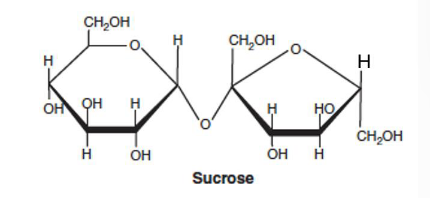
Sucrose
a non reducing sugar bc it can’t polymerize further
non reducing sugar
when the condensation between two sugars involves hydroxyl groups bound the anomeric carbons of both sugars, then there is no more reactive anomeric carbons
Hydrolysis occurs thru
enzymes and requires water
Opposite of polymerization/condensation
hydrolysis
Water is added across the glycosidic bond
One side gets H and the other side gets OH → each sugar gets back its hydroxyl group
hydrolysis restores
two glucose molecules each with an anomeric carbon again
sugar on the left
is always the one that donated the hydroxyl group of the anomeric carbon
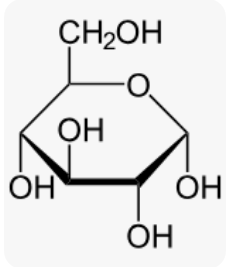
glucose
If a sugar has its anomeric carbon locked in the glycosidic linkage
you must specify whether it is alpha or beta
alpha and beta
tell you the direction of the OH on the anomeric carbon in a cyclic sugar
Beta
the anomeric OH and the reference CH2OH are on the same side
Alpha
the anomeric OH and the reference CH2OH are on opposite sides
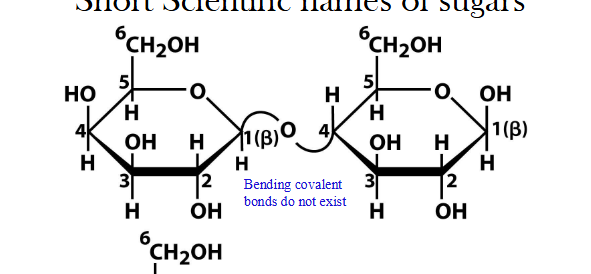
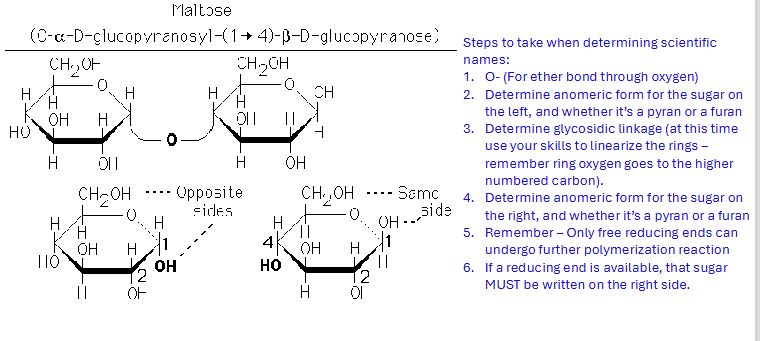
Figuring out the right sugar carbon number
find the anomeric carbon, “cut” the ring so each carbon gets its oxygen back, then number, and find which carbon the glycosidic linkage is
Right sugar conformation rule
If the sugar on the right has a free anomeric carbon, no need to include its alpha/beta conformation in the linkage name
naming: sugar on the left will always be
1-something or 2-something because by convention it donated its anomeric hydroxyl group
naming: sugar on the right
students must use ring-linearization and numbering to determine which carbon donated its hydroxy
Systematic Nomenclature
Polysaccharides
Indefinite length, strings of many identical monomers or dimer repeats (homopolymers or heteropolymers)
oligosaccharides
shorter discrete-length polymers
Examples of polysaccharides
cellulose, starch, glycogen, and chitin
Starch
Used to store glucose in plants
Glycogen
Used to store glucose in animals
Glycogen and starch linkages
Both have alpha(1→4) linkage
left handed helix
Digestibility of glycogen and starch
can be digested by animals
Cellulose
occurs as sheets that has beta(1→4) linkage and provides structural support to plants but cannot be digested by animals
animals cant digest cellulose bc
animals have enzymes that break alpha 1→ 4 not enzymes that break beta 1→ 4
Branching in starch and glycogen
alpha(1→6) linkage which is important bc more branching means more non reducing ends and that helps with rapid mobilization
cellulose has no branching
it has intra chain hydrogen bonds and inter chain hydrogen bonds → very sturdy
homopolymers of glucose
starch, glycogen, and cellulose
chitin
homopolymer derivative of glucose and is used for the exoskeleton of bugs
chitin linkage
beta 1→ 4 glycosidic linkage
Peptidoglycan
consists of small protein segments that are bound to sugars and is present in bacteria
gram positive
have a cell wall made of peptidoglycan
gram negative
have a very thin peptidoglycan layer and an outer membrane