BIS 2A EXAM 2
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
reducing agent
the one with electrons in the reactant side giving electrons to the product
the compound that gets reduced
the product that gains electrons
redox
high electronegativity = more likely to be reduced and low electronegativity = tendency to be oxidized
if delta E is pos
delta g is neg and spontaneous and exergonic
NADH
carries 2 electrons and 1 proton plus one associated proton, the form when NAD+ is reduced
Metabolism
the addition of any chemical reaction that is happening in your body
the precursor that is common in the metabolism in all organisms
glucose
allosteric enzyme
other shape (non-competitive inhibition)
Statins
chemicals which are completive inhibitors, they’re going for the same active site that you have in your enzymes in the formation of cholesterol
ends in -ase
an enzyme
cellular respiration
cytoplasm: glycolysis,
Mitochondria Matrix: Citric acid cycle
Cristae and interspace membrane: oxidative phosphorylation
glycolysis
everything is going to happen in the cytoplasm
for the first part of obtention of energy
you don’t need a mitochondria
every time you consume food it’s broken apart into glucose and then part of that glucose will build into ATP
because you can’t storage ATP since it will explode
glucose transporters
glucose has an electrical charge so you need transporters one that is called Facilitated diffusion( not using energy)- glucose transporters (GLUTs) through uniporters and. another through symporters Sodium glucose link transporters (SGLTs) which moves glucose inside cells and goes against the gradient concentration which requieres energy
thioester bond (high-energy covalent bond)
through cysteine (sulfur and hydrogen) and histidine (carbon and nitrogen ring) will be formed nitrogen and hydrogen will form a hydrogen bond and a substrate will attach to sulfur creating the thioester bond the formation is going to be an endergonic reaction
Enzymes just change for the transformation of the substrate
once that happens enzyme goes back to its original shape and you can reduce that enzyme
forming pools
NAD pool is going to pick up electrons that are produced from that fragmentation of your food and these electrons are going to reduce this molecule which will transform into NADH (you are moving energy)
pyruvate
molecule with only 3 carbons so when you produce it through glycolysis you split the glycogen molecule into 2
if it has pyruvate and no mitochondria
it can use electrons to produce lactate (a waste of that chemical reaction)
lactic acid
through lactate bacteria growing in a system with no oxygen and using sugar as their nutrient source, (anaerobic)
fermentation
happens in the absence of oxygen (anaerobic) - alcohol and CO2 the goal of fermentation is to produce ATP -
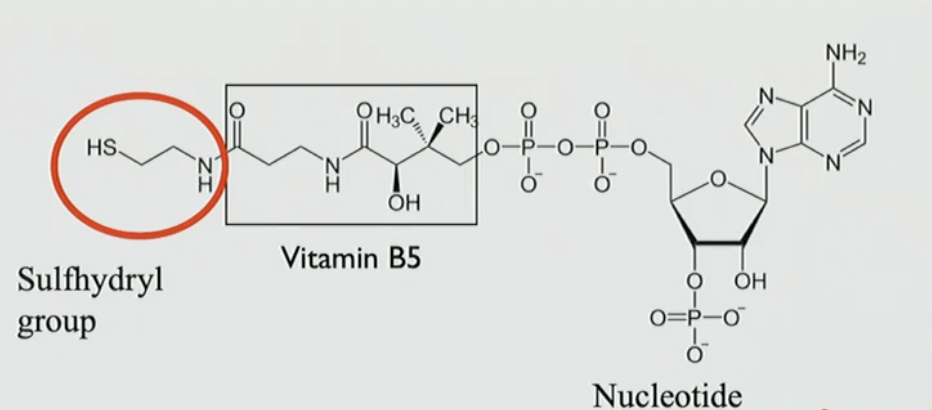
coenzyme -A
sulfhydryl group (HS-N H) vitamin B5 and a nucleotide
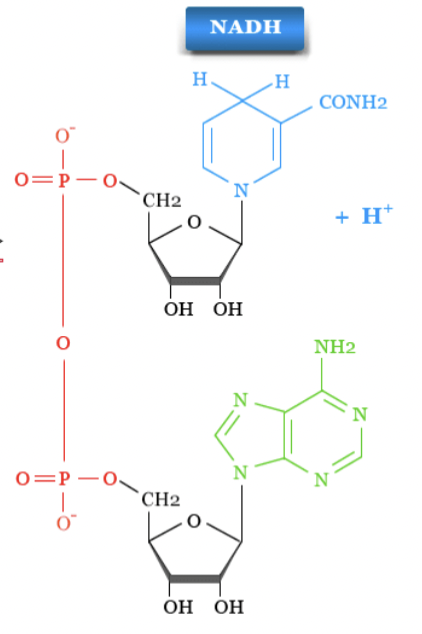
NADH
has 2 electron (H and H sticking out of nicotinamide ring) and +H (proton)
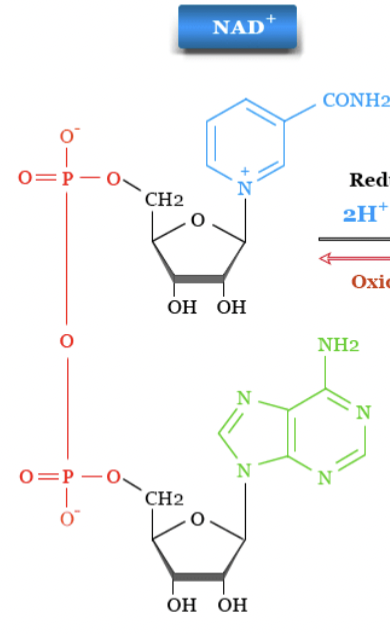
NAD+
has + charge on N ( nicotinamide ring) has no H sticking out go it
decarboxylation
taking out one carbon (carbon dioxide) because once you don't have more energy to extract from a molecule that is going to become waste or a byproduct
inside the inner membrane - matrix
the place where the citric acid cycle happens (Krebs and tricarboxylic acid cycle), receptor is oxaloacetate which has 4 carbons, acetyl givens 2 carbons to another molecule making it citric acid which has 6 carbons
The Cytochrome C reductase complex helps
pump protons and because you are pushing these protons in the intermembrane space (not outsdide the mitochondria instead in between the inner and outer membrane) and as a result your re creating a gradient of concentration
Electron Transport Chain
is aerobic we need oxygen to have the last receptor for electrons to form water and to start with that gradient of concentration of protons
ATP synthase complex
has a specific channel for portions to exit, the protons are going to use that gradient concentration fore every proton that you’re putting in back is going to propel the energy to transform ADP and ATP and then you’re going to have one ATP channel protein that is going to permit the exit of that molecule
the flow of energy
Glucose —— NADH/FADH2———- ETC —— chemiosmosis —— ATP
Irreversible reactions
are those with a large negative delta G meaning that they’re highly energetically favorable and proceed overwhelmingly in the forward direction (products) under cellular conditions
allosteric part of an enzyme
any other place for bonding for different molecules of the active site will be an allosteric site
if you have low ATP the reaction will happen faster and if you have high ATP the reaction will be slower
because you don’t want to create unnecessary energy because you don’t have somewhere to put it
difference between fermentation and respiration
when talking about the Krebs cycle and ETC we need a last receptor for electrons which will be oxygen for us but in fermentation (bacteria archaea or fungus) they’re going to be able to produce lactic acid or ethanol as waste doesn’t use oxygen as the last receptor for electrons
chlorophyll a has
2 different wavelengths where it is going to be effective (430 and 680)
Bacteria have all the cytochromes in the membrane
they’re going to have a space between the membrane and the cell wall to prepare the gradient of concentration for protons and from there going from glycolysis to the electron transport chain to chemiosmosis and to produce more energy
oxidative phosphorylation
is not exclusive for eukaryotic organisms
the only primary pigment that we have
chlorophyll A (any other pigment is considered secondary), they’re hydrophobic because they have a long chain of lipids
anoxygenic photosynthesis
produce photosynthesis but they’re not going to release oxygen into the atmosphere, they use light to transform matter but don’t produce oxygen
cellular respiration and glycolysis is for
every organism including the photosynthetic ones
photosynthetic infrastructure of plants
presence of chloroplast which has a double membrane and their own DNA, contains stroma which is the empty spaces in-between the stacks (which will be full of water, salts, and ions ) has the thylakoid (the individual disks) which contain the pigments for photosynthesis (a stack is a granum which are all going to be interconnected)
stomata
valve-like pores found in leaves which allow CO2 in for photosynthesis and releases oxygen, but when it opens evaporation of water is happening (dehydration)
photosynthesis only happens in the
visible spectrum everything that you can see which they use this light to excite electrons through the chloroplasts
NADP+ (P is for phosphate)
is an electron carrier, requires a terminal electron acceptor
NADPH (P is for phosphate)
take electrons to build bigger molecules using CO2
photosystems
have a reaction center which has primary pigments ( two chlorophyll A) around that you will have an antenna complex which is a group of different pigments that can have a variation depending on the plant (secondary pigments) then it concentrates a particular light that is going to have enough energy to generate the movement of electrons (you need a bunch of helpers that can work with diff wavelengths) once they have that energy they pas to chlorophyll A which will be in charge
lumen of your thylakoid
is where that split of water is going to form a boron gradient
ATP runs the cell in virtually every aspect of metabolism
if this cycle is not fast enough which could cause a deficit in ATP and a surplus of NADPH which means you can’t transfer electrons because the only way to do it is using NADPH+ and you don’t have any more so you fix using the Z-scheme
Z-scheme
is linear because it’s not coming back, it’s starting and ending, is light-dependent, they require photosystem 1 and 2, water is necessary (because electrons are coming from there) and produces ATP, oxygen and NADPH
Cyclic photophosphorylation
happens only in photosystem 1 is an anaerobic process meaning it doesn’t need or produce oxygen and doesn’t produce NADPH, instead goes back in the electrons transport chain where they can travel using cytochromes and int eh process produce more ATP doesn’t use new electrons just is re-excited linear and cyclic process happens together
light independent reactions (Calvin cycle)
do not happen in the dark they just don’t need light to work happens in the stroma
light dependent reactions happen in
the thylakoid
G3P
produces glucose and it can have other derivatives such as fatty acid and amino acid
cellular respiration happens in
the mitochondria
carbon and oxygen in glucose is going to come
from CO2 part of that oxygen from CO2 is going to form water and other part of water product is going to come from water reactant. O2 product comes from water reactant
an irreversible reaction from G6P
is going to produce pentose phosphate pathway which is another opportunity to form CO2, NADPH and form nucleotides which are needed to create DNA,RNA and ATP the formation of nucleotides following procedures aren’t irreversible done when you have enough ATP (so it goes through the PPP - pentose phosphate pathway instead of glycolysis)
NADPH donate electrons in anabolic pathways
form fatty acid synthesis, steroid hormone synthesis, and cholesterol synthesis which is important for your liver, kidneys, gonads(testis,ovaries) and mammary glands
glycolysis precursors
glucose-6-P, glyceraldehyde 3-phophate, phosphoenolpyruvate, fructose -6phosphate, 3-phosphoglycerate, pyruvate
pentose phosphate pathway precursors
ribose-5phosphate, erythorose-4 phosphate
linking glycolysis and CAC precursor
Acetyl-Co-A
CAC (citric acid cycle ) precursor
a-Ketoglutarate, oxaloacetate, succinyl-CoA
DNA
double helix (two strands) and has 4 bases (cytosine, guanine, adenine, and thymine) and is going to use the sugar deoxyribose
RNA
has only one strand and four bases (cytosine, guanine, adenine, and uracil with nitrogen ) and the sugar used is going to be ribose
pyrimidine ring
have only one ring in sugars (cytosine, uracil, and thymine)
purine ring
two rings together (guanine and adenine)