BIOC17 Lab Exam
1/123
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
124 Terms
What is the stage micrometer used for?
calibrating the ocular micrometer at each objective
increments on the stage micrometer
major divisions: 100 μm
minor divisions: 10 μm
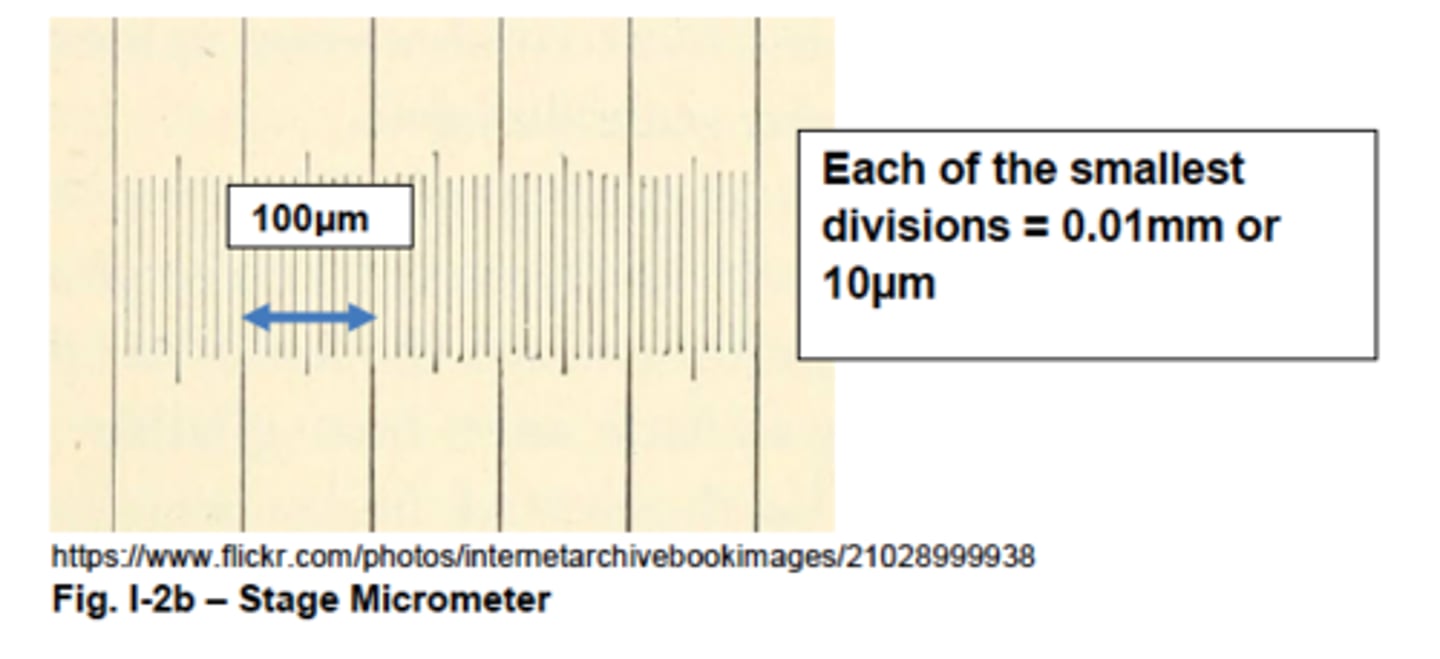
process of calibrating ocular micrometer
1. find two incidences where the gradations on the ocular micrometer match up w. the ones on the stage micrometer
2. make calculations of the value of one ocular unit (minor division) for both incidences
3. if the values are different, take the mathematical mean
difference between simple staining and negative staining
simple staining: staining the microbe; uses basic stain
negative staining: staining the background; uses acidic stain
basic makeup of a stain
solutions with a solvent and a coloured molecule
- solvent tends to be water or ethanol
- coloured molecule tends to be chromogen
What is a chromogen? (2 components)
coloured component of a stain consisting of:
1. auxochrome: charged portion of the molecule to ionically/covalently bind cell portion or background to be dyed
2. chromophore: portion of the chromogen giving the stain its colour
DEF basic stain
stain w. a (+) charged auxochrome in its chromogen; attract bacterial cell b/c cell wall (-) charged
examples of commonly used basic stains (3)
1. crystal violet
2. safranin
3. methylene blue
example of commonly used acidic stains (2)
1. Nigrosine
2. Congo Red
DEF acidic stain
stain w. a (-) charged auxochrome in its chromogen; repels (-) charged bacterial cell walls so that the background is stained, instead
purpose of heat-fixation (3)
1. kills the bacterial cells
2. coagulates the cytoplasmic proteins to make more visible
3. helps cells adhere to the slide to be stained
consequences of heat-fixation
- can distort cell
- can cause cell shrinkage
- not usable if cells need to be alive during observation
For what procedures are heat-fixing prohibited? (3)
1. negative stain
- don't want distorted/shrunken cells
2. capsule stain
- don't want shrunken cells that may appear to have halo
3. hanging drop
- don't want dead cells
methods of determining bacterial motility (3)
1. hanging drop
2. flagellar stain technique
3. motility test
difference between true motility and brownian motion
true motility: single flagellum/flagella to enable directional motion
brownian motion: vibrating on the spot due to bombardment of water molecules in their aqueous environments
What type of slide is used for hanging drop?
depression slide
microbes displaying true motility vs. Brownian motion
motile: Escherichia coli
Brownian (non-motile): Staphylococcus epidermidis
Is the 100X objective used for the hanging drop?
no; if can't focus on the 100X, the drop will no longer be suspended
What stain is used for flagella staining? Why?
Ryu stain mordant to stain in multiple layers to flagella so that the flagella appears thicker to be visible thru bright-field microscopy
types of flagellar arrangements:
monotrichous
amphitrichous
lophotrichous
petritrichous
monotrichous: single flagellum at one end of the cell
amphitrichous: flagella at both ends of the bacteria
lophotrichous: tufts of flagella at the end of the cell
petritrichous: flagella emerging from all over the cell surface
What does flagella staining tell us about the bacteria's motility?
if flagellum present, the organism is motile
What is Wescodyne?
iodine-based anti-germicidal agent for disposal of solid and liquid waste including used tips
What is a bright-field light microscope?
specimen is dark and contrasted by surrounding bright viewing field; image produced by bright-field light microscopy is from light transmitting thru the specimen
objectives on a bright-field light microscope (4)
1. scanning objective lens (4X)
2. low power objective lens (10X)
3. high-dry objective lens (40X)
4. oil immersion lens (100X)
Which objectives can the coarse adjustment knob be used on?
scanning (4X)
low power (10X)
Which objectives should the fine adjustment knob be used on?
high-dry (40 X)
oil immersion (100X)
total magnification of specimen
total magnification = magnification by the objective lens × magnification by the ocular lens
limit of resolution of a microscope
minimum distance between two points needed for the microscope to discern as separate entities
calculation of limit of resolution
D = (λ) ÷ (NA condenser + NA objective)
- D: limit of resolution (nm)
- λ: microscope's wavelength of light (nm)
- NA: numerical aperture
DEF numerical aperture
measure of light-gathering ability of a lens, which is dependent on the index of refraction (bending of light as it moves thru a material)
Is a larger or smaller numerical aperture better for a smaller limit of resolution?
larger
index of refraction for air
1
index of refraction for oil
1.52
index of refraction for glass
closer to 1.52 than 1
Why is oil immersion used for the 100X objective? (2)
1. to prevent scratching the objective on the slide
2. to allow greater resolution power (lower limit of resolution) b/c the index of refraction btw. glass and oil is more similar than btw. air and oil, thereby increasing numeric aperture
role of the condenser on a microscope
concentrates incandescent light emitted from microscope's light source on the bottom
role of the diaphragm on a microscope
situated in the condenser to control the focusing of light onto the specimen, where a smaller amount of light focused thru is more desirable for low-contrast, unstained specimen
importance of the gram stain in bacteriology
most important differential stain and typically the first one used on specimen brought into lab
sources of gram variability (mix of pink and purple cells) (4)
1. cultures older than 24 hrs b/c G+ cell wall starts losing integrity
2. bacterial smears too thick so stain/decolourizing agent has difficulty penetrating
3. bacteria used form endospores and can't easily take up primary dye
4. bacteria used had reduced thickness of peptidoglycan during growth
gram stain procedure (9 steps)
1. Crystal Violet stain
2. rinse with dH₂O
3. Gram's Iodine stain
4. rinse with dH₂O
5. 95% ethanol for brief while
6. rinse with dH₂O
7. safranin stain
8. rinse with dH₂O
9. observe under oil immersion
purpose of Crystal Violet stain in gram staining
primary stain: penetrates both G+ and G- bacteria to produce violet colour
purpose of Gram's Iodine stain in gram staining
mordant: complexes with Crystal Violet in cytoplasm and cell wall; G+ has higher affinity for Crystal Violet after applying gram's iodine
purpose of 95% ethanol in gram staining
decolourizer: dehydrates the peptidoglycan layer by tightening/shrinking to trap CV-I in G+ (washed out of G- b/c outer membrane becomes leaky)
CAVEAT: over-decolourization leads to washing away of dye from G+ as well
purpose of safranin in gram staining
secondary stain, counterstain: dyes pink for colourless bacteria (G-)
examples of G+ bacteria (5)
1. Bacillus subtilis
2. Micrococcus roseus
3. Micrococcus lutea
4. Escherichia coli
5. Staphylococcus epidermidis
6. Staphylococcus epidermidis
7. Lactococcus lactis
What is a capsule?
thick layer of polysaccharides or glycoproteins surrounding and adhering to bacteria (type of glycocalyx)
- protects cell from desiccation
- protects cell from predators
- allows adherence to favourable surfaces
- increase virulence of microbe
Why are capsule difficult to stain? How to stain?
water soluble and uncharged; thus, background stain is needed so capsule appears as a halo (heat-fixing not allowed)
capsule stain procedure (6 steps)
1. Congo red stain
2. add bacteria and air dry
(3. stupid manual states to wash with dH₂O)
4. Maneval's stain
(5. stupid manual states to gently rinse with dH₂O and blot)
6. observe under oil immersion
purpose of Congo red stain in capsule staining
acidic dye to make background red
purpose of Maneval's stain in capsule staining
lowers pH even more to make Congo red stain purple; dyes inside of bacterial, cell, too (but not the capsule)
example of capsule-forming bacteria
Lactococcus lactis
composition of agar in nutrient agar
1.5% agar
composition of motility test agar
0.4% agar
- lower to allow bacteria motility thru it
tetrazolium salt (TTC)
purpose of tetrazolium salt (TCC) in motility test agar
electron acceptor for bacteria growth
- upon its reduction, becomes red and insoluble (when oxidized, colourless and soluble)
- if red, that means bacteria had been in that part of the agar (for tracking bacterial movement)
interpreting the results of a motility test (2)
1. tube is red throughout: mobile bacteria
2. tube has only red colour in stab line: Brownian movement

motility test procedure (2 steps)
1. introduce 1-inch bacteria w. inoculating needle into inner cylinder or slant tube
2. incubate overnight @37°C
examples of motile bacteria (2)
1. Escherichia coli
2. Bacillus subtilis (slow)
mycolic acid in waxy cell walls
results in hydrophobic walls that may prevent penetration by charged dyes
staining mycolic acid
cannot use basic stains b/c walls are hydrophobic
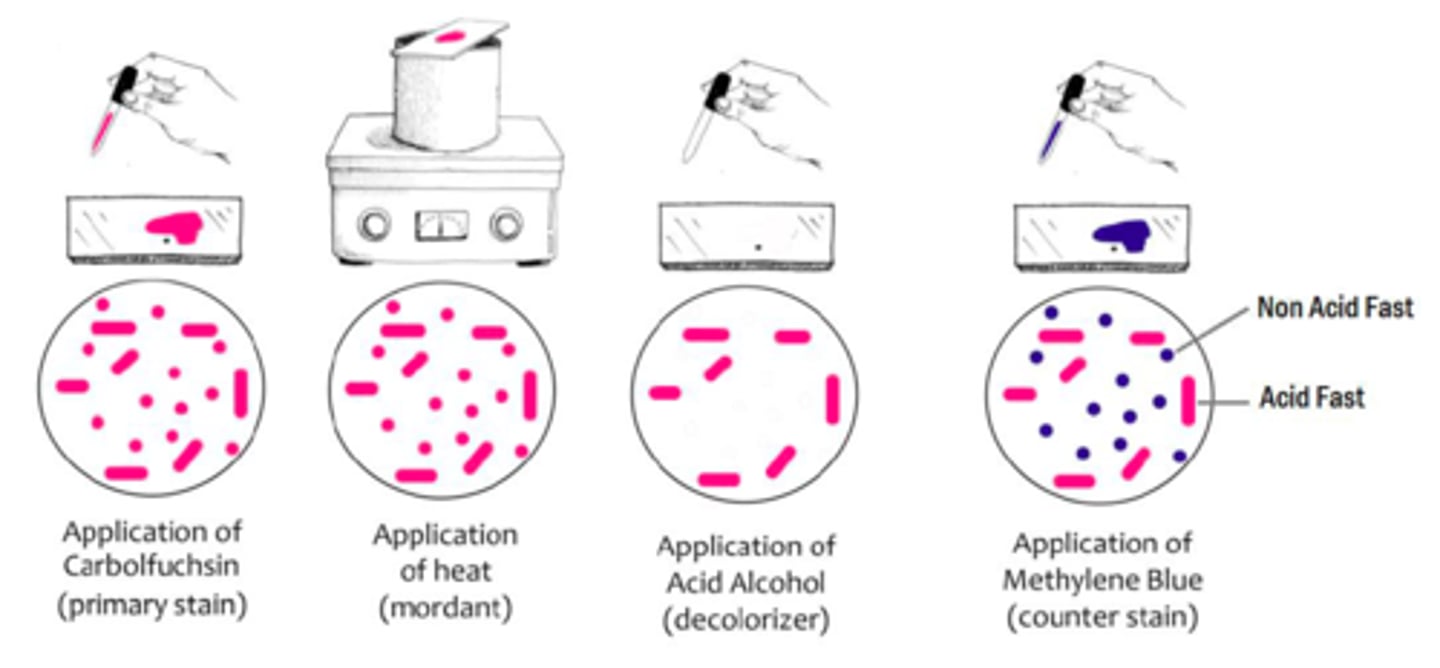
Ziehl-Neelson Method of acid-fast staining (8 steps)
1. heat heat-fixed bacterial smears over boiling dH₂O
2. while still heating, add carbolfuchsin stain
3. rinse with dH₂O
4. acid alcohol until run-off is clear
5. rinse with dH₂O
6. methylene blue stain
7. blot dry
8. observe under immersion oil
Kinyoun's Cold Method of acid-fast staining (7 steps)
1. saturate heat-fixed bacteria with Kinyoun's carbolfuschin
2. rinse with dH₂O
3. acid alcohol until run-off is clear
4. rinse with dH₂O
5. methylene blue stain
6. rinse with dH₂O and blot dry
7. observe under immersion oil
difference between Kinyoun's Cold Method and Ziehl-Neelson Method of acid-fast staining
Kinyoun carbolfuchsin has higher concentration of phenol to allow penetration of stain thru waxy cell wall w/o heat
purpose of carbolfuchsin in acid-fast staining
primary dye: pink dye comprises basic fuchsin and phenol, which stains both acid-fast + and - cells
purpose of heat in acid-fast staining
melts away the waxy mycolic acid to allow the stain to enter the cell
purpose of acid alcohol in acid-fast staining
decolourizer: decolorizes acid-fast negative cells b/c once wax cools, carbolfuchsin is trapped w/in was in acid-fast positive cells
purpose of methylene blue in acid-fast staining
counterstain: blue dye to colourize acid-fast negative cells
example of acid-fast positive bacteria
Mycobacterium (genus, level 2 biosafety)
Where is the endospore on B. subtilis?
subterminal
significance of endospore
heat- and chemical-resistant form produced by bacteria to adverse conditions (lack of nutrients, wastes, high temperature, antibiotics etc.) to allow the dormancy of the cell
endospore composition
keratin
dipicolinic acid (high in Ca2+)
How long for endospore to become visible?
48 hrs: endospore w/in the cell
5 days: endospores outside the cell
endospore stain procedure
1. stain heat-fixed bacteria w. malachite green over boiling water
2. cool and rinse w. decolourizer dH2O
3. counterstain with safranin
4. rinse with dH2O and blot dry
5. observe under oil immersion
example of endospore-positive bacteria
B. subtilis
catalase
breaks down H2O2 into water and oxygen, neutralizing bactericidal H2O2; synthesized by aerobic and facultative anaerobic bacteria
Which organisms produce catalase?
aerobic or facultatively anaerobic bacteria
examples of organisms with catalase activity (strong) (4)
1. S. epidermidis
2. B. subtilis
3. M. lutea
4. M. roseus
ways to isolate pure culture from mixed culture
1. streak plating
2. spread plating
What is a colony forming unit?
origin of a colony on a streak plate
streak plate technique
streak on a third of the plate; flame inoculating loop; rotate 90° and streak on 2nd third of the plate pulling from the 1st streak; flame inoculating loop; rotate 90° and streak on 3rd third of the plate pulling from 2nd streak; invert-incubate at 37°C overnight
spread plate technique
spread diluted microbial sample across surface of agar
difference between blow-out (serological/to deliver) pipettes and Mohr pipettes
blow-out/serological/to-deliver: gradations start at tip (first number is 9mL or 1mL starting at the end of the tip); can empty all the fluid out
Mohr: gradations don't start at tip; only cylindrical parts have measured volume (first number is 10mL or 0mL starting at the end of the tip); don't empty fluid out of tip
λ phage infection
recognizes maltose receptor on E. coli outer membrane so that DNA can be ejected and lytic cycle can ensue
significance of NYZ agar
supplemented with MgSO4 and maltose to induce receptor for phage
presence of plaques in plaque assay
successful infection means cell lysis, present as plaques
plaque assay procedure
1. pre-heat NYZ plates at 37°C; keep soft agar tubes in 50°C baths
2. add dH2O to bacteriophage lambda
3. finish making bacteriophage lambda dilutions
4. add E. coli to the bacteriophage lambda dilutions and water to control
5. incubate @37°C for 15 minutes
6. transfer each dilution to soft agar
7. transfer soft agar onto NYZ plate and swirl until solid
8. incubate @37°C overnight
determining plaque forming units
divide statistically acceptable count (30-300) by final dilution factor
life cycle phases of Escherichia coli (4)
1. lag phase: intense metabolic activity but little/no cell division
2. log phase: growth at geometric, highest rate
3. stationary phase: death rate = dividing rate
4. decline rate: cell death exceeds rate of cell division
types of total count (2)
1. direct count (microscopic, Petroff-Hausser Counting Chamber)
- measures both viable and non-viable cells
2. viable count (plating)
- measures only viable cells
turbidity method of cell count
indirect method of measuring total cell population
needs to be calibrated against viable count
wavelength between 450 and 600 nm
minimum cell density for turbidity to be detected
E7 bacterial cells/mL
DEF generation time
time needed for cells to double w/in the log phase
generation time calculation
GT = 0.3/slope of the semi-log graph
original cell density thru direct count calculation
OCD = (# cells counted) / (# squares dilution factor volume of square)
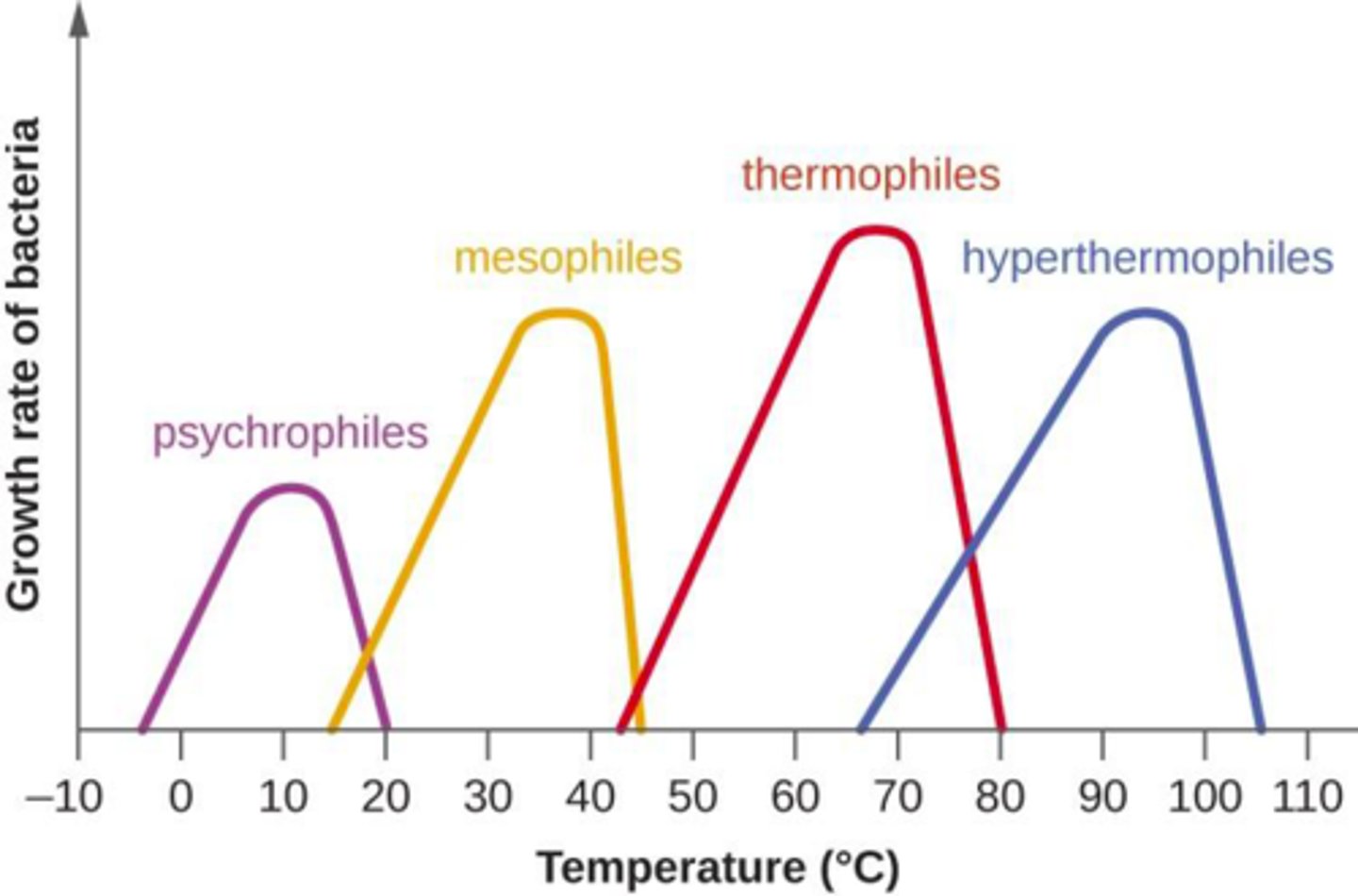
classification of bacteria into preferred temperature range (5)
1. psychrophiles: cold-loving
- grow only below 20°C
2. psychotrophs: cold to warm dwellers
- grow in environments from 0°C to >30°C
3. mesophiles: medium temperature thrivers
- grow in environments from 15°C to 45°C
4. thermophiles: hot-loving
- grow in environments above 40°C
5. extreme thermophile
- grow best above 80°C
classification of bacteria into preferred pH range (3)
1. acidophiles
- optimal growth at pH of 3.0
2. neutrophiles
- optimal growth at pH of 7.0
3. alkalophiles
- optimal growth at pH of 8-10.5
Which part of the cell is most affected by pH?
protein (H-bonding, denaturation)
DEF plasmolysis
shrinkage of cell membrane away from cell wall as water leaves the cell
classification of bacteria based on osmotic tolerance (2)
1. osmotolerant: can withstand high osmotic pressures (wide range of salinities)
2. non-osmotolerant: can only live in low solute concentrations so that fluids don't diffuse out of cell
classification of bacteria based on love of NaCl (2)
1. halophiles: require >3% NaCl to survive
2. extreme halophiles: require up to 25% NaCl to survive
DEF thermal death time
time needed to kill a specific number of microorganisms (initial density) at a specific temperature