units 1-8 ap biology
1/428
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
429 Terms
How do cohesion and adhesion help plants?
They enable capillary action to pull water from roots to leaves.
Why does the R-group matter?
It determines the chemical properties and folding of a protein.
DNA vs RNA structure?
DNA is double-stranded (deoxyribose, T); RNA is single-stranded (ribose, U).
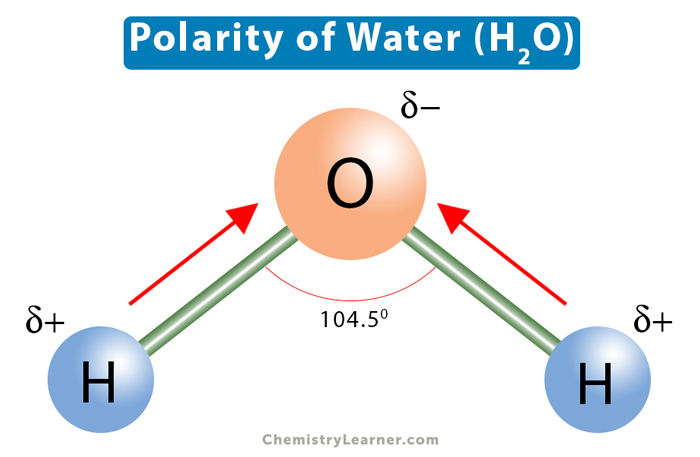
Polarity
Uneven charge distribution where Oxygen is negative (-) and Hydrogen is positive (+).
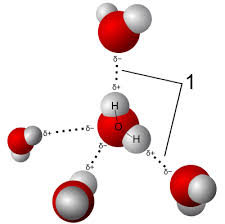
Hydrogen Bond
Weak attraction between polar molecules (e.g., between water molecules).
Cohesion
Water sticking to water.
Adhesion
Water sticking to other surfaces.

Surface Tension
Result of cohesion; allows things to float on water.
High Specific Heat
Water resists temperature changes.
Monomer
Small building block (e.g., amino acid).
Polymer
Large molecule of linked monomers (e.g., protein).
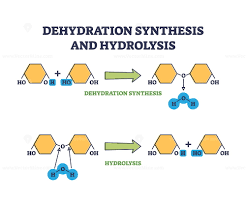
Dehydration Synthesis
Removing water to join monomers.
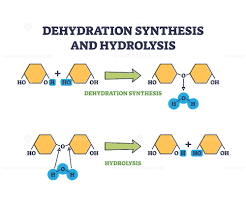
Hydrolysis
Adding water to break polymers apart.
Carbohydrate
Energy and structure; composed of Carbon, Hydrogen, and Oxygen (C, H, O).
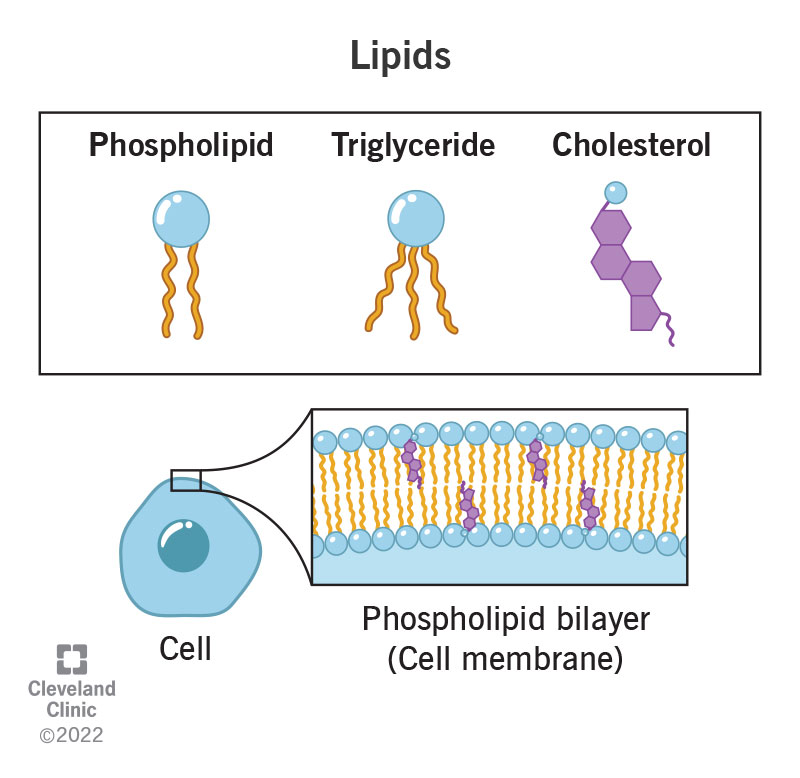
Lipid
Hydrophobic; energy storage and membranes;
Protein
Polymers of amino acids; composed of Carbon, Hydrogen, Oxygen, Nitrogen, and Sulfur (C, H, O, N, S).
Nucleic Acid
Genetic information (DNA/RNA); composed of Carbon, Hydrogen, Oxygen, Nitrogen, and Phosphorus (C, H, O, N, P).
Protein Mutation
If a hydrophilic amino acid is swapped for a hydrophobic one, the protein will fold differently, likely losing function.
Denaturation
High heat or pH changes break hydrogen bonds, changing protein shape and stopping function.
Evaporative Cooling
High heat of vaporization allows sweat to remove body heat efficiently.
α (Alpha) Linkages
Curved/helical chains found in starch and glycogen; easily broken down by enzymes.
β (Beta) Linkages
Straight rigid fibers found in cellulose; humans cannot digest.
Ribosome
Site of protein synthesis.
Endoplasmic Reticulum (ER)
Rough (proteins) and Smooth (lipids/detox).
Golgi Complex
Folding, chemical modification, and packaging of proteins.
Mitochondria
Site of ATP production (double membrane).
Lysosome
Contains hydrolytic enzymes for digestion/recycling.
Vacuole
Storage (water/waste); large central vacuole in plants.
Chloroplast
Site of photosynthesis (algae/plants).
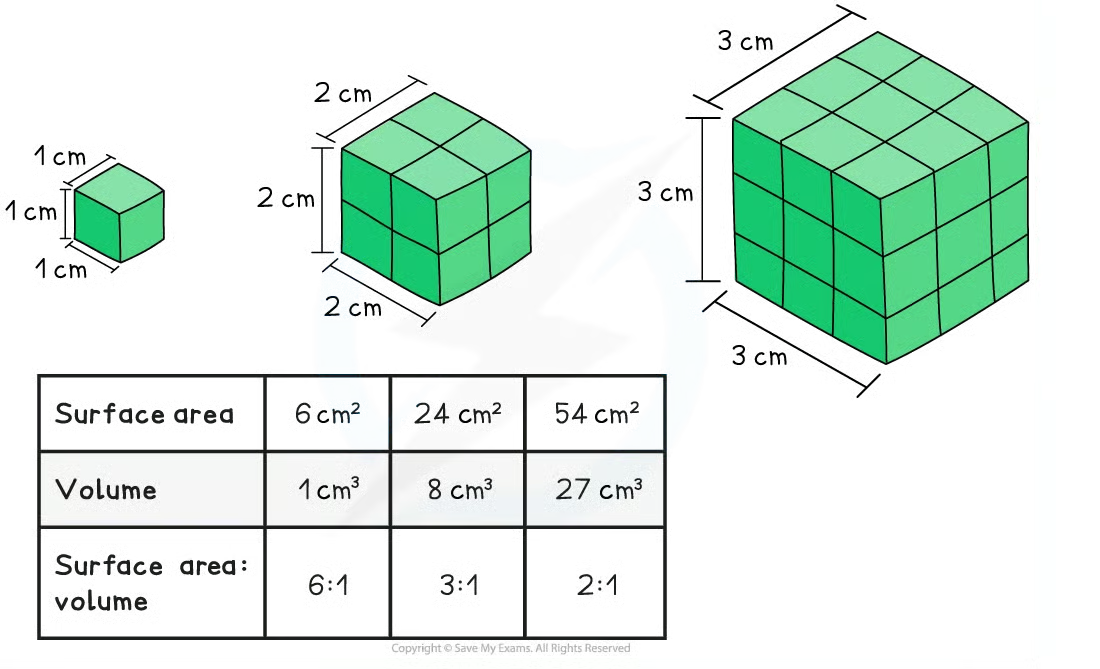
Surface Area-to-Volume Ratio
Smaller cells are more efficient at exchange.
Phospholipid Bilayer
Amphipathic membrane (hydrophilic heads, hydrophobic tails).
Selective Permeability
Ability of membrane to regulate what enters/exits.
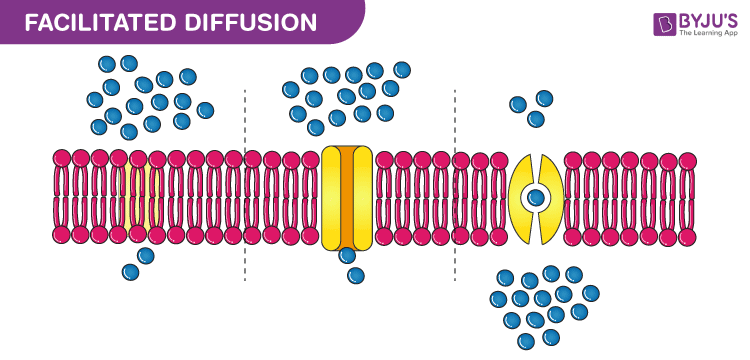
Facilitated Diffusion
Passive transport via membrane proteins.
Active Transport
Requires ATP to move molecules against gradient.
Endocytosis/Exocytosis
Bulk transport into/out of cell.
Tonicity
Ability of solution to cause a cell to gain/lose water (Hyper, Hypo, Iso).
SA:V Ratio Effect on Cell Size
As a cell grows, volume increases faster than surface area, making it harder to move materials in/out.
Organelle Interaction
Describing how a protein moves from a Ribosome -> ER -> Golgi -> Plasma Membrane.
Water Potential Movement
Water moves from areas of High Water Potential (low solute) to Low Water Potential (high solute).
Membrane Evolution
The Endosymbiotic Theory (Mitochondria/Chloroplasts having their own DNA and double membranes).
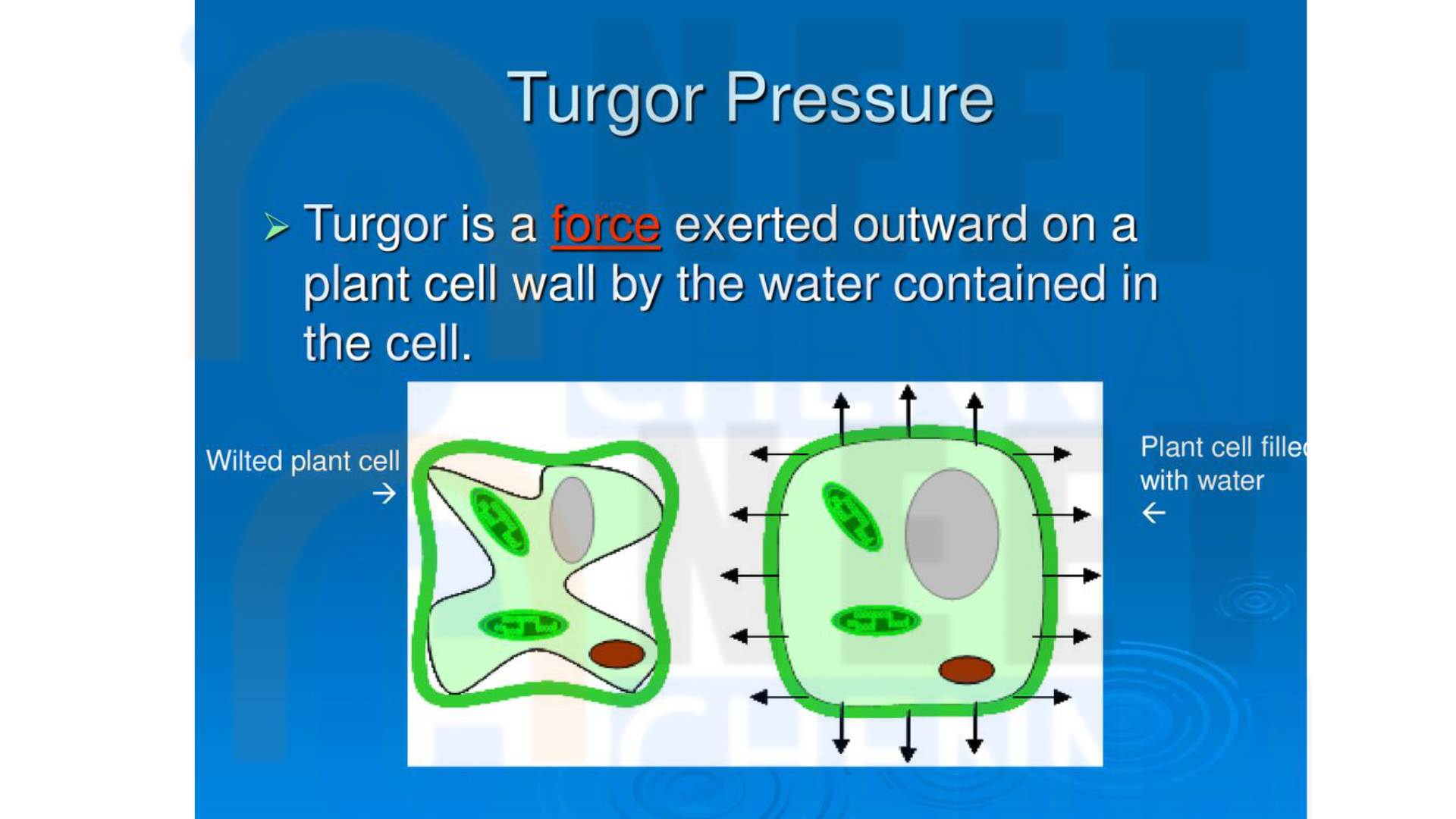
Turgor Pressure
Internal pressure against the cell wall caused by water entering a plant cell.
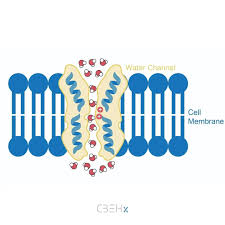
Aquaporins
Specialized protein channels that facilitate the rapid transport of water, move with concert graduation and until equilibrium
Ion Channels
Proteins that allow specific ions to pass through the membrane down their gradient.
Hypertonic
Higher solute concentration outside; causes cell to lose water.
Hypotonic
Lower solute concentration outside; causes cell to gain water.
Turgor Pressure Role
It provides structural support to plants by keeping cells rigid in hypotonic environments.
Aquaporins Necessity
While water can slowly diffuse across the membrane, aquaporins allow for the massive, rapid flow of water needed for osmoregulation.
Osmosis
The net diffusion of water across a selectively permeable membrane.
Hypotonic Solution Effect
Water moves IN; animal cells may burst (lyse); plant cells become turgid (ideal).
Hypertonic Solution Effect
Water moves OUT; animal cells shrivel; plant cells undergo plasmolysis.
Isotonic Solution Effect
Equal solute; no net water movement;
Water Hydrogen Bonds Property
Water is polar, allowing hydrogen bonding between molecules.
Water as a Good Solvent for Polar Molecules
Its polarity allows it to surround and interact with other polar molecules, dissolving them.
Carbohydrate Structure and Function Relation
Monosaccharides are energy sources; polysaccharides like starch store energy, and cellulose provides structural support in cell walls.
Saturated vs Unsaturated Fats
Saturated fats have no double bonds, are solid at room temp; unsaturated fats have double bonds, are liquid at room temp.
Phospholipid Function in Cell Membrane
They form a bilayer with hydrophilic heads facing out and hydrophobic tails inside, providing a selective barrier.
Enzyme Role in Chemical Reactions
They lower activation energy and stabilize the transition state.
Temperature and pH on Enzyme Activity
Extreme temperatures or pH can denature enzymes, reducing or stopping activity.
ATP Energy Storage and Release
Energy is stored in the phosphate bonds; breaking the terminal phosphate bond releases energy for cellular processes.
Prokaryotic vs Eukaryotic Cells
Prokaryotes lack a nucleus and membrane-bound organelles; eukaryotes have both.
Free vs Bound Ribosomes in Eukaryotic Cells
Free ribosomes make proteins for use in the cytosol; bound ribosomes make proteins for export or membranes.
Maintaining Homeostasis in Water Balance
Through osmosis; water moves from high to low concentration across semi-permeable membranes.
Fluid Mosaic Model Explanation
The cell membrane is a dynamic structure with proteins embedded in a phospholipid bilayer that allows movement and flexibility.
Dehydration Synthesis vs Hydrolysis
Dehydration synthesis builds polymers by removing water; hydrolysis breaks polymers by adding water.
Enzyme Specificity to Substrates
The active site has a shape complementary to the substrate (lock-and-key or induced fit model).
Functional Groups Effect on Macromolecule Properties
They determine polarity, solubility, and chemical reactivity, influencing biological function.
DNA vs RNA Structure and Function
DNA is double-stranded, stores genetic info; RNA is single-stranded, involved in protein synthesis.
Cells Using Energy from Chemical Reactions
They couple exergonic reactions (release energy) to endergonic reactions (require energy), often using ATP.
Enzymes Role in Metabolic Pathways
They control the rate of each step, ensuring efficient energy use and product formation.
Water Cohesion and Adhesion Importance for Plants
Cohesion helps water move through xylem; adhesion allows water to stick to cell walls, aiding transport.
Temperature and Solvent Effects on Chemical Reactions
Higher temperatures increase reaction rates; solvents can stabilize or destabilize reactants and products.
Enzyme
A biological catalyst (usually a protein) that speeds up reactions by lowering activation energy.
Substrate
The specific reactant an enzyme acts on.
Active Site
The region of the enzyme where the substrate binds.
Denaturation
When an enzyme loses its shape (and function) due to high heat or extreme pH.
Activation Energy ($E_a$)
The initial energy required to start a chemical reaction.
Competitive Inhibitor
A molecule that binds to the active site, blocking the substrate.
Noncompetitive (Allosteric) Inhibitor
A molecule that binds to a different site, changing the enzyme's shape so the substrate can't fit.
First Law of Thermodynamics
Energy cannot be created or destroyed, only transformed.
Second Law of Thermodynamics
Every energy transfer increases the entropy (disorder) of the universe.
Photosynthesis
The process of capturing light energy to make organic compounds (glucose).
Cellular Respiration
The process of breaking down glucose to produce ATP.
ATP (Adenosine Triphosphate)
The main energy currency of the cell.
Light-Dependent Reactions
Occur in the thylakoid. Use light and water to produce NADPH, ATP, and $O_2$ (waste).
Calvin Cycle (Light-Independent)
Occurs in the stroma. Uses ATP, NADPH, and $CO_2$ to produce G3P (sugar).
Glycolysis
Occurs in the cytosol. Breaks glucose into pyruvate (produces 2 ATP, 2 NADH).
Krebs Cycle (Citric Acid Cycle)
Occurs in the mitochondrial matrix. Produces $CO2$, ATP, NADH, and $FADH2$.
Oxidative Phosphorylation
Occurs on the inner mitochondrial membrane (ETC). Uses $O2$ and electrons from NADH/$FADH2$ to make ~30-34 ATP.
Temperature Effect on Enzyme Activity
Activity increases with temp up to an optimal point, then drops sharply as the enzyme denatures.
Final Electron Acceptor in ETC
Oxygen (it combines with $H^+$ to form water).
No Oxygen Consequence
The cell undergoes Fermentation (Lactic Acid or Alcoholic) to recycle NAD+ so glycolysis can continue.
Mitochondria Structure
Outer Membrane (smooth), Inner Membrane (Cristae), Matrix (Krebs Cycle), Intermembrane Space (H+ ion gradient).
Chloroplast Structure
Thylakoids (site of Light Reactions), Stroma (Calvin Cycle), Chlorophyll (main pigment, absorbs light).
Calvin Cycle Process
Carbon Fixation (CO2 + RuBP via Rubisco). 2. Reduction (ATP, NADPH -> G3P). 3. Regeneration (recycles G3P to RuBP).
Fermentation Goal
To recycle NAD+ so that Glycolysis can keep running and making a small amount of ATP.
Lactic Acid Fermentation
Occurs in muscle cells; produces lactate.
Alcoholic Fermentation
Occurs in yeast; produces Ethanol and CO2.
Carotenoids
Yellow/orange/red pigments that absorb extra light energy to protect chlorophyll from damage.
Rubisco
The enzyme that 'fixes' inorganic carbon into the Calvin Cycle.
RuBP
The 5-carbon molecule that starts the Calvin Cycle.