Final Monosaccharides
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
oligosacchride
multiple monosacchrides linked
polysacchrides
lots of monosacchrides linked
glycoproteins
oligiosacchride + protein linked
glycoplipid
oligiosacchride linked to lipids
when a fischer projection had D sterochem
the OH group is on the right
which one is D sterochem
left one

how to determine D/L sterochem
-compare the asymmetric center furthest from the carbonyl group, OH right=D
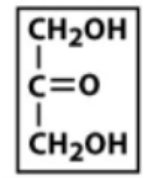
Ketoses
carbonyl group at C2
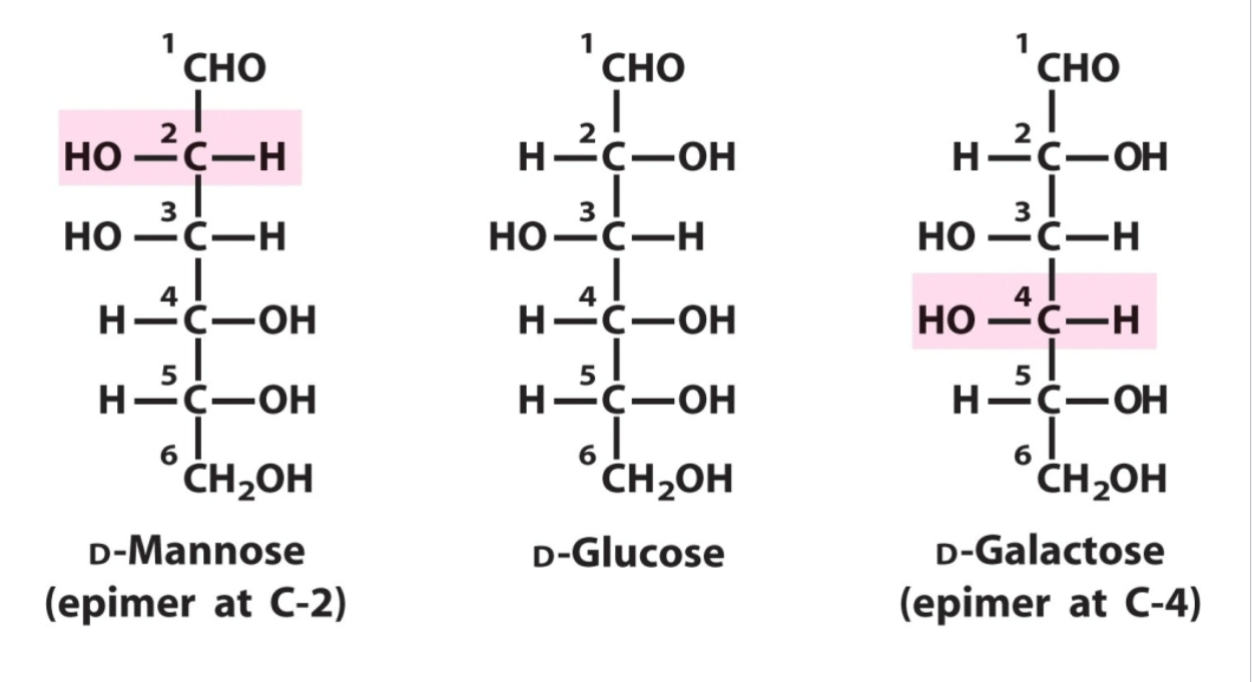
epimers
differ only by configuration around one asymetric carbon, interconverting requires breaking a covalent bond
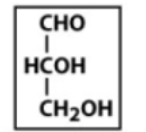
aldehyde sugars
glucose, mannose, galactose, etc
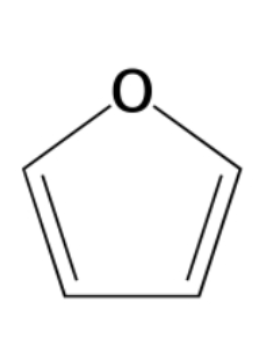
furanose
pyranose
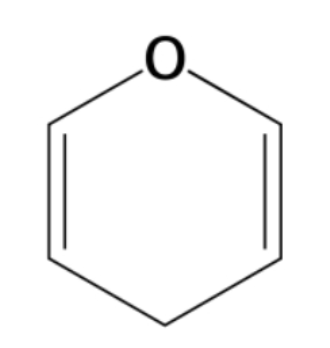
how to convert fisher to ring
-right in fischer- below the ring
-CH2OH up = D, CH2OH down= L
-anomeric same side as C6= beta
mutrarotation
-interconverison of anomers
at phyisological condiditons which anomer is more common
beta is typically more 64%
in chair strucutres, which positions are more crowded
axial positions are more crowded than equitorial
sweet taste receptors are
G coupled reaction between 2 subunits, T1R2 and T1R3, think about a venus flytrap to ligands in extracellular domain, creates a closed confromation which activates CPCR
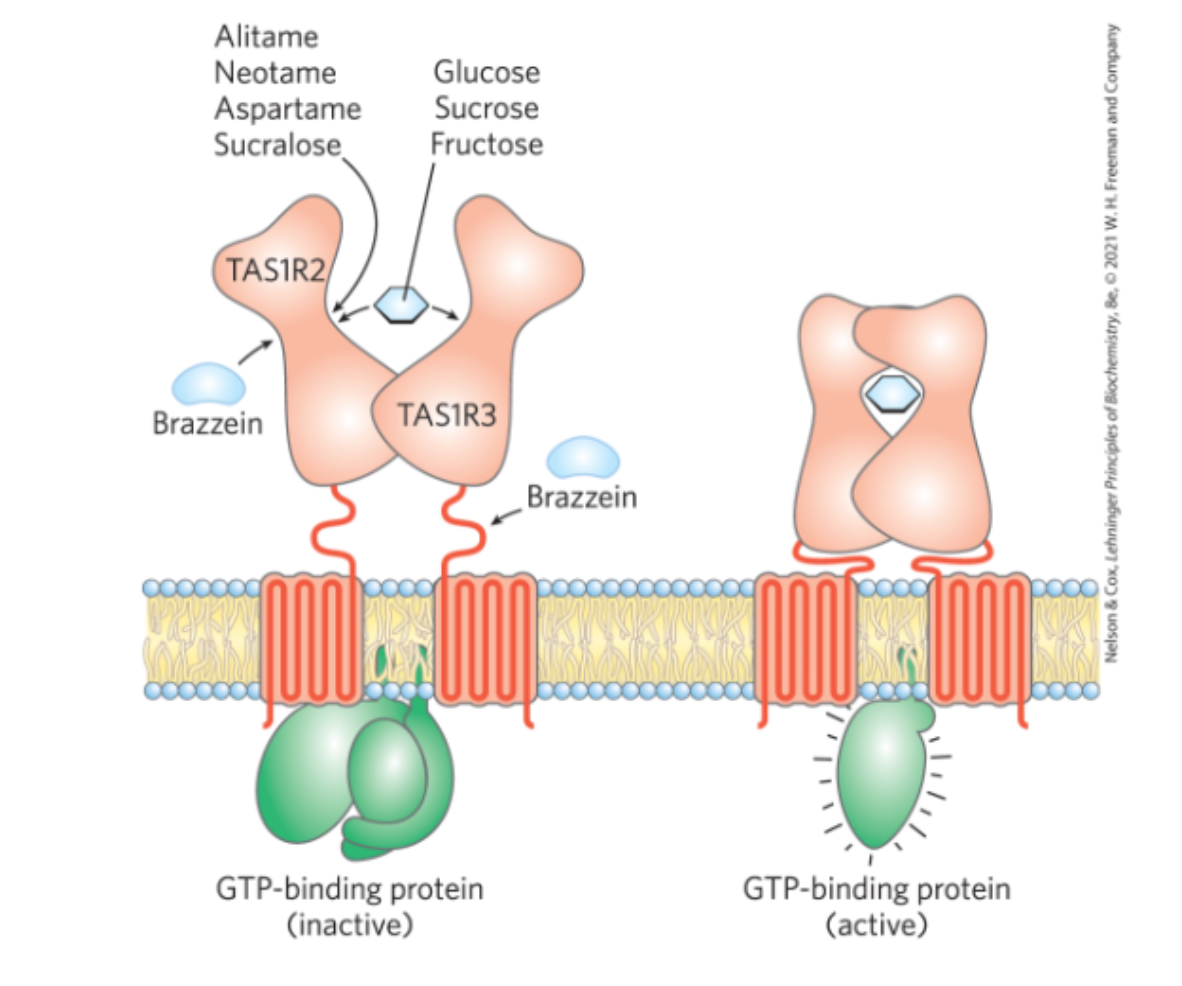
artifical + sweet taste receptors
artifical sugars bind in similar/ overlapping pockets + mimic a closed state, STEROCHEM dependent
sweet taste enhancers + receptors
bind next to the natural ligans in that same pocket
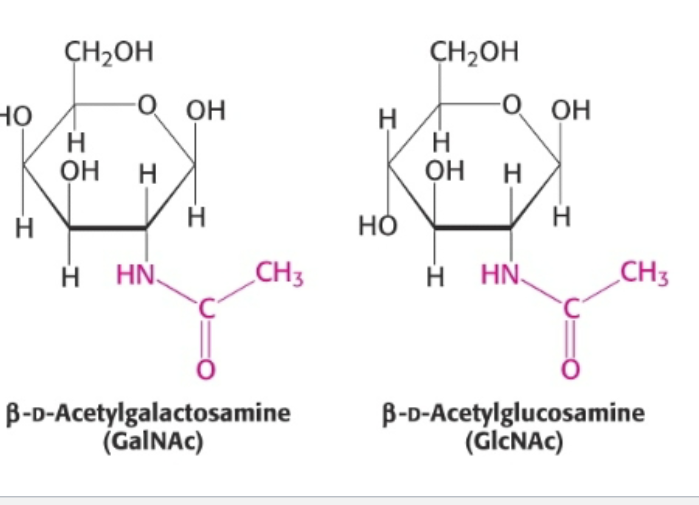
amino modified sugars
-OH that lies at the C2 position is replaced with NH2 or acetylated amine (NCOCH3)
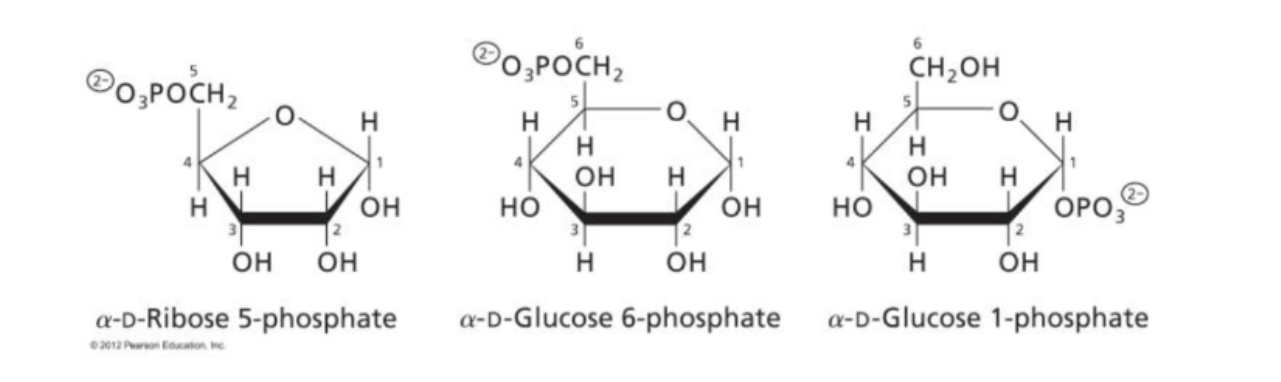
phosphorylated modified sugars
can occur at any position, adds OPO3 with -2 charge, ex alpha-D-Glucose 1 phosphate
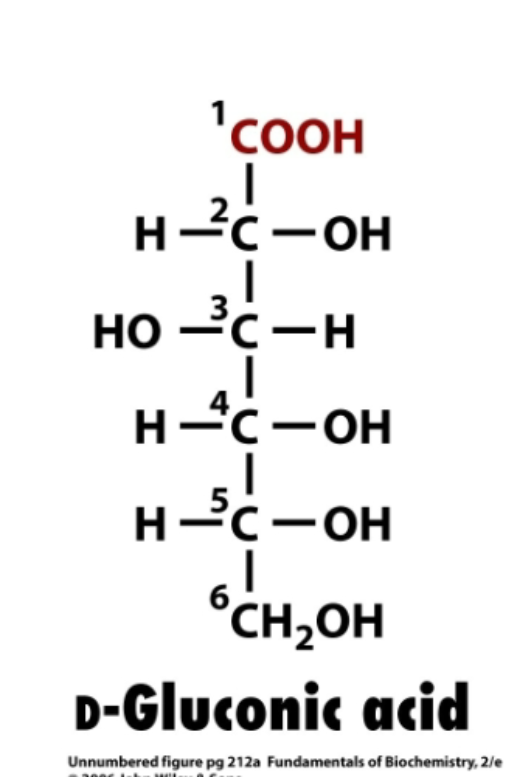
Aldonic acids
alehyde (top, c1, COH) oxidized to carboxylic acid (COOH)
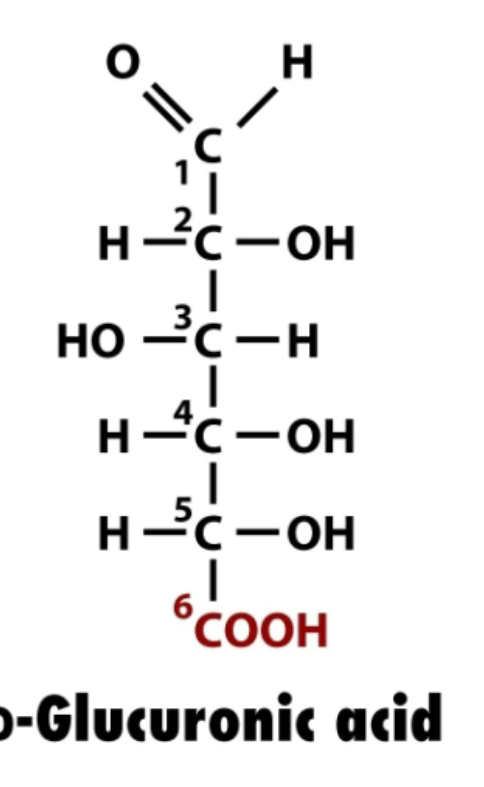
uronic acids
primary alchol oxidized to carboxylic acid (bottom CH2OH C6) to COOH
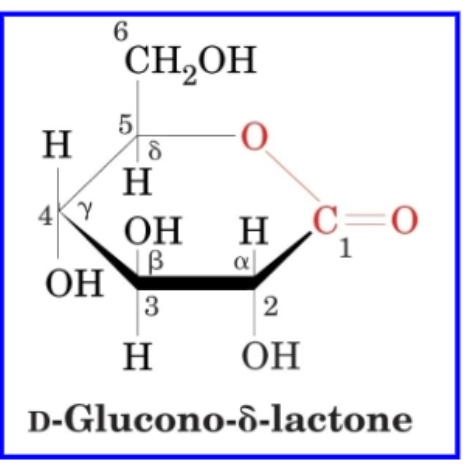
what forms when aldonic acids + uronic acids cyclize
lactones
carbohydrate reduction
loses OH at C6 position, creates alditols
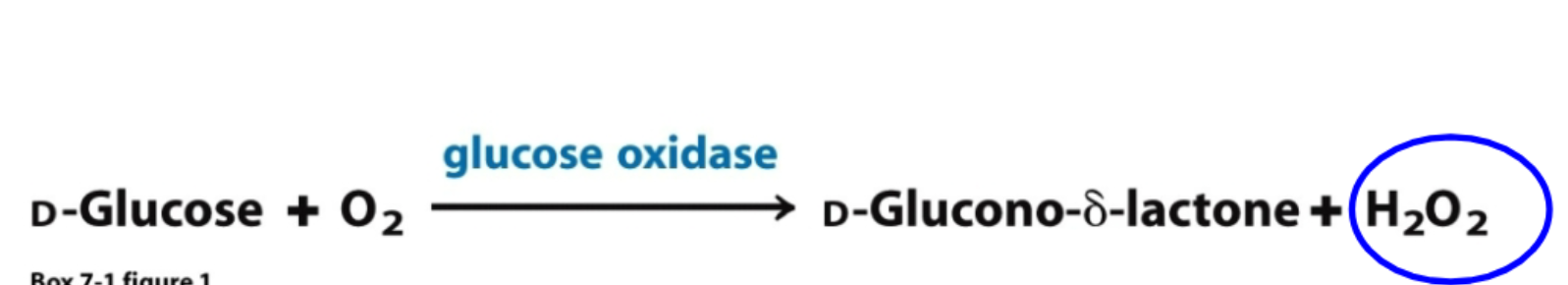
instant blood glucosee measurement
glucose gets oxidized, resulting H2O2 oxidizes a colorless dye, more intense color higher blood glucose
slow HbA1c blood glucose test
reflects average glucose exposure over 3 months, nonfasting