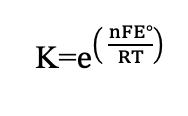Chem Final Equations
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
Avogradro’s Number (Constants)
NA=6.022×1023 mol−1
Gas constant (Constants)
R = 0.08206 L atm / mol K R = 8.314 J / mol K
Faraday Constant (Constants)
F = 96,485 C /mol e
Temperature conversion (Constants)
0°C = 273.15 K
Pressure conversion (Constants)
Pressure conversion (Constants)
1 atm = 760 Torr = 760 mmH
Heat (phase change) calendar (Heat/Energy)
q=nΔHfus
q=mΔHfus
q=nΔHvap
q=mΔHvap
Heat (temperature change) (Heat/Energy)
q = mCΔT
Molarity (Solutions & Concentration)
M = mol solute/L solution
Molality (Solutions & Concentration)
M = mol solute/kg solvent
Mole fraction (Solutions & Concentration)
Xa = mol A / total moles
Mass percent (Solutions & Concentration)
% mass = Mass solute (or solvent)/ Total mass x 100
Freezing point depression (Boiling & Freezing Point Changes)
ΔTf=imKf
Boiling Point Elevation (Boiling & Freezing Point Changes)
ΔTb=imKb
New freezing temp (Boiling & Freezing Point Changes)
Tf=Tf∘−ΔTf
Osmotic Pressure
Π=iMRT
General rate expression (Reaction Rates)
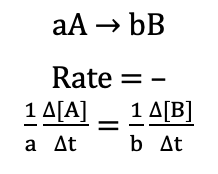
Rate Law (Reaction Rates)
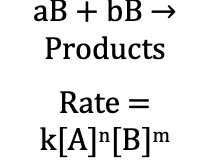
Comparing rate experiments (Reaction Rates)
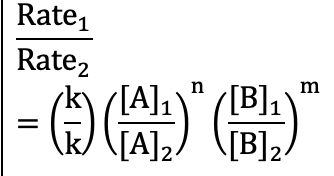
Zero Order (Integrated Rate Laws)
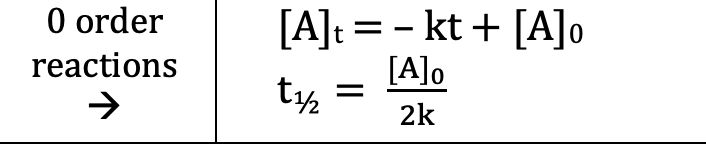
First order (Integrated Rate Laws)
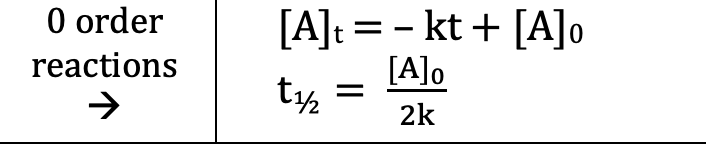
Second Order (Integrated Rate Laws)
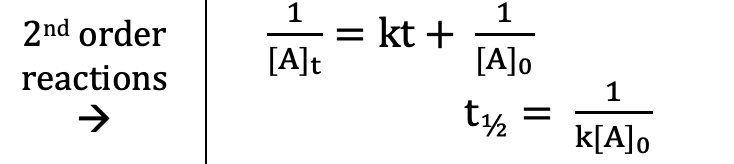
Arrhenius Equation
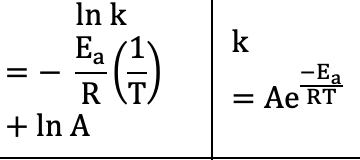
Log Rules
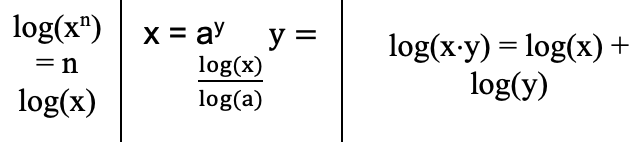
Equilibrium constant (Equilibrium)
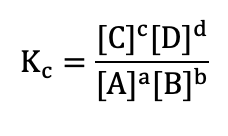
Gas equilibrium (Equilibrium)
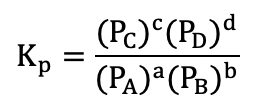
Relationship (Equilibrium)
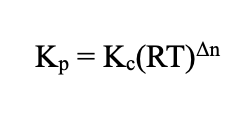
Water equilibrium
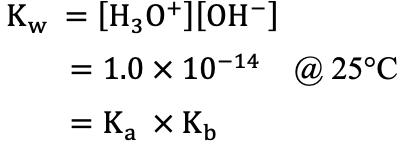
Acid Constant (Acid–Base Equations)
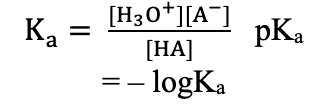
Base Constant (Acid–Base Equations)
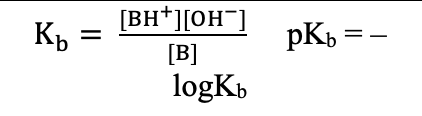
pKa / pKb (Acid–Base Equations)
pKa=−logKa
pKb=−logKb
pH / pOH (Acid–Base Equations)
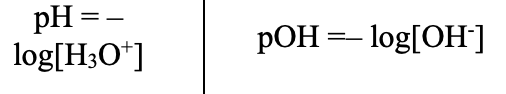
Relationships (Acid–Base Equations)
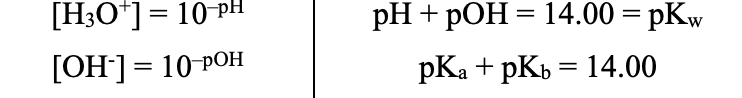
Henderson–Hasselbalch
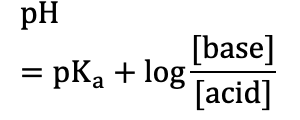
Weak acid/base shortcut
When x is small
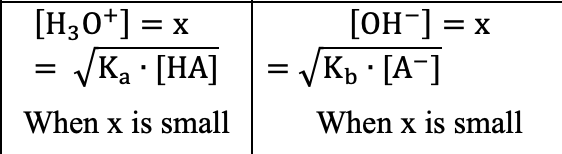
Free energy (Thermodynamics)
ΔG=ΔH−TΔS
Standard free energy (Thermodynamics)
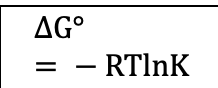
Nonstandard (Thermodynamics)
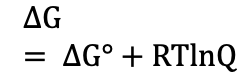
Phase equilibrium temp (Thermodynamics)
T=ΔH/ΔS
Cell Potential (Electrochemistry)
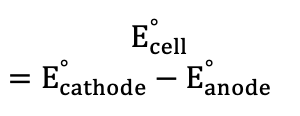
Free energy relationship (Electrochemistry)
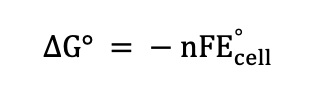
Nernst Equation (Electrochemistry)
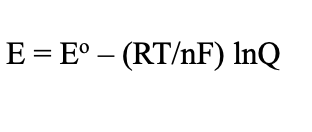
Equilibrium relationship (Electrochemistry)
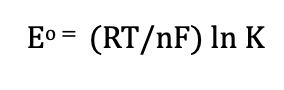
Alternate equilibrium form (Electrochemistry)