chapter 4- alcohols
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
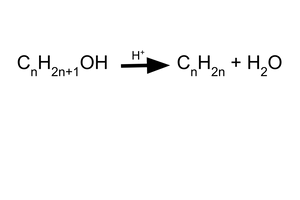
general formula of alcohols?
CnH2n+1OH
what are the 3 types of alcohols?
primary, secondary, tertiary
this is dependent on which carbon atom the -OH is bonded to
what prefix is used when the compound has an -OH group, in addition to another functional group?
-hydroxy
why are alcohols polar?
because of the electronegative hydroxyl group which pulls the electrons in the C-OH bond away from the carbon atom
what type of bonding do alcohols contain?
hydrogen bonding
the strongest kind of intermolecular force
this is why alcohols have low volatility
in what reaction do alcohols react with compounds containing halide ions?
substitution reaction
the hydroxyl group is replaced by the halide, so the alcohol is transformed to form a haloalkane
this reaction also requires the addition of an acid, such as sulfuric acid
water is a by-product of the reaction
what is the reaction where alkenes are made from alcohols?
elimination reaction
the alcohol is mixed with an acid catalyst (either concentrated sulfuric acid or concentrated phosphoric acid). the mixture is then heated
when an alcohol dehydrates, it eliminates water
the water molecule is made up from the hydroxyl group and a hydrogen atom that was bonded to a carbon atom adjacent to the hydroxyl carbon
this means that there are often 2 possible alkene products from one elimination reaction depending on which side of the hydroxyl group the hydrogen is eliminated from
alkene products could also form E/Z isomers
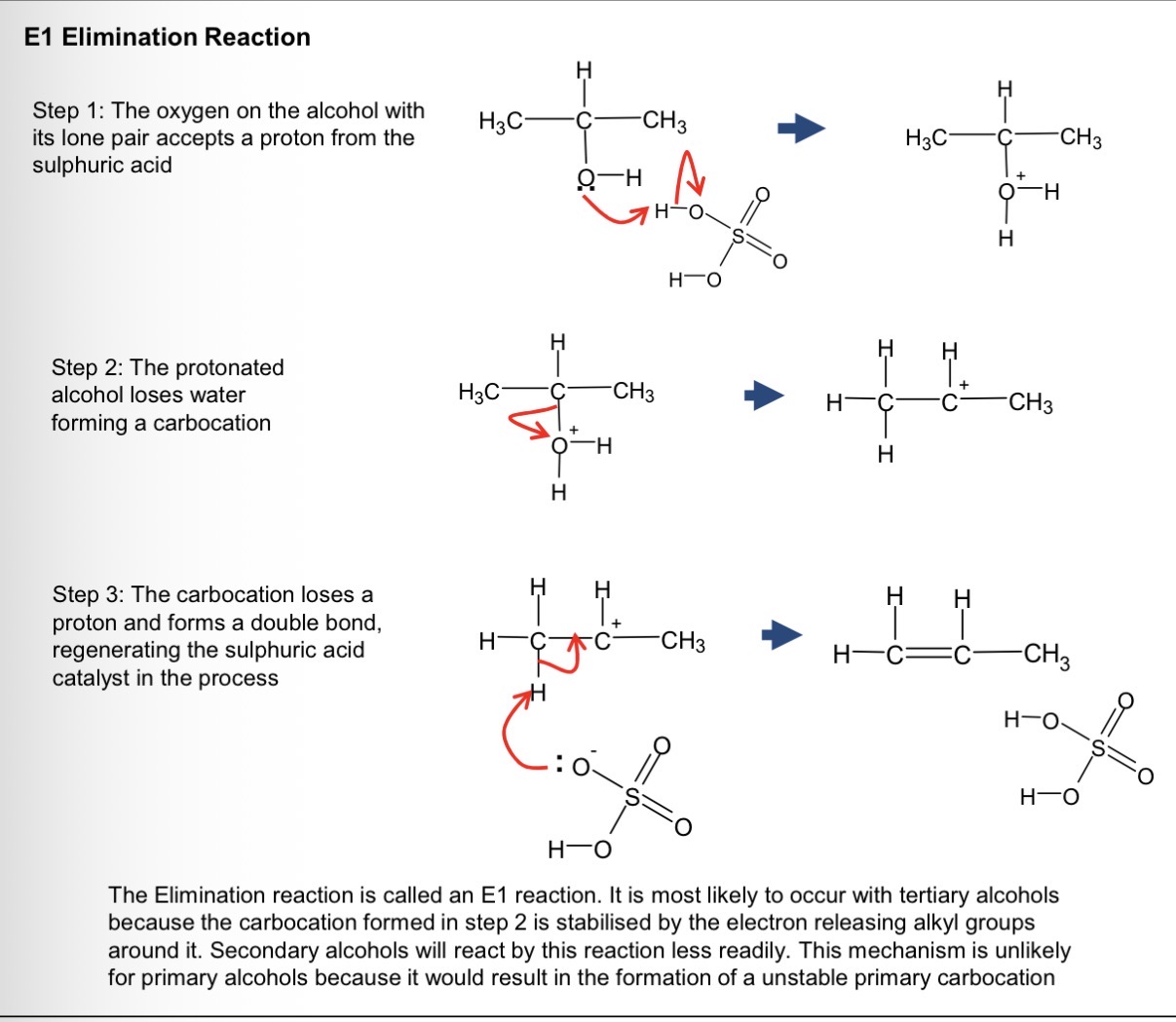
mechanism of elimination?
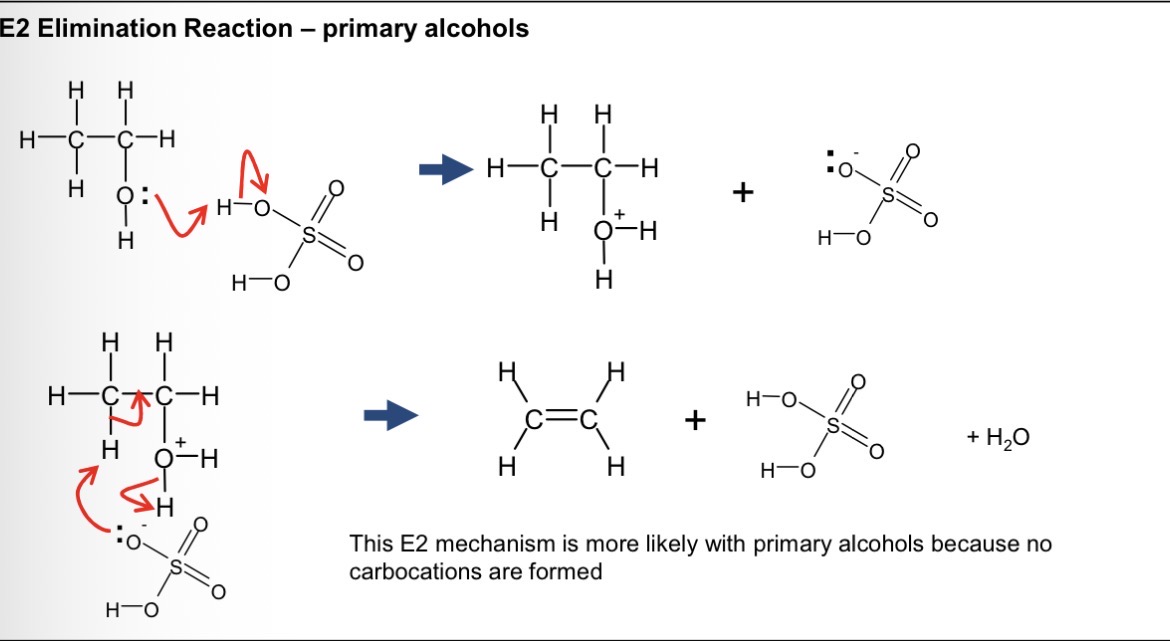
mechanism of elimination for primary alcohols?
why do we need a catalyst for elimination reaction?
the catalyst protonates the -OH group to make it easier to eliminate the group
the acid catalyst is reformed when the H+ is lost as the alkene forms in the last step
when alcohols burn in oxygen, what do they produce?
carbon dioxide and water
what happens when alcohols are burned in insufficient oxygen?
then we get incomplete combustion
depending on how much oxygen is present, we could get a mixture of carbon monoxide, carbon (soot) and water
what is a primary alcohol?
an alcohol where the carbon is bonded to the functional group
e.g- ethanol
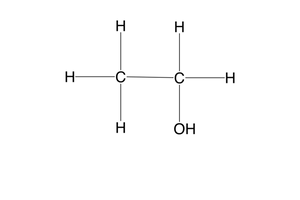
what is a secondary alcohol?
an alcohol where the carbon bonded to the functional group, is bonded to 2 other carbons
e.g- propan-2-ol
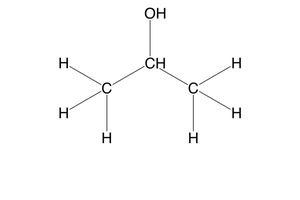
what is a tertiary alcohol?
an alcohol where the carbon bonded to the functional group is bonded to 3 other carbons
e.g- 2-methyl-propan-2-ol
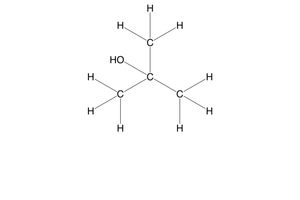
what happens when primary alcohols are oxidised?
they are oxidised to aldehydes, then carboxylic acids
what happens when secondary alcohols are oxidised?
they are oxidised to ketones only
what happens when tertiary alcohols are oxidised?
they won’t be oxidised
functional group of aldehydes and ketones?
CnH2nO
what is the oxidising agent needed for oxidising alcohols?
acidified dichromate
e.g- K2Cr2O7/H2SO4
how can you control how far an alcohol (primary) is oxidised by controlling the reaction conditions?
distil for an aldehyde
reflux for carboxylic acid
how to produce an aldehyde?
carry out the reaction under distillation
this condenses any reacted aldehyde, which forms as a gas, into a new flask
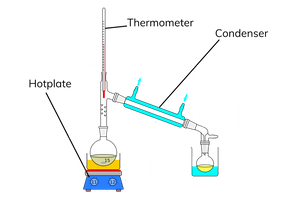
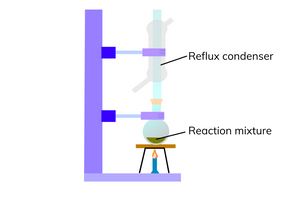
how to produce a carboxylic acid?
carry out the reaction under reflux
this involves the use of a reflux condenser
the reflux condenser condenses any gases and forces them back into the reaction vessel
this puts the aldehyde back in the flask to be oxidised again
what is the only way to oxidise tertiary alcohols?
by burning them
tollens reagent?
ALDEHYDES only are oxidised by tollens reagent into a carboxylic acid
the silver ions are reduced to silver atoms
with aldehydes, a silver mirror forms coating inside the test tube
with ketones, there is no visible change
fehling’s solution?
ALDEHYDES ONLY are oxidised by fehling’s solution into a carboxylic acid.
the copper (II) ions are reduced to copper (I) oxide
in aldehydes, the blue CU2+ ions in the solution change to a red precipitate of CU2O
ketones do not react
how can the presence of a carboxylic acid be tested?
by the addition of sodium carbonate
it will fizz and produce carbon dioxide
what 3 things does reflux do?
thermally accelerate the reaction by conducting it at a constant elevated temperature
keeps volatile substances in the reaction vessels
keeps volatile flammable solvents away from the heat source
reflux apparatus set up and purification?
apparatus:
round bottom flask
heat source
condenser
purification:
separating funnel to separate organic and aqueous layer
drying with anhydrous salt, like magnesium sulfate
redistillation, collect fraction distilling at 102C
distillation conditions- forming aldehyde and carboxylic acid?
forming aldehyde:
h2so4/k2cr2o7
heat AND distillation
organic product is …..
equation is ….
forming carboxylic acid
h2so4/k2cr2o7
heat under reflux
organic product is ….
equation is ….
distillation set up:
diagram of apparatus with condenser
condenser had water flow
collection of organic product
product separated to prevent further oxidation
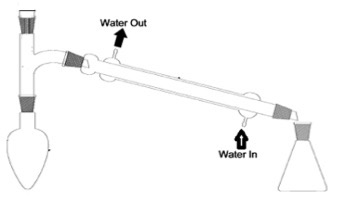
reagents and conditions for oxidation of primary alcohol to aldehyde?
reagents: potassium dichromate (VI) solution and dilute sulfuric acid
conditions: use a LIMITED amount of dichromate, warm gently and use distillation
reagents and conditions for oxidation of aldehyde to carboxylic acid?
reagents: potassium dichromate (VI) solution and dilute sulfuric acid
conditions: use an EXCESS of dichromate and heat under reflux
why is an aldehyde likely to be produced with distillation apparatus?
volatile compounds can escape/distil out
why is a carboxylic acid most likely to be produced with reflux apparatus?
volatile compounds can not escape/distil out
complete oxidation will be achieved/oxidised to the acid
equation for formation of bromoethane from ethanol?
(haloalkane to alcohol)
NaBr + H2SO4 —→ NaHSO4 + HBr
CH3CH2OH + HBr ——→ C2H5Br + H2O
why do alcohols have relatively low volatilities compared to alkanes?
due to their ability to form hydrogen bonds between alcohol molecules
solubility of alcohols in water compared to alkanes?
the smaller alcohols (up to 3 carbon) are soluble in water because they can form hydrogen bonds with water
the longer the hydrocarbon chain, the less soluble the alcohol