Lecture 10 – Proteins Secondary, tertiary, and other structures
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
Prions
Proteinacious infectious particles
We think of almost all agents that cause infection to alive (bacteria, unicellular eukaryotes, etc.), or at least mimic a living organism (virus)
Prions are completely different
Are Prions infectious?
Yes
◦ They’re infectious, even though they’re just a protein. No nucleic acid.
◦ They don’t “replicate” so much as cause your own normally folded proteins to become misfolded
Important to distinguish: They’re infectious, but very difficult to transmit ◦ i.e. I probably can’t sneeze infectious particles onto you
How does a generic prion disease work?
Prion proteins exist as normal proteins in normal cell membranes
Through either spontaneous misfolding, genetic abnormality, or introduction of infectious proteins, there is an over accumulation of unfolded and non-digestible protein matter
◦ Can’t be targeted by ubiquitin and subsequently removed by proteosomes ◦ Can’t be degraded effectively in lysosomes
How are prions infectious?
PrP(C) is the normal variant.
◦ When it comes into contact with the infectious version PrP(Sc) , this version changes the normal protein’s shape into the infectious version
◦ Now there are two infectious particles! Continue process over and over…
How many codified prions diseases are there in humans?
In humans, somewhere between 9-10 different diseases, but occur through three main routes
◦ Spontaneously developed
◦ Genetically derived ◦
Due to infectious particles
◦ All are relatively rare
The more interesting prion diseases:
◦ Kuru
◦ sCJD – Spontaeous Cruetzfeldt-Jacob disease
◦ Also has a familial version
◦ FFI - Fatal familial insomnia
◦ Also has a sporadic version
◦ Iatrogenic CJD
Kuru
Probably the most well known… ◦
Ritualistic Cannibalism – of the brain
◦ Suspected to be caused by one spontaneous case of CJD… ◦
Then, their brain was eaten, spread to rest of group
Spontaneous Cruetzfeldt-Jacob disease
The most common codified prion disease
FFI - Fatal familial insomnia
◦ Similar symptoms, but includes bouts of insomnia, which ultimately cause the death of the patient
Iatrogenic CJD
Derived from infectious particles left of medical equipment
General pathology of prion diseases
PrP(C) → PrP(Sc) , then what?
◦ PrP(Sc) accumulates…
Beta sheets fold themselves into beta helices
These “beta helices” then form fibrils, long strands of non-degradable proteins
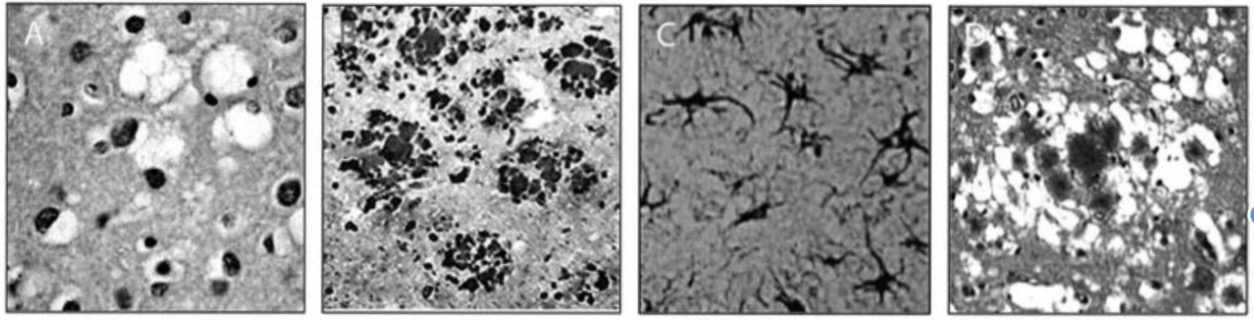
Tissue pathology
Vacuoles – The typical damage that occurs in the disease
◦ Hence – “spongiform”
The void regions form from cells that have died, having left an empty region in its place
Tissues in prions diseases
Spongiform degeneration
Prp plaques (formed by over accumulation of protein structures)
Gliosis
Florid Plaque
Behavioural effects of general prion diseases
Cognitive dysfunction is the most well known, and includes
◦ Irritability and anger
◦ Delusions
◦ “as if someone is taking over my mind”
◦ Hallucinations
Eventually, after so much damage to the brain has occurred, patients pass within 1-2 years of diagnosis
How do prion diseases occur?
Many of these diseases can happen spontaneously, they are poorly understood, and completely incurable
Primary structures of proteins:
◦ The string of polypeptides coded from DNA, potentially modified post-translation, and the subsequent sulphur bridges (covalent bonds) that form within the strand
Secondary structures of protein
Local repeating structures that form once the final polypeptide sequence is complete
◦ Typically formed via hydrogen bonding between N and O groups, and occasionally C groups, in the amino residues
Tertiary structures of protein
The final three dimensional structure that forms once the repeating structures are in place
Quaternary structures of proteins
Multiple fully folded subunits coming together to form complexes
Bond Angles in proteins
Φ =
Ψ =
Φ = phi Bond of N group to C alpha
Ψ = psi Bond of C alpha to C-O
Secondary protein structures What are included in these groups?
Our two most common:
1. Alpha helices (right handed)
2. Beta sheets
Next in line:
3. 310 helix
4. Polyproline II (or polypeptide II) helix
5. Left handed alpha helices
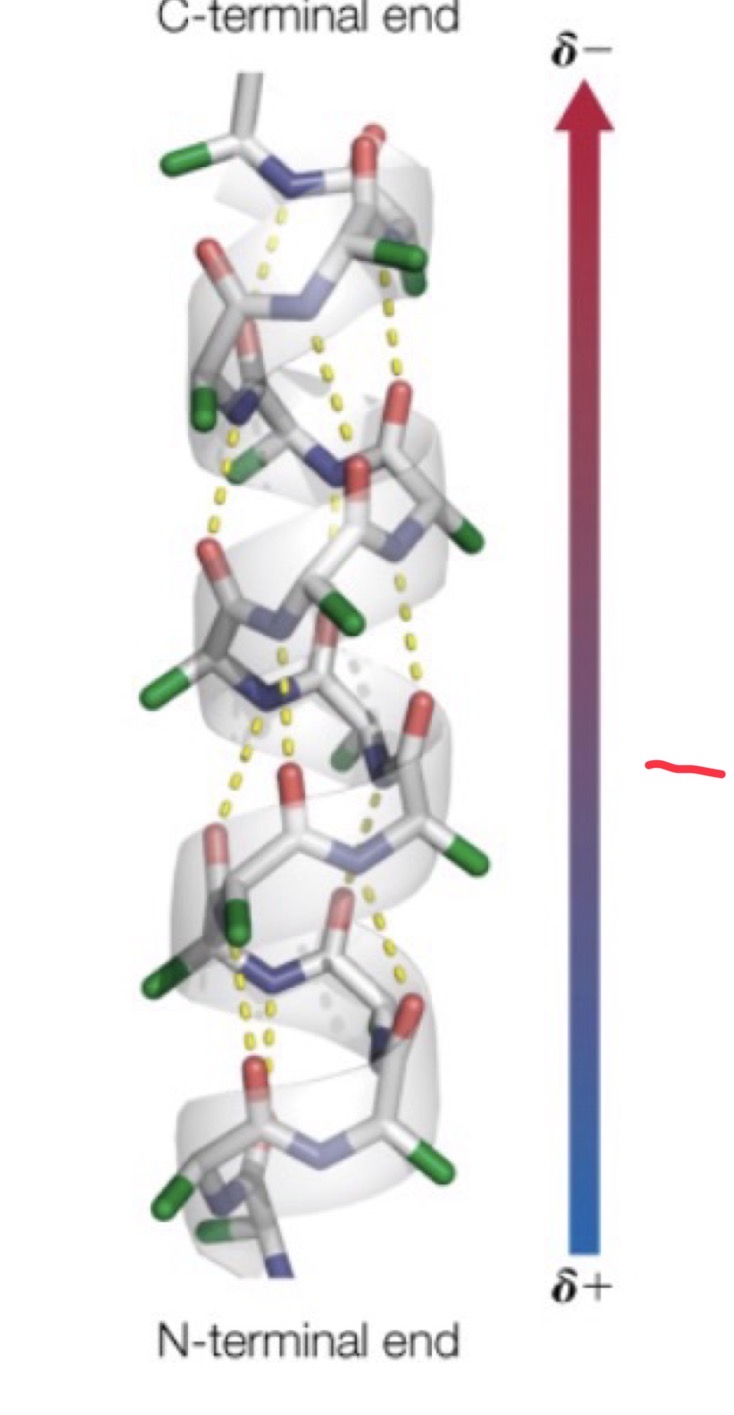
Alpha helices, how are they formed?
Hydrogen bonds formed between:
◦ Oxygen of carboxyl group and nitrogen of amino group
◦ Side chains (R groups) stick out perpendicular to the helix
◦ Depending on which groups are present, the helix could be hydrophilic, phobic, or amphipathic.
◦ When we refer offhand to alpha helices, we are almost always referring to right-handed helices
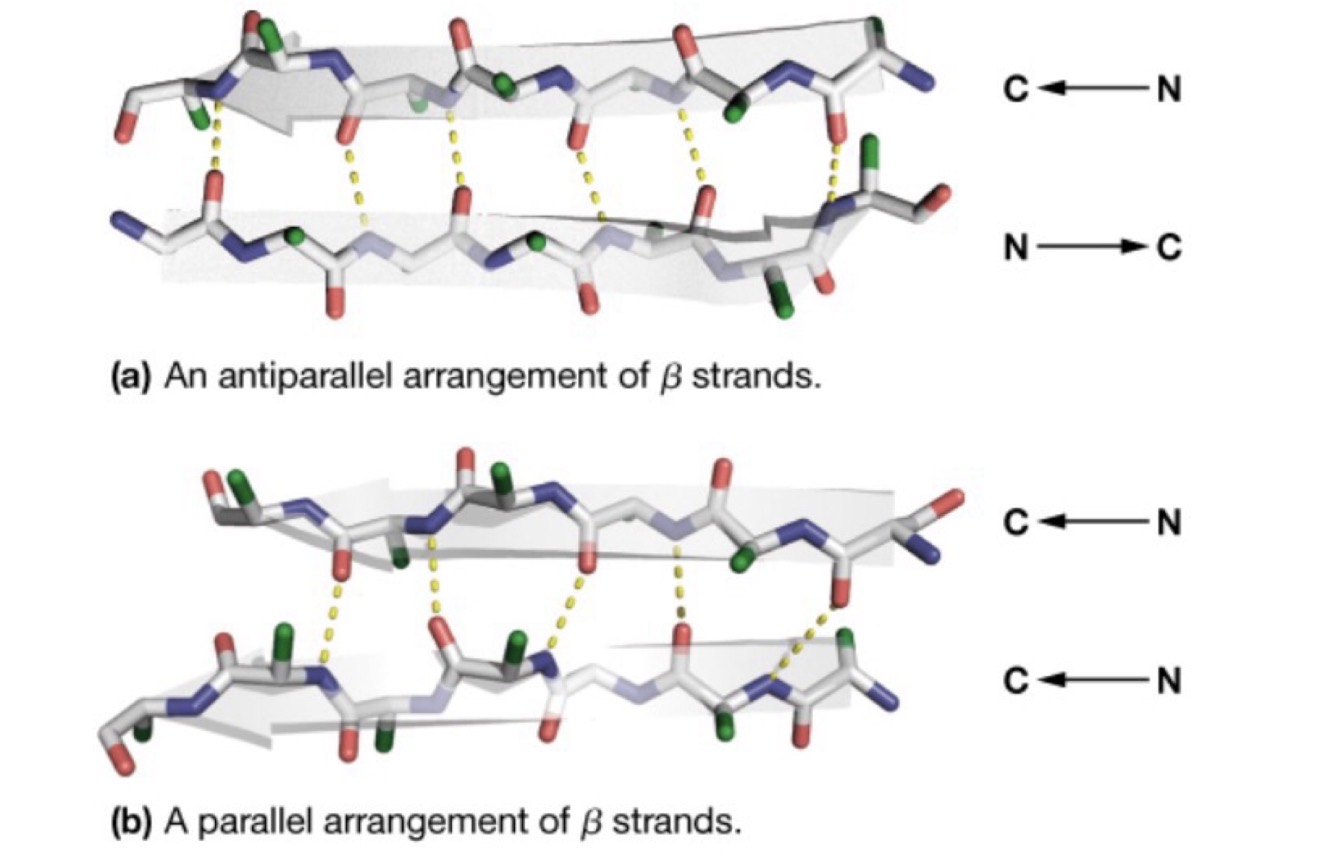
Beta Sheets: Anti-parallel vs. parallel sheets
Anti-parallel: N terminal and C terminal of the cross-bonded sheets run in different directions
Parallel: N terminal and C terminal of the cross-bonded sheets run in the same direction
Bonds between sheets are formed between one of
1. The nitrogen atom of the amino residue with the oxygen of the carboxyl group or
2. The oxygen of the carboxyl group with the beta carbon of the other side (less common)
310 helix
the more uncommon helix
◦Typically, the third most common secondary structure found in amino acids
◦Hydrogen bonds are formed between nitrogen group of prolines or amino groups and a carboxyl group
◦Why 310?
◦3 Amino groups per turn of the helix, 10 atoms found between the hydrogen-bonded atoms
Polyproline II helix
◦No stabilizing hydrogen bonds between turns
◦Composed of primarily proline amino residues, with some glycine
Left-handed alpha-helix
the most uncommon helix!
◦Rare, considerably less stable than its right handed version
How do we know which amino acids exist in which structures?
Ramachandran Plots
How do Ramachandran Plots work?
◦Because we know the predicted bond angles induced between the N-Cα (phi) and the Cα-COO (psi) bonds by the five specific secondary structures of all of the amino acids, we can isolate these structures in proteins via crystallography
◦They give us plots of which amino acids are present in the secondary structures
steric clashing
We also know that some bond angles are simply not possible
This is steric clashing that occurs between the residues of the amino acids
What is a sterically non allowed conformation?
Φ =0, Ψ =0 is not allowed in any polypeptide chain because of the steric crowding between main-chain atoms. A tripeptide is shown, where the central amino acid has Φ =0, Ψ =0
The carbonyl oxygen of residue #1 would clash with the steric residue of the amide hydrogen of residue #3
Ramachandran plot
A map of this type can be used to describe the backbone conformation of any polypeptide residue as well as the secondary structures of proteins.
The coordinates are the values of the bond angles ϕ and ψ,
The white areas correspond to sterically allowed conformations
The gray dots in the background show the observed ϕ and ψ angles for over 10,000 alanine residues found in protein X-ray crystal structures.
The circles withsymbols correspond to the secondary structures
B strand ϕ and ψ angles
ϕ -150 to -100
ψ -120 to 160
Alpha helix ϕ and ψ angles
ϕ -70 to -60
ψ -50 to -40
310 Helix ϕ and ψ angles
ϕ -70 to -60
ψ -30 to -10
Polypetide II helix ϕ and ψ angles
ϕ -80 to -60
ψ 130 to -160
Ramachandran plots for glycine and proline.
Glycine has the greatest number of allowed ϕ, ψ angle combinations, whereas proline has the fewest.
When we think of proteins, we often think:
◦Enzymes
◦Extracellular to intracellular signalling
◦Secondary messenger signalling
◦Hormones and hormone signalling
◦Compound transport
These are almost always globular proteins, which include tertiary and sometimes quaternary structures
Other protein structures can be finalized for their main purpose with, mostly, only secondary changes
Fibrous proteins
amino acid structures which are elongated “strings” of polypeptides
The three main fibrous proteins:
◦Keratin
◦Fibroin
◦Collagen
What amino acids are found in fibrous proteins?
Usually, these proteins have AAs with small, and uncharged, R-groups, which can include:
◦Glycine
◦Alanine
◦Serine
◦Proline
With other amino acids being present in varying numbers depending on the exact nature of the fibre.
Alpha (α) and Beta (β) keratins
•Alpha keratins
•Compose the hair and fingernails of mammals
•Beta (β) keratins
•Talons, beaks, feathers
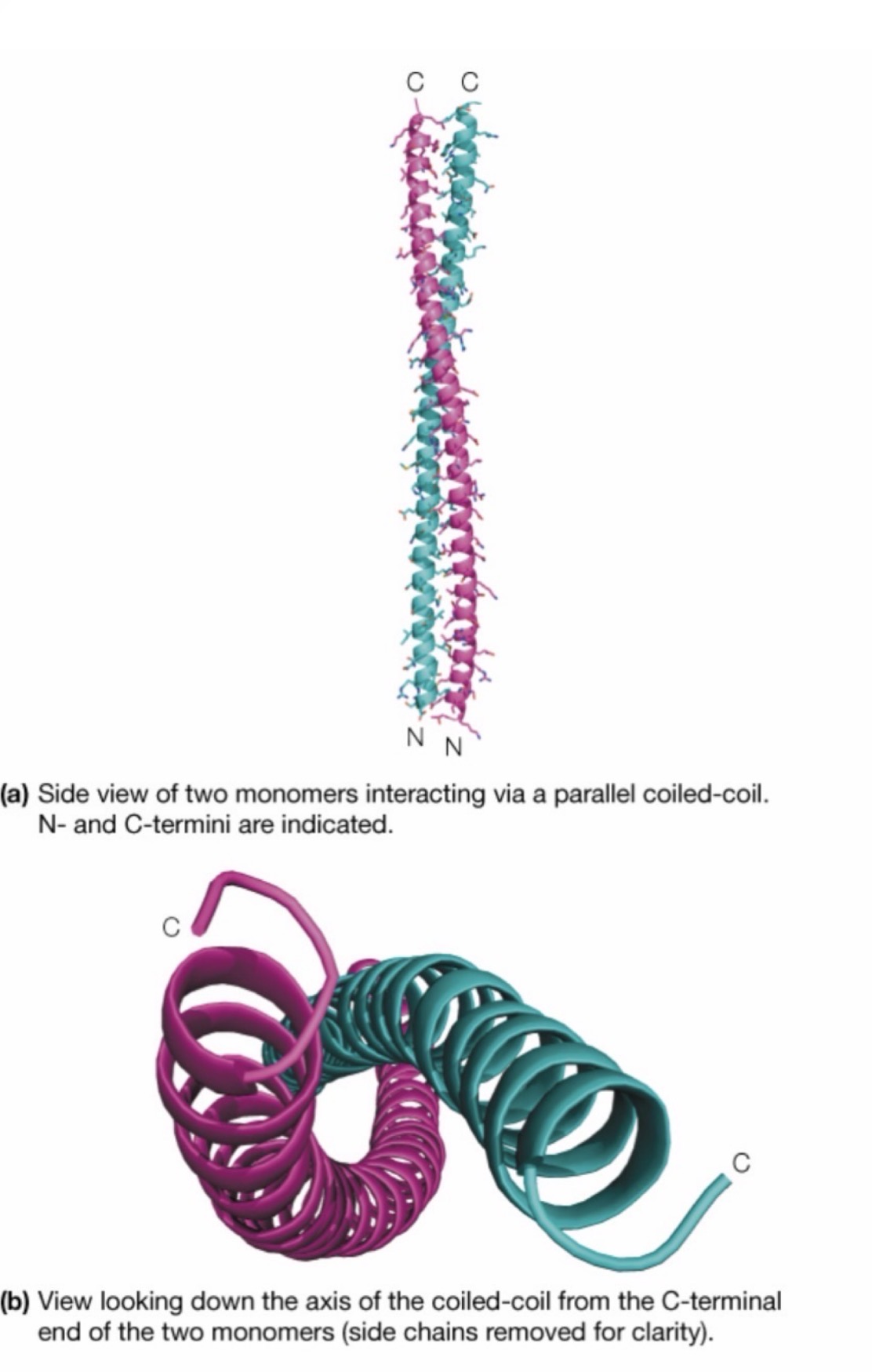
alpha (α) Keratin
Common structural protein – Approximately 300 residues long
Composed of a polypeptide that is almost entirely an alpha helix, hence the name
Two of these strands come together and intertwine, due to some of their small and hydrophobic side chains (glycine, serine)
The larger side chains point outwards
Beta Keratin
A continuous strand of proteins, but rather than alpha sheet being the main structures, it’s the beta helix
Less important for mammals…
More important for other vertebrates:
Helps form the feathers, beaks, and claws of birds and/or reptiles
Fibroin
Again, another secondary structure less important for mammals, but more for invertebrates
◦Silk worms and spiders
Composed almost entirely of anti-parallel B-sheets aligned one on top of each other
◦Again, composed of small amino acids
◦Gly-Ala-Gly-Ala-Gly-Ser-Gly-Ala-Ala-Gly-(Ser-Gly-Ala-Gly-Ala-Gly)8
◦Slightly different patterns can allow for different structural properties
Structural unit of fibroins
Stacked b sheets. alternating layers of interdigitating adenine or glycine side chains
Fibres are very flexible because bonding betweens the sheets involves only the weak van der waals interactions between side chains
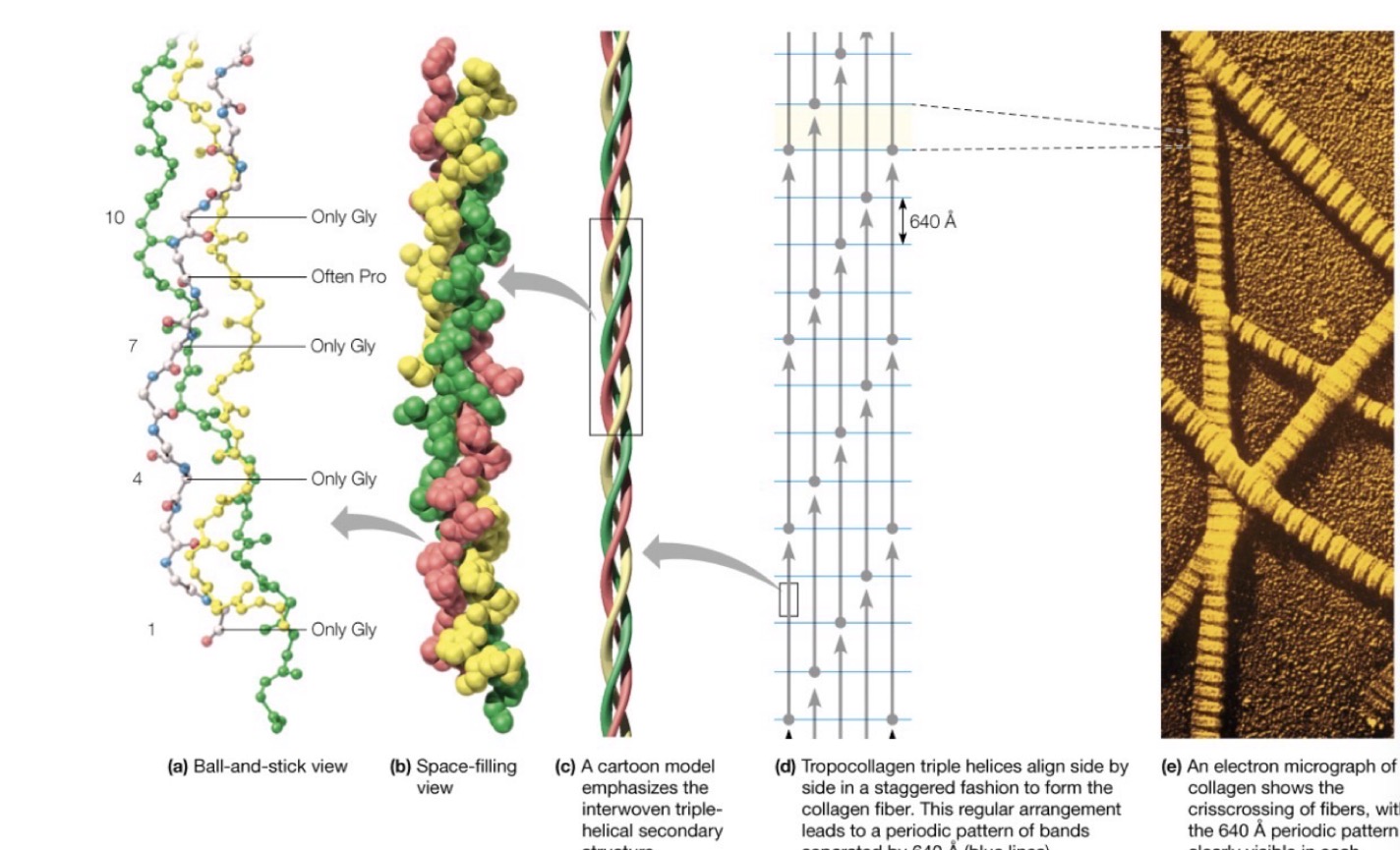
Collagen
Makes up the largest components of the fibrous proteins in animals
◦Connective tissue holding skin together, forms the matrix for holding together bone, and is an important connective piece between bones (tendons and ligaments)
It’s also (sort of) what you’re eating when you have Jell-O
How is collagen composed?
◦A triple helix of intertwining polypeptides
◦A repetitive string of amino acids composed of primarily
◦Glycine, Proline, and a modified version of proline (-4-hydroxyproline)
Gly-X-Y àGly-X-Y à etc. etc
However, variants in this pattern can result in different collagen structures
vitamin C
The enzymes that catalyze the hydroxylations of proline and lysine residues in collagen require vitamin C, L-ascorbic acid
important for collagen
The structure of collagen fibers.
The basic unit of the collagen fiber is a triple helix of three polypeptide chains
how much fibrous proteins are in an organism?
the amount of fibrous proteins in an organism is relatively small…
Globular proteins compose the vast majority of proteins in organisms, including humans
What are Globular proteins involved in?
These molecules are involved in enzymatic and transportation work, and can include proteins required for/as:
◦Signalling (insulin)
◦Anabolism and catabolism (enzymes in metabolism)
◦Signal receptors (neurotransmitter receptors)
◦Destruction / degradation of unnecessary / harmful substances (typically enzymes or antibodies)
Examples of diverse globular proteins
Myoglobin- Transport
Neuraminidase- Enzyme
Triosephosphate isomerase - enzyme
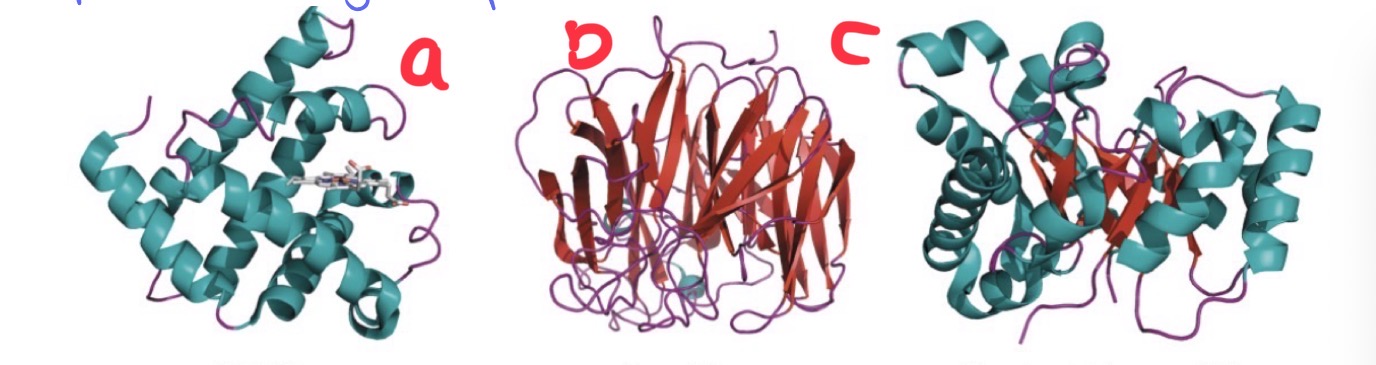
How do we classify globular proteins?
1.The types of secondary structures present
I.Alpha helices, beta sheets, 310 helix, etc.
2.The number of domains present
I.A specific tertiary 3D structure, typically useful for transport or enzymatic activity
3.The types of repeating patters present within the domains
I.i.e. types of a-helices and b-sheets present
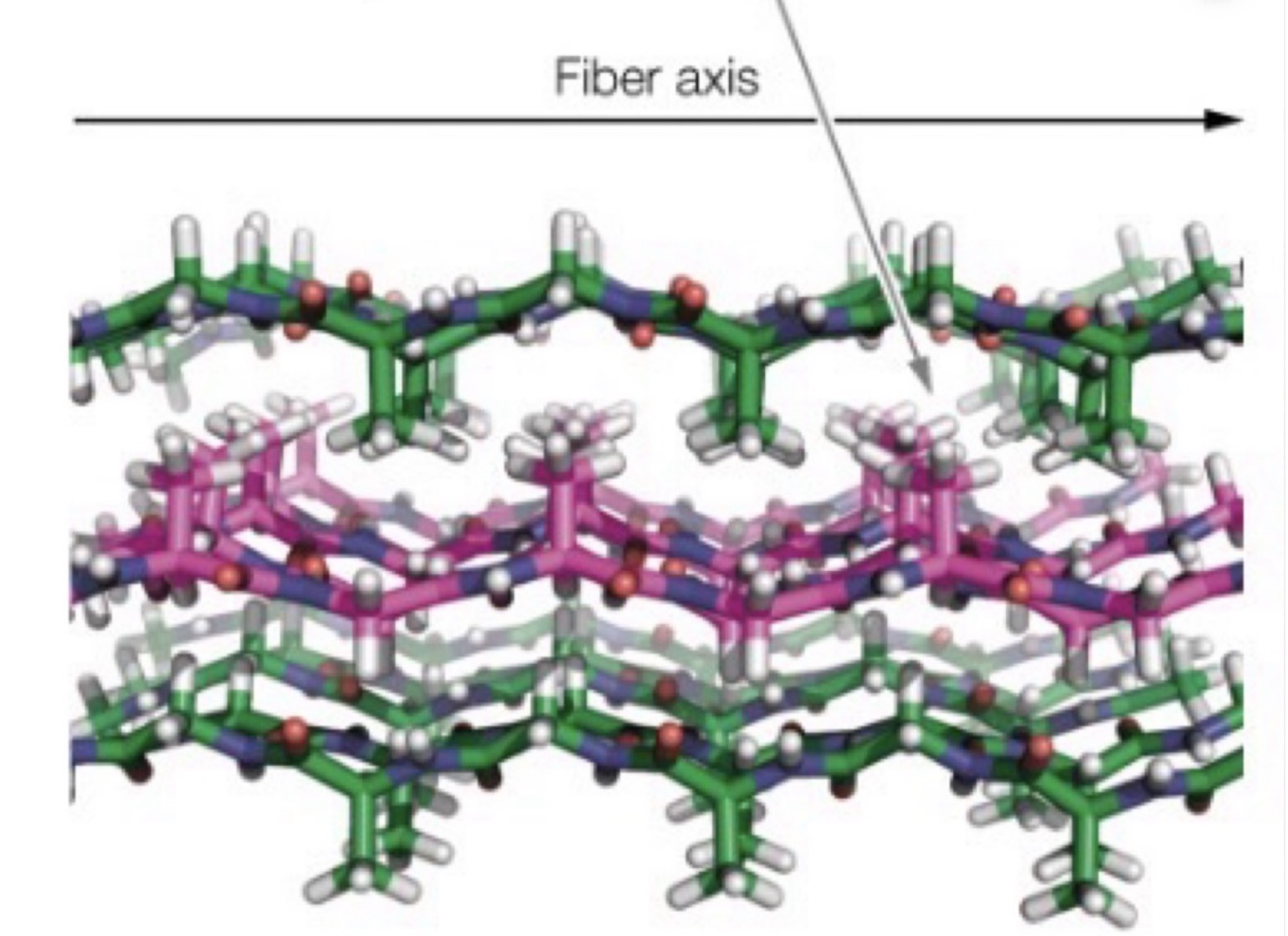
What is this?
A three-dimensional model of the stacked B sheets of fibroin showing alternating layers of interdigitating alanine or glycine side chains.
Three sheets are shown with H atoms in gray, N atoms in blue, O atoms in red, and C atoms in either green or magenta.
Fibers are very flexible because bonding between the sheets involves only the weak van der Waals
interactions between the side chains.